Mini-review: The Mucosal Immune Response in Health and Disease
The mucosal surfaces of the body are designed to provide resistance to microbial infection. They are also home to complex microbial ecosystems, known as the microbiota. Higher organisms have evolved a unique mucosal immune system, that interacts with the commensal microbiota, to promote health as well as protect the vast mucosal surfaces of the body which include the gut, the skin, nasal and oral cavities and the female reproductive tract. This mini-review provides a structural overview of mucosal immunity, specifically in the gut, and outlines how our understanding of the mucosal immune system is driving therapy for various diseases.
Introduction to mucosal immunity
The immune system can be considered a complex and diverse system designed to protect the body from dangerous pathogens. The mucosal surfaces, which can exceed 300 m2 in humans, are particularly vulnerable to infection (McGhee and Fujihashi 2012). Therefore, higher mammals have evolved a distinct mucosal immune system (MIS) for protection against toxic elements that enter the body through mucous membranes. The MIS comprises the largest immune organ in the human body (O’Hara and Shanahan 2006). It can be viewed as a single layer epithelium covered by mucus and anti-microbial proteins that is reinforced by various aspects of innate and adaptive immunity (McGhee and Fujihashi 2012). Within the MIS, there is a community of commensal, symbiotic and pathogenic microorganisms, called the microbiota, that share space with the host in areas such as the gut, skin, nasal and oral cavities as well as the female reproductive tract. The human large intestine contains the largest amount of microbes at approximately 1012 bacteria/ cm3 (Nicholson et al. 2012). This intestinal microbiota varies between individuals, and plays key roles in defense against pathogens as well as food digestion and nutrition (Janeway et al. 2001).
In its simplest form, the MIS can be divided into inductive and effector sites based on their anatomical and functional properties (Figure 1). The mucosal inductive sites are collectively called mucosa-associated lymphoid tissue (MALT) and include gut-associated lymphoid tissues (GALT), nasopharyngeal-associated lymphoid tissue (NALT) and lymphoid sites. The MALT provides a continuous source of memory B and T cells that then move into effector sites (McGhee and Fujihashi 2012). Mucosal effector sites include the lamina propia regions of the gastrointestinal, upper respiratory and reproductive tracts as well as secretory glandular tissues. These sites contain antigen-specific mucosal effector cells such as IgA-producing plasma cells, and memory B and T cells (McGhee and Fujihashi 2012).
Much of our current understanding of the MIS and the interaction of the host immune system with its microbiota has evolved from studies involving germ-free animals in which the impact of a single microbe species or defined species mixtures can be examined (Hooper et al. 2012). High throughput genomic, transcriptomic and metabolomic tools have also expanded our knowledge of the complexity of the microbiota and how this impacts systemic and mucosal immunity. These methods have provided significant insight into how the mucosal immune system is regulated as well as how to apply these findings for effective therapy. This mini-review explores these areas of mucosal immunity providing the latest information in this evolving area of biology. Particular emphasis is placed on intestinal mucosal immunity, as the gut mucosa is the largest mucosal immune site in the body, and is the most widely studied.
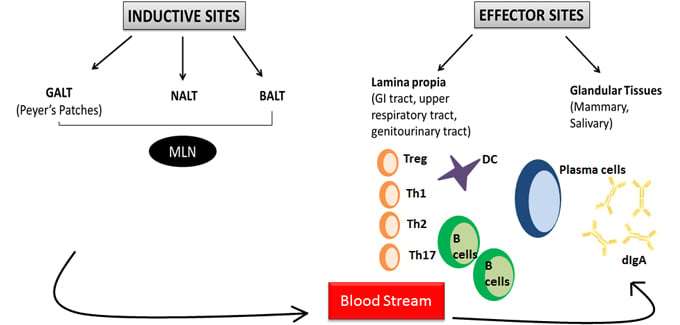
Fig. 1. Overview of the mucosal immune system. Adapted from McGhee JR and Fujihashi K 2012. The mucosal immune system (MIS) is divided into inductive and effector sites. Both sites are interconnected thus enabling the MIS to protect vast surface areas. GALT, gut-associated lymphoid tissues; NALT, nasopharyngeal- associated lymphoid tissue; BALT, bronchous-associated lymphoid tissue; MLN, mesenteric lymph nodes.
