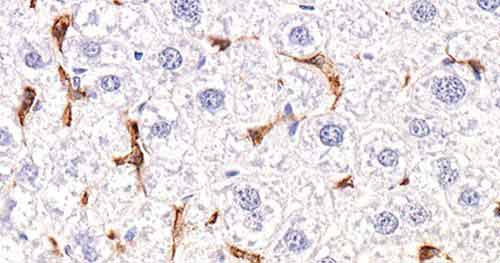
Mini-review: Anti-F4/80 Antibody | Clone CI:A3-1
F4/80: The marker and the antibody
F4/80 is a well-characterized and extensively referenced membrane protein and is the best known mature mouse macrophage and microglial marker. The anti-F4/80 antibody clone CI:A3-1 is the original monoclonal produced against the F4/80 antigen in Siamon Gordon’s laboratory at the University of Oxford (Gordon and Austyn, 1981). Today, Bio-Rad is the only commercial manufacturer of the original clone for this important monoclonal macrophage antibody.
Thirty formats/sizes of F4/80 antibody products are currently available, from purified and low endotoxin versions to a wide range of popular fluorescent labels. These fluorescent labels are suitable for flow cytometry and include choices such as FITC, RPE, DyLight® and Alexa Fluor® dyes, as well as the exclusive tandem conjugates RPE-Alexa Fluor® 647 and RPE-Alexa Fluor® 750.
Characteristics of the anti-F4/80 antibody clone CI:A3-1
In addition to recognizing kupffer cells, langerhans cells, peritoneal macrophages, and splenic red pulp macrophages, the anti-F4/80 antibody clone CI:A3-1 is distinguished from other F4/80 monoclonal antibodies in that it stains microglia (macrophages of the central nervous system) (Lawson et al. 1990). This may indicate that clone Cl:A3-1 recognizes a different F4/80 epitope from other F4/80 antibody clones.
In addition to its extensive use in flow cytometry, clone Cl:A3-1 is also suitable for functional studies and is available in a low endotoxin format for this purpose. The rat anti-mouse F4/80 antibody clone CI:A3-1 is also paraffin-reactive, allowing for immunohistochemical studies of paraffin-embedded sections of brain and other tissues.
Hundreds of scientific references support the use of the antibody clone Cl:A3-1 in a wide range of applications and for a variety of experimental studies. Despite intensive investigation, however, the function of the F4/80 antibody target protein is not fully understood.
F4/80: Gene and protein
F4/80 is known under several alternate names: Ly71, Gpf480, TM7LN3, DD7A5-7, EGF-TM7, and EMR1.
The F4/80 gene is located on mouse chromosome 17 and encodes a polypeptide of 931 amino acids that is processed into a mature protein of 904 amino acids. The predicted mass of the protein is 98.9 kDa, but the F4/80 protein runs at an apparent weight of 160 kDa in SDS-PAGE. The mass difference is probably accounted for by extensive glycosylation. Two papers describe the early cloning of the gene and the characterization of the protein in 1996 and 1997, McKnight et al. and Lin et al. respectively.
The N- terminus of the F4/80 polypeptide contains seven tandem EGF-like domains and shows a high degree of homology to proteins such as fibrillin-1 and fibulin-2. The C- terminal sequence demonstrates homology to members of the TM7 superfamily, such as the G protein-coupled receptors for peptide hormones, e.g. parathyroid hormone, calcitonin, and glucagon (McKnight et al. 1996).
The later discovery of other proteins with the same domain arrangement defined a new protein family named EGF-TM7, whose members include F4/80, EMR1 (the human F4/80 homolog), and human CD97 (McKnight et al. 1998).
The presence of an adhesion and a signaling domain in the F4/80 sequence suggests that it functions as signaling molecule upon binding with other as yet unidentified cell-surface proteins. Ligands identified for other members of the EGF-TM7 protein family support this hypothesis, making it reasonable to conclude that F4/80 could have similar adhesion and signaling properties (McKnight et al. 1998).
|
|
F4/80: Functions
F4/80 knockout mice appear vital, healthy, and fertile. The development and anti-microbe activity of macrophages in these animals also seems to be normal (Schaller et al. 2002).
However, Warschkau and Kiderlen (1999) have shown that administration of the anti-F4/80 antibody inhibits the production of cytokines (namely TNFa, IL-12, and IFN) from whole spleen cell cultures of SCID mice exposed to heat killed Listeria monocytogenes. Modulated cytokine response is believed to be dependent on the interaction of NK cells and macrophages in culture.
