Mini-review: The T Cell Marker, CD3 Antigen and Antibodies
Cluster of differentiation 3 (CD3) is a multimeric protein complex, known historically as the T3 complex, and is composed of four distinct polypeptide chains; epsilon (ε), gamma (γ), delta (δ) and zeta (ζ), that assemble and function as three pairs of dimers (εγ, εδ, ζζ). The CD3 complex serves as a T cell co-receptor that associates noncovalently with the T cell receptor (TCR) (Smith-Garvin et al. 2009). The CD3 protein complex is a defining feature of the T cell lineage, therefore anti-CD3 antibodies can be used effectively as T cell markers (Chetty and Gatter 1994).
T cell activation overview
TCRs cannot bind free epitopes/antigen; instead TCRs bind enzymatically cleaved fragments of larger polypeptides associated with major histocompatibility complexes (MHC), which is synonymous with the human leukocyte antigen (HLA) system in humans (Rudd 1990, Gao et al. 2002). This interaction occurs in a space that has become known as the immunological synapse. MHC class I molecules are expressed on all nucleated cells of the body and present antigens to CD8, a transmembrane glycoprotein expressed predominately on cytotoxic T cells. This activation of cytotoxic T cells subsequently results in the destruction of virally infected cells. MHC class II are found on macrophages, B cells and dendritic cells.
These immune cells present antigens to helper T cells with CD4 stabilizing the MHC/TCR interaction, which ultimately results in an antibody mediated immune response. Other co-stimulatory molecules, such as CD45, CD28 and CD2 aid in T cell activation in the immunological synapse and initiate the formation of the TCR signalosome, a macromolecular protein complex responsible for intracellular signaling. Read our TCR mini-review for detailed information on TCR activation, signaling, development and diversity.
TCR protein structure
TCRs are heterodimers composed of either an alpha and beta polypeptide chain, occurring in approximately 95% of the TCR population, or a gamma and delta polypeptide chain (Pitcher and van Oers 2003, Malissen 2008). Each polypeptide contains a constant (C) and variable (V) region (Figure 1). The constant region is anchored in the cell membrane, while the variable region extends extracellularly and is responsible for antigen binding. The short cytoplasmic tail of the TCR lacks the ability to signal, requiring intracellular signaling to be initiated by the CD3 protein complex.
CD3 protein structure
CD3 proteins have an N-terminal extracellular region, a transmembrane domain and a cytoplasmic tail where the immunoreceptor tyrosine activation motifs (ITAMs) are located. The extracellular domains of CD3 ε, γ and δ contain an immunoglobulin-like domain, so are therefore considered part of the immunoglobulin superfamily.
The intracellular ITAMs are characterized by a consensus amino acid sequence of YXXL/I X6-8 YXXL/I (where X represents any amino acid residue). The cytoplasmic segments of CD3 ε, CD3 γ and CD3 δ contain a single ITAM, whereas the cytoplasmic domain of the CD3 ζ subunit contains three ITAMs, resulting in ten ITAMs in total, making the complex exquisitely sensitive to antigen binding (Figure 1).
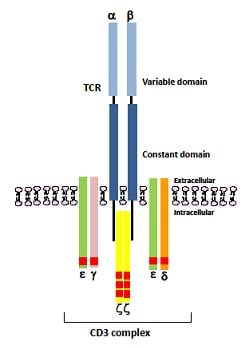
Fig. 1. A schematic representation of a TCR heterodimer comprising of an alpha (α) and beta (β) polypeptide chain with each polypeptide containing a constant and a variable region. The CD3 protein is composed of three pairs of dimers (εγ, εδ, ζζ) that are responsible for intracellular signaling, initiated by the phosphorylation of immunoreceptor tyrosine activation motifs (ITAMs) shown as red rectangular domains (ten in total).
CD3 genes and function
The CD3 protein complex was first identified by immunoprecipitation experiments using human T cells. The determination of glycosylation state of the individual CD3 ε, CD3 γ and CD3 δ polypeptide chains was subsequently determined using endoglycosaminidases and specific anti-CD3 antibodies. CD3 ε, CD3 γ and CD3 δ are highly homologous and are believed to be derived from a single ancestral gene, mapping to human chromosome 11 band q23, through gene duplication (Clevers et al. 1988).
CD3 ε is a non-glycosylated polypeptide chain of 20 kDa. The existence of an epitope on the ε polypeptide that is conserved among many species has made it possible to obtain the antibody clone, CD3-12 which has a very broad species cross-reactivity for the CD3 marker (Jones et al. 1993). Both CD3 γ and CD3 δ are glycosylated and have a molecular weight of 25-28 kDa and 20 kDa respectively (Norman 1995).
CD3 ζ (also known as CD247) is a non-glycosylated polypeptide with a molecular weight of 17 kDa that shares no sequence similarity with the other CD3 polypeptide chains. CD3 ζ was first discovered by virtue of the fact that it coprecipitates with the TCR complex. CD3 ζ maps to human chromosome 1 band q22-q25 underscoring the fact that CD3 ζ is a distinct genetic component of the CD3 protein complex (Weissman et al. 1988).