Mucosal immunity in the gut
Scientists have long since known of the microbial nature of the human body. Louis Pasteur and Harvey Cushing wrote about these concepts in the late 19th and early 20th centuries, both speculating on whether it would be possible to separate the microbes from the host (Cushing and Livingood 1900, Pasteur 1885, Macpherson and McCoy 2014). We now know that this would be impractical as microbes are beneficial to the host in many ways. They play keys roles in providing protection against pathogenic bacteria in the gut, and also serve a nutritional role by synthesizing key vitamins or their components (Janeway et al. 2001). However, these microbes can also be detrimental to the host under certain circumstances (Casadevall and Pirofski 2000).
It is estimated that 1014 microbes live within the human lower intestines (Macpherson and McCoy 2014). Accordingly, the MIS has evolved mechanisms to avoid deleterious responses from food antigens, to detect and destroy pathogenic organisms gaining entry to the body through the gut and to harness the beneficial effects of commensal microbes (Janeway et al. 2001).
Because of these diverse functions, the gut mucosa is known to be the largest immunological environment of the body. It employs a number of key immune cells and molecules to maintain immune homeostasis and protect against prolonged inflammation, which can lead to inflammatory bowel diseases (IBD) and other intestinal diseases. Below we describe the structure of the gut immune system and its immunoregulatory network.
Organization of the gut immune system
The intestinal surface of the gut is covered with finger-like projections known as villi, which play a predominant role in nutrient absorption. These villi and the underlying tissue also host the body’s largest population of immune cells (McGhee and Fujihashi 2012). Along the intestinal mucosa are also dome-like structures called Peyer’s patches. These are filled with lymphoid tissue, and thus are the key sites for coordinating immune responses to pathogens in the gut, as well as maintaining tolerance to food and commensal bacteria. The villi of the gut contain a network of blood vessels that transport nutrients from food to the rest of the body. Lymphatics from the villi and Peyer’s patches drain into the mesenteric lymph node.
Within villi are loose connective tissue called the lamina propia and at the base of the villi are crypts which contain stem cells that can replenish the epithelium. The epithelium and its thick overlying mucus form a protective layer against microbial invasion. Immune cells can be found inside the matrix of the Peyer’s patches, and include T and B cells, macrophages and dendritic cells (DCs). Peyer’s patches also contain specialized phagocytic cells known as M (microfold) cells, which transport macromolecules across the epithelial barrier, through a process called transcytosis, and deliver them to underlying DCs. The structure of the gut MIS is depicted in figure 2.