F4/80 is also expressed by eye-derived antigen-presenting cells (APCs) involved in anterior chamber-associated immune deviation (ACAID), a process that elicits tolerance for an antigen inoculated into the eye. ACAID is known to be antigen specific (Wang et al. 2001). Lin et al. (2005) went on to demonstrate the involvement of F4/80 in ACAID, in particular in the generation of efferent T regulatory (Treg) cells that are the effectors of tolerance.
Without knowing the cognate ligand of F4/80, Lin et al. (2005) could not describe a molecular mechanism for the involvement of F4/80 in ACAID, but they hypothesized that F4/80 is implicated in the interaction between tolerogenic APCs and NKT cells in the spleen that is essential for the development of the CD8+ Treg cells seen in ACAID.
In the same report, Lin et al. (2005) showed that F4/80 is also needed for CD8+ Treg cell induction in low-dose oral tolerance. Furthermore, F4/80 is expressed on APCs in the Peyer’s patches (Makala et al. 2003) and on the majority of dendritic cells (DCs) in the central nervous system (Suther et al. 2003). These findings suggest that F4/80 could be involved in tolerance in systems distinct from ACAID and could be an essential part of the interaction between NK cells,macrophages, and F4/80 expressing APCs.
F4/80: Expression
F4/80 is expressed at high levels on the surface of various macrophages: kupffer cells, splenic red pulp macrophages, microglia, gut lamina propria, and langerhans cells in the skin. Macrophages of the connective tissue, heart, kidney, reproductive, and neuroendocrine systems also express F4/80 (Gordon and Austyn, 1981).
Although F4/80 is broadly represented in macrophages, its expression varies depending upon the state of maturation, developmental processes, and type of macrophage. Langerhans cells are known to down regulate F4/80 expression after antigen uptake. This down regulation precedes migration to the local lymph nodes and is in accord with the hypothesized adhesion and signaling function of the protein (Gordon and Austyn, 1981).
Monocytes that circulate in the bloodstream also express F4/80 on the surface, but the level is lower than on tissue macrophages, indicating another correlation between the level of F4/80 and the adhesion properties of the corresponding mononuclear blood cell (Gordon and Austyn, 1981).
F4/80: Microglia
Clone CI:A3-1 ability to stain microglia is due a unique and advantageous feature of F4/80, as several other antigens, such as CD4, are down-regulated once they pass through the blood-brain barrier (Perry and Gordon, 1991).
F4/80+ microglia are found throughout white and grey matter. Separate regions of the parenchyma contain F4/80+ cells varying in morphology; this allows us to distinguish microglia from other macrophage populations in the choroid plexus and leptomeninges (Gordon et al, 1992). Lawson et al (1990) were able to produce a map of the distribution and morphology of microglia in the adult mouse CNS due to F4/80 labelling.
Microglia are associated with a number of disorders such as Alzheimer’s Disease, Multiple Sclerosis and Prion Disease, which means that CI:A3-1 is a key F4/80 antibody clone to use in neurological research.
Pow et al (1989), found that F4/80+ microglia in the posterior pituitary selectively endocytose terminals of neuroendocrine cells containing oxytocin/vasopressin, indicating that hormonal stimulation could be a functional response of F4/80+ microglia.
|
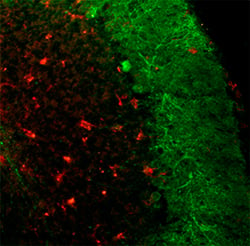
A
|
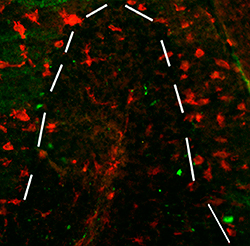
B
|
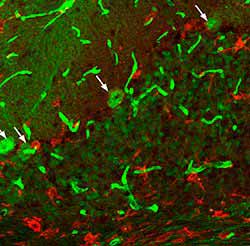
C
|
Figure 2. These images show sections from 50-d-old npc -1 mutant mice stained with anti-F4/80 antibody (red) with the aim of marking microglia. A) Shows an infiltration of microglia in the granule cell layer and white matter tract. B and C) Shows numerous microglia marked by anti-F4/80 present throughout the cerebellum, the dashed line indicating the edge of the granule cell layer (Dennis et al. 2005).
EMR1: The human homolog of F4/80
The human homolog of F4/80 was cloned and named EMR1 by Baud et al. (1995). EMR1 is located on chromosome 19 and its sequence shows 68% overall identity to F4/80. EMR1 also contains six EGFRepeats and seven transmembrane segments supporting a purported function in adhesion and signaling that is in line with members of the EGF-TM7 family.