CD3 eta (η) is an alternatively spliced product of the gene that encodes CD3 ζ (Clayton et al. 1991) and has an apparent molecular weight of 23 kDa (Orloff et al. 1989). CD3 ζ is predominantly found as a homodimer, but a fraction (approximately 10%) of CD3 ζ is found complexed as a heterodimer with CD3 η and in this form is thought to confer different signaling properties (Orloff et al. 1989).
CD3 omega (Ω) is a non-glycosylated polypeptide chain of 28 kDa that associates with the TCR complex during TCR assembly in the endoplasmic reticulum, but does not associate with the TCR complex at the cell surface (Pettey et al. 1987, Clevers et al. 1988, Neisig et al. 1993). It is thought that CD3 Ω might facilitate complex assembly and/or be involved in the retention of unassembled CD3 protein complexes in the endoplasmic reticulum leading to their subsequent degradation in the lysosomes.
ITAMs are required for initiation of the signaling cascade as they recruit protein tyrosine kinases, signaling intermediates and adapter molecules. Other immune receptors, such as FcR and BCR, contain far fewer ITAMs than the TCR: CD3 complex, however, T cell development and activation is influenced by the number, location and type of ITAMs present within the complex (Guy and Vignali 2009). This suggests that the TCR is able to vary its signaling leading to diverse T cell responses.
Signal transduction pathways mediated by the CD3 protein complex
Following antigen stimulation conformational changes within the cytoplasmic tails of the CD3 polypeptides occur. This is induced by protein tyrosine kinases (PTKs) that trigger the MHC/TCR interaction. These PTKs belong to the Src family, such as Lck and Fyn, and phosphorylate conserved tyrosine residues present within the ITAMs in the CD3 complex, creating a docking site for proteins with Src homology 2 domains, e.g. ζ -associated protein of 70 kDa (ZAP-70 ) (Pitcher and van Oers 2003, Smith-Garvin et al. 2009).
Recruitment of ZAP-70, a syk kinase family member, binds to CD3-ζ. Bound ZAP-70 phosphorylates the transmembrane adapter protein linker of activated T cells (LAT) which then allows the cytosolic adapter protein SH2 domain containing leukocyte phosphoprotein of 76 kDa (SLP-76) to bind to it. These adaptors form the backbone of the signalosome complex that nucleates the subsequent activation of downstream effector molecules necessary for T cell activation.
Phosphorylated LAT binds to phospholipase C γ 1 (PLCγ1), the p85 subunit of phosphoinositide 3 kinase, growth factor receptor-bound protein 2 (GRB2) and the GRB2 related adaptor downstream of Shc (GRAP2). SLP-76 is then recruited to phosphorylated LAT by Gads. SLP-76 interacts with PLC γ 1 as well as Vav1, Nck, Il2 induced tyrosine kinase and adhesion and degranulation-promoting adapter protein (ADAP).
The formation of this signaling complex results in the activation of PLC gamma 1-dependent signaling pathways including calcium and diacylglycerol (DAG) induced responses, cytoskeletal rearrangements and integrin activation pathways that mediate cell to cell and cell to matrix interactions. The ligation of co-stimulatory molecules, such as CD28, enhances the activity of these pathways, while hematopoietic progenitor kinase-1 (HPK1), which associates with SLP-76, acts as a negative regulator of T cell activation.
CD3 protein expression
Initial expression of CD3 occurs in the cytoplasm in a peri-nuclear location of pro-thymocytes. As T cell maturation proceeds, cytoplasmic CD3 expression is lost and the CD3 antigen is found on the cell surface. Pro-thymocytes differentiate into common thymocytes, and then into medullary thymocytes, and it’s at this latter stage the CD3 antigen begins to migrate to the cell membrane.
The specificity of the CD3 antigen for T cells and its appearance at all stages of T cell development makes it an ideal T cell marker for both the detection of normal T cells and T cell neoplasms (lymphomas and leukemias), and makes it a useful immunohistochemical marker for T cells in tissue sections (Salvadori et al. 1994, Vernau and Moore 1999).
CD3 is also weakly expressed by some macrophages, Purkinje cells in the brain and by Hodgkin’s and Reed-Sternberg cells, both of which are cells found in Hodgkin’s lymphoma usually derived from cells of the B cell lineage.
Clinical applications for the CD3 protein complex and its role in disease
As mentioned above, the CD3 protein complex is an important T cell marker for the classification of malignant lymphomas and leukemias (T cell neoplasms). CD3 can also be used for the identification of T cells in coeliac disease (Leon et al. 2011), lymphocytic colitis and collagenous colitis (Mosnier et al. 1996, Sapp et al. 2002).
Animal studies have shown that anti-CD3 antibodies induce tolerance to allografts (Nicolls et al. 1993). OKT3, an anti-CD& antibody directed against CD3 ε, has been clinically approved for use in humans for the induction of immunosuppression in solid organ transplantation for the prevention and treatment of rejection (Norman 1995). Interestingly, susceptibility to type I diabetes has been associated with the CD3 ε genetic locus (Wong et al. 1991) and anti-CD3 antibodies have been shown to ameliorate the symptoms of this and other auto-immune disorders (Sprangers et al. 2011).
Defects in the CD3 genes are one of the causes of Severe Combined Immune Deficiency (SCID), which is an autosomal recessive disorder characterized by a severe defect in T cell production or function (de Saint Basile et al. 2004, Fischer et al. 