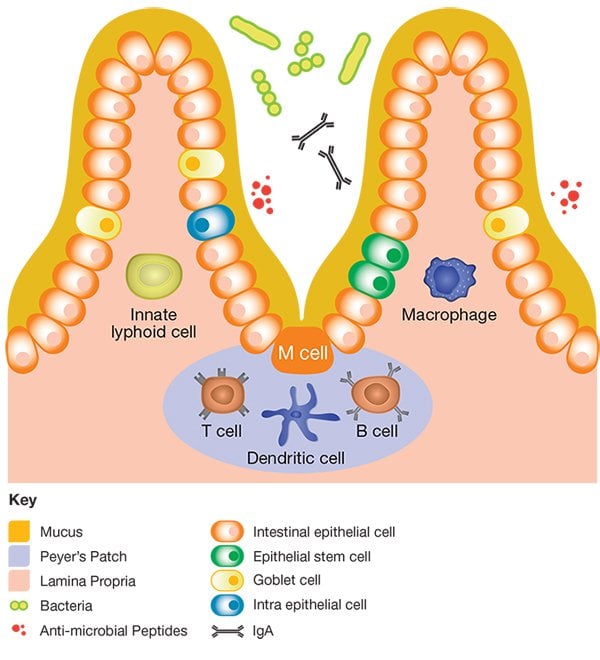
Fig. 2. The structure of the gut MIS
Regulation of gut immune homeostasis and tolerance
Similar to their role in other parts of the body, DCs are critical for maintaining immune homeostasis within the gut. They sample antigens between epithelial cells and process and present these antigens to cognate T cells. This results in the activation of an anti-inflammatory response, or tolerogenic activation, that aids in the maintenance of immune homeostasis. The DCs then travel to the T cell zones of the Peyer’s patches where they present antigen to T-cells and stimulate their differentiation to T regulatory cells (Tregs). Disruption of Treg function is associated with IBD in animals and humans as immune tolerance is perturbed (Mizrahi and llan 2009). T cells then migrate to the lamina propia of the villi through the lymphatic system, where they secrete the immunosuppressive cytokine Interleukin (IL)-10. IL-10 suppresses the function of immune cells in the lamina propia and the epithelial layer itself. Therefore, it is necessary for maintaining immune quiescence and preventing unnecessary inflammation.
The Wnt-β-catenin pathway in intestinal DCs also plays a role in maintaining immune tolerance in the gut. β-catenin expression in intestinal DCs induces the expression of anti-inflammatory mediators such as retinoic acid, IL-10 and transforming growth factor (TGF)-β (Santhakumar et al. 2010). Activation of β-catenin in intestinal DCs was also shown to induce Treg function and suppress the function of inflammatory effector T cells (Santhakumar et al. 2010).
Table 1 highlights the cells and structures in the gut MIS and outlines their function and specific location.
Gut Mucosal Immune Structures and Cells |
Associated Markers |
Function |
Location |
|---|---|---|---|
|
Villi |
N/A |
Houses self-renewing population of epithelial cells |
Projections of the gut wall that project into the lumen |
|
Peyer's patches |
N/A |
Key sites for coordinating immune responses to pathogens in the gut |
Component of GALT. Located throughout the Ileum region of the small intestine |
|
Lamina propia |
N/A |
Contains lymphatic network that absorbs digestive products. Also hosts many immune cells and is consequently the site of immune responses |
Located beneath the epithelium |
|
Crypts |
N/A |
Houses replicating stem cells, Paneth cells and goblet cells |
Invaginations of the epithelium around the villi. Lie within a lamina propia in small intestines |
|
Mesenteric lymph nodes |
N/A |
Important for initiating immune response to pathogens in the gut, specifically adaptive immune response |
Between the layers of the small intestine |
|
Stem cells in crypts |
Lgr5+, CD44+, CD24lo, CD166+, GRP78lo/- (only in mouse), CD117- (only in mouse) |
Continuously divides to provide all the epithelial cells in the crypts and on the villi. Induces the active self-renewal of the epithelium |
Located at the bottom of the crypt between Paneth cells |
|
Paneth cells |
IL-22R+, CD24lo, p-STAT3+ |
Produce anti-microbial peptides such as human beta-defensin |
Intestinal crypts |
|
Goblet cells |
CD117+ (on a subset in the colon), Muc2, Spdef, Krt20, Slc26a3 |
Secrete the mucus layer that protects the epithelium |
Intestinal crypts |
|
Intestinal effector T cells |
α4β7, CCR9+ |
Mediators of immune homeostasis, protection against pathogens |
Lamina propia, mesenteric lymph nodes, Peyer’s patches |
|
Intestinal T regulatory cells |
Suppresses immune response to commensal microbes. Maintains intestinal immune homeostasis |
Lamina propia, mesenteric lymph nodes, Peyer’s patches |
|
|
Intestinal B cells |
Primary source of IgA antibodies in the lamina propia |
Lamina propia, mesenteric lymph nodes, Peyer’s patches |
|
|
Intestinal dendritic cells (DCs) |
CD11c+, CD11b- (a subset expresses this marker), CD103+, CD80+ |
Selectively regulates T cell homing to the small intestine. Present microbial peptides to T cells for priming and activation |
Lamina propia, mesenteric lymph nodes, Peyer’s patches |