Hamann et al. (2007) studied the expression and surface abundance of EMR1 using quantitative real-time PCR and flow cytometric analysis. They observed that EMR1 is surprisingly absent on mononuclear phagocytic cells, such as monocytes, macrophages, and myeloid dendritic cells. According to their report EMR1 expression is restricted to eosinophilic granulocytes, making it a highly specific eosinophil marker in humans.
While the absence of F4/80 on human monocytes and macrophages merits further investigation, the presence of F4/80 on eosinophils is not new to scientists. Murine eosinophils are known to stain with F4/80 and even Mac-1, but at lower intensity than is typical for macrophages and monocytes (McGarry et al. 1991). Bio-Rad offers anti human EMR1 antibody for the study of the human equivalent of F4/80.
References
-
Austyn, J.M. and Gordon, S. (1981) F4/80, a monoclonal antibody directed specifically against the mouse macrophage.
Eur. J. Immunol. 11: 805-815 -
Baud, V. (1995) EMR1, an unusual member in the family of hormone receptors with seven transmembrane segments.
Genomics. 20;26(2):334-44 -
Dennis Ko et al. (2005) Cell-Autonomous Death of Cerebellar Purkinje Neurons with Autophagy in Niemann-Pick Type C Disease.
-
Hamann, J. (2007) EMR1, the human homolog of F4/80, is an eosinophil-specific receptor.
Eur J Immunol. 37(10):2797-802 -
Hume DA et al. (1984c) The mononuclear phagocyte system of the mouse defined by immunohistochemical localisation of antigen F4/80. Macrophages of endocrine organs.
-
Lawson L.J et al. (1990) Heterogeneity in the distribution and morphology of microglia in the normal adult mouse brain.
-
Lin H.H et al. (1997) Identification and characterization of a seven transmembrane hormone receptor using differential display.
Genomics. 41(3):301-8 -
Lin H.H et al. (2005) The macrophage F4/80 receptor is required for the induction of antigen-specific efferent regulatory T cells in peripheral tolerance.
J Exp Med. 201(10):1615-25 -
Makala, L.H et al. (2003) Phenotype and function of murine discrete Peyer’s patch macrophage derived - dendritic cells.
J Vet Med Sci. 65(4):491-9 -
McGarry M.P. and Stewart C.C. (1991) Murine eosinophil granulocytes bind the murine macrophage-monocyte specific monoclonal antibody F4/80.
J Leukoc Biol. 1991 50(5):471-8 -
McKnight A.J et al. (1996) Molecular cloning of F4/80, a murine macrophage-restricted cell surface glycoprotein with homology to the G-protein-linked transmembrane 7 hormone receptor family.
J Biol Chem. 271(1):486-9 -
McKnight, A.J. and Gordon, S. (1998) The EGF-TM7 family: unusual structures at the leukocyte surface.
J Leukoc Biol 63(3):271-80 -
Perry VH, Gordon S (1991) Macrophages and the nervous system.
-
Pow DV et al. (1989) Microglia in the neurohypophysis associate with and endocytose terminal portions of neurosecretory neurons.
-
Gordon S et al. (1992) Antigen Markers of Macrophage Differentiation in Murine Tissues.
-
Schaller E et al. (2002) Inactivation of the F4/80 glycoprotein in the mouse germ line.
Mol Cell Biol. 22(22):8035-43 -
Suther T et al. (2003) The brain as an immune privileged site: dendritic cells of the central nervous system inhibit T cell activation.
Eur J Immunol. 33(11):2998-3006 -
Wang Y et al. (2001) Blood mononuclear cells induce regulatory NK T thymocytes in anterior chamber-associated immune deviation.
J Leukoc Biol. 69(5):741-6 -
Warschkau H. and Kiderlen, A.F. (1999) A monoclonal antibody directed against the murine macrophage surface molecule F4/80 modulates natural immune response to Listeria monocytogenes.
J Immunol. 163(6):3409-16 -
Wilbanks G.A. and Streilein, J.W. (1991) Studies on the induction of anterior chamber-associated immune deviation (ACAID). 1. Evidence that an antigen-specific, ACAID-inducing, cell-associated signal exists in the peripheral blood.
J Immunol. 146(8):2610-7 -
Wilbanks G.A et al. (1991) Studies on the induction of anterior chamber-associated immune deviation (ACAID). II. Eye-derived cells participate in generating blood-borne signals that induce ACAID.
J Immunol.146(9):3018-24 -
Wilbanks G.A et al. (1992) Studies on the induction of anterior chamber-associated immune deviation (ACAID). III. Induction of ACAID depends upon intraocular transforming growth factor-beta.
Eur J Immunol. 22(1):165-73