|
Intestinal macrophages |
Regulate inflammatory responses to bacteria, protect mucosa against harmful pathogens and scavenge dead cells and foreign debris |
Sub-epithelial lamina propia |
|
|
Microfold (M) cells |
Glycoprotein 2 (GP2)+, α (1,2)-fucose-containing carbohydrate moiety (NKM 16-2-4)+ |
Specialized epithelial cells that take up antigen from the lumen of the intestine and deliver them to the MALT |
M cells reside above Peyer’s patches |
|
Intestinal epithelial cells (IECs) |
Secrete anti-microbial peptides, secrete cytokines in response to microbes, recruit DCs, directly present antigens to T cells. Barrier function and maintenance of immune homeostasis |
Lining of the small intestine |
|
|
Innate lymphoid cell type 2 (ILC 2) |
CD127, ST2 (IL-33R), CRTH2 (only in humans), CD117 (only in mouse) |
Induces mucus production by goblet cells. Contributes to the immune response to helminth worms in the gut |
Lamina propia |
|
Innate lymphoid cell type 3 (ILC 3) |
Involved in the development of intestinal lymphoid organs. Involved in maintenance of gut immune homeostasis and response to pathogens |
Lamina propia |
Table 1. Structures and cells of the intestinal mucosal immune system
GALT, gut associated lymphoid tissue; MHC II, major histocompatibility complex class II
Gut MIS response to pathogenic infection
Dysregulation or a breakdown of homeostasis in the gut can lead to intestinal inflammation if the disruption is prolonged and uncontrolled. Disruption of the epithelial barrier either by mechanical, chemical or pathogenic factors can induce inflammation. Genetic susceptibility can also play a role in inducing inflammation in the gut (Bouma and Strober 2003). Upon encounter with bacteria, the epithelium becomes activated, leading to bacterial influx. It then releases alarmins such as IL-25, which activate immune cells. Since their main function is to initiate an immune response, alarmins are constitutively available and passively secreted upon tissue or cellular damage.
Surrounding Tregs restrict their secretion of the anti-inflammatory cytokine IL-10, thus enabling an immune response to proceed. DCs also become activated and secrete key inflammatory cytokines such as IL-6, IL-12 and IL-23. Effector T cells further escalate the immune response to bacteria by secreting other inflammatory cytokines, namely tumor necrosis factor-alpha (TNF-α), interferon-gamma (IFN-γ) and IL-17. Neutrophils are then recruited, as they are critical for the clearance of bacteria. They undergo a unique form of pathogen induced cell death known as NETosis. This cell death mechanism refers to the ability of neutrophils to produce neutrophil extracellular traps (NETs), which are chromatin structures filled with anti-microbial molecules. The NET kills the bacteria but also causes collateral damage to tissues in the process. Any remaining neutrophils die by apoptosis once the majority of bacteria are removed, and they are cleared by macrophages. Epithelial integrity is then restored by the replacement of damaged cells with new cells from the stem cells of the intestinal crypts. Tregs are then recruited to calm the immune response.
Role of innate lymphoid cells in gut pathogen defense and homeostasis
The recently discovered innate immune lymphoid cells (ILCs) are critical for orchestrating immune responses and maintaining homeostasis. They are divided into 3 subsets referred to as ILC1-3, in which ILC2 and ILC3 play key roles in maintaining gut immune homeostasis (Sonnenberg 2014). In the MIS, ILC3s interact with DCs to maintain the gut epithelial barrier. Upon encountering antigen from the gut microbiota, DCs secrete IL-23, which stimulates ILC3s to produce IL-22 (Sonnenberg et al. 2011). IL-22 then activates the epithelium, leading to the secretion of anti-microbial peptides that kill bacteria directly. IL-22 also enhances IL-23 production by DCs, and it is this cytokine-mediated interplay between ILC3s and DCs that protects the gut epithelial barrier from pathogenic bacteria.
ILC3s also interact with macrophages to induce gut immune homeostasis. Antigens from the gut microbiota induce IL1- beta secretion by macrophages, and this triggers the secretion of Granulocyte macrophage colony - stimulating factor (GMCSF) in ILC3s. In a feedback loop, GMCSF then signals macrophages to induce retinoic acid, which stimulates the differentiation of Tregs (Mortha et al. 2014).
ILC2s on the other hand, contribute to the response to helminths (parasitic worms such as nematodes; pinworms and hookworms). In response to the alarmin IL-25, ILC2s produce proteins that induce mucus production from goblet cells. This also initiates the activation of DCs that prime effector T cells and recruit mast cells and eosinophils, which induce muscle contraction and thus expulsion of the helminth worms. ILC2s are also involved in tissue repair following clearance of the helminth worms. In response to IL-33, they secrete amphiregulin which is involved in restoring the damaged tissue (Monticelli et al. 2015). To learn more about ILCs, read our mini-review on these interesting immune cells.
Inducing mucosal immunity for treating disease
Because the MIS is so critical for protecting the body from pathogenic invasion and maintaining homeostasis at mucosal surfaces, it is understandable that dysregulation of key components of the MIS is associated with disease. Some conditions linked to MIS dysfunction include autoimmune diseases, IBD and allergies as well as a variety of infectious diseases. The current knowledge of the function of the MIS can be exploited to develop effective therapies for treating these diseases. Below we discuss therapeutic strategies based on vaccination and protein and cellular targets that have been approved for disease treatment or are currently in development.
Vaccination
Vaccines currently play an important role in the control of a variety of infectious diseases. A large number of pathogens enter the body through the aerodigestive and reproductive tract; therefore, vaccines designed to induce both mucosal immune responses and systemic immunity can induce broad ranging protection against infectious agents. Mucosal vaccines currently licensed for human use include oral vaccines against Vibrio cholera, Salmonella typhi, poliovirus and rotavirus, as well as nasal vaccines for treating influenza (Azegami et al. 2014).
These vaccines function by interacting directly with the MIS. M cells in the follicle-associated epithelium (FAE) typically take up antigens delivered by mucosal vaccines from the lumen of the digestive or respiratory tract. These vaccine antigens are then transferred to antigen presenting cells (APCs) that reside below the FAE, such as DCs. The antigens are then processed and presented to CD4 and CD8 T cells at inductive sites (Holmgren and Czerkinsky 2005). In the GALT, orally administered antigens induce effector or memory cells that express the integrin α4β7. These activated lymphocytes are imprinted with specific gut-homing molecules associated with the integrin and chemokine systems. For example, Chemokine (C-C motif) ligand 25 (CCL25) is selectively present in the small intestine, where it is involved in the intestinal homing of IgA-committed B cells and plasma cells (Hieshima et al. 2004). When vaccine antigens are administered intranasally, antigen sensitized lymphocytes in the NALT express the α4β1 integrin, a receptor for vascular cell adhesion molecule 1 (VCAM-1) (Azegami et al. 2014).
Mucosal vaccines have been shown to induce rapid immune responses, typically between 48-72 h post-inoculation (Azegami et al. 2014), suggesting that they are likely beneficial in preventing the spread of endemic infections. However, they generally contain killed or attenuated micro-organisms that possess toxin-like molecules, which can lead to adverse immune responses. Therefore, further studies are needed to improve the safety of mucosal vaccines through the development of delivery technologies such as plant-based vaccines (Azegami et al. 2014).
Targeting key immune cells and proteins
Key diseases mediated by dysregulation of the MIS include IBD. IBD describes Crohn’s disease (CD) and ulcerative colitis (UC), which are chronic diseases that lead to structural damage with destruction of the bowel wall (Neurath 2014). The signaling pathways that control wound healing, intestinal inflammation and barrier function in IBD involve intestinal epithelial cells (IECs), goblet cells and Paneth cells that receive signals from the local microenvironment of the gut. The signals are mediated by cytokines, toll-like receptor (TLR) ligands and growth factors that induce apoptosis, proliferation or cell expansion, respectively. Defects in goblet cells lead to impaired mucin production, and alterations in Paneth cells lead to reduced production of anti-microbial peptides such as defensins (Neurath 2014). Consequently, IBD is characterized by an inflamed mucosa and alterations in barrier functions, which is predominantly mediated by IECs.
Healing of the inflamed mucosa and restoration of barrier functions are emerging goals for therapy of IBD. In IBD, structural changes in IECs are induced by proinflammatory cytokines such as tumor necrosis factor (TNF), IFN-γ, and IL-13, which are upregulated in the inflamed mucosa. Therefore, anti-TNF antibody has been applied to the treatment of IBD and has been shown to improve IEC-mediated barrier function in IBD patients (Zeissig et al. 2004). However, many patients do not respond to anti-TNF therapy, thus prompting the exploration of new targets to treat IBD (Neurath 2014). Current strategies being investigated include the application of anti-cytokine agents such as antibodies to IL-13, IL-6R, IL-21 and IL-12/IL23, all of which contribute to the chronic inflammation observed in IBD (Siegmund et al. 2001, Stolfi et al. 2011, Kawashima et al. 2011). Other methods have focused on increasing Tregs and anti-inflammatory cytokines in IBD, JAK inhibition and TLR 9 stimulation (Neurath 2014). Inhibition of T cell homing via the integrins α4β7 and the addressin mucosal vascular addressin cell adhesion molecule 1 (MAdCAM-1) have also been investigated (Neurath 2014). While treatments based on these studies are not currently in clinical application, it is expected that they will have a significant impact on the future therapy of IBD patients.
Because they play protective roles in the MIS, dysregulated ILC function or alterations in the composition of intestinal ILC populations can also induce IBD and even intestinal cancer (Sonnenberg 2014). Studies indicate that dysregulation or expansion of pro-inflammatory ILC populations may directly promote UC through IL-17, IL-22, TNF-α and IFN-γ production (Sonnenberg 2014, Goldberg et al. 2015). However, the exact mechanisms that promote their dysregulation of their function or composition in the gut have not yet been fully elucidated.
Sustained activation of ILCs due to chronic infection or impaired intestinal barrier function has been shown to induce the development and progression of cancer within the GI tract such as colorectal and gastric cancers, as well as hepatic carcinoma (Sonnenberg 2014, Vallentin et al. 2015). ILC3 production of IL-22 has been implicated as a key factor in the maintenance of cancer in a mouse model of colon cancer induced by bacteria (Goldberg et al. 2015). IL-17 production by ILC3s has also been shown to be required for tumorigenesis (Chan et al. 2014). Collectively, these studies indicate that ILCs are novel targets for the treatment of both intestinal cancers and IBD. Accordingly, therapeutic strategies to restore the proper function of these cells in the MIS are currently being explored (Goldberg et al. 2015).
The MIS is an essential compartment of the overall immune system and plays a key role in maintaining health. However, disruption of its function results in a number of diseases. Further understanding of the MIS is currently an active area of research, and it is hoped that as our knowledge increases, so will the ability to treat diseases caused by MIS dysfunction.
References
- Azegami T et al. (2014). Challenges in mucosal vaccines for the control of infectious diseases. Int Immunol 26, 517-528.
- Bouma G and Strober W (2003). The immunological and genetic basis of inflammatory bowel disease. Nat Rev Immunol 3, 521-533.
- Casadevall A and Pirofski L (2000). Host-pathogen interactions: Basic concepts of microbial commensalism, colonization, infection and disease. Infect Immun 68, 6511-6518.
- Chan IH et al. (2014). Interleukin-23 is sufficient to induce rapid de novo gut tumorigenesis, independent of carcinogens, through activation of innate lymphoid cells. Mucosal Immunol 7, 842-856.
- Cushing H and Livingood LE (1900). Experimental and surgical notes upon the bacteriology of the upper portion of the alimentary canal, with observations on the establishment there of an amicrobic state as a preliminary to operative procedures on the stomach and small intestine. John’s Hopkins Hospital Rep 9, 543-549.
- Goldberg R et al. (2015). The unusual suspects - innate lymphoid cells as novel therapeutic targets in IBD. Nat Rev Gastroenterol Hepatol 12, 271-283.
- Hieshima K et al. (2004). CC chemokine ligands 25 and 28 play essential roles in intestinal extravasation of IgA antibody-secreting cells. J Immunol 173, 3668-3675.
- Holmgren J and Czerkinsky C (2005). Mucosal immunity and vaccines. Nat Med 11, S45-S53.
- Hooper LV et al. (2012). Interactions between the microbiota and the immune system. Science 336, 1268-1273.
- Janeway CA Jr et al. (2001). Immunobiology: The Immune System in Health and Disease. 5th edition. (New York: Garland Science).
- Kawashima R et al. (2011). Interleukin-13 damages intestinal mucosa via TWEAK and Fn14 in mice- a pathway associated with ulcerative colitis. Gastroenterology 141, 2119-2129.
- Macpherson AJ and McCoy KD (2014). Standardised animal models of host microbial mutualism. Mucosal Immunol 8, 476-486.
- McGhee JR and Fujihashi K (2012). Inside the Mucosal Immune System. PLoS Biol 10, e1001397.
- Mizrahi M and llan Y (2009). The gut mucosa as a site for induction of regulatory T-cells. Curr Pharm Des 15, 1191-1202.
- Monticelli LA et al. (2015). IL-33 promotes an innate immune pathway of intestinal tissue protection dependent on amphiregulin-EGF R interactions. Proc Natl Acad Sci USA 112, 10762-10767.
- Mortha A et al. (2014). Microbiota-dependent crosstalk between macrophages and ILC3 promotes intestinal homeostasis. Science 343, 1249288.
- Neurath MF (2014). New targets for mucosal healing and therapy in inflammatory bowel diseases. Mucosal Immunol 7, 6-19.
- Nicholson JK et al. (2012). Host-gut microbiota metabolic interactions. Science 336, 1262-1267.
- O’Hara AM and Shanahan F (2006). The gut as a forgotten organ. EMBO Rep 7, 688-693.
-
Pasteur L (1885). Observations relatives à
ala note de M. Duclaux. Compt Rend 100, 69. - Santhakumar M et al. (2010). Activation of β-catenin in dendritic cells regulates immunity versus tolerance in the intestine. Science 329, 849-853.
- Siegmund B et al. (2001). Neutralization of interleukin-18 reduces severity in murine colitis and intestinal IFN-gamma and TNF-alpha production. Am J Physiol Regul Integr Comp Physiol 281, R1264-R1273.
- Sonnenberg GF et al. (2011). Border patrol: regulation of immunity, inflammation and tissue homeostasis at barrier surfaces by IL-22. Nat Immunol 12, 383-390.
- Sonnenberg GF (2014). Regulation of intestinal health and disease by innate lymphoid cells. Int Immunol 26, 501-507.
- Stolfi C et al. (2011). Involvement of interleukin-21 in the regulation of colitis associated colon cancer. J Exp Med 208, 2279-2290.
- Vallentin B et al. (2015). Innate Lymphoid Cells in Cancer. Cancer Immunol Res 3, 1109-1114.
- Zeissig S et al. (2004). Downregulation of epithelial apoptosis and barrier repair in active Crohn’s disease by tumour necrosis factor alpha antibody treatment. Gut 53, 1295-1302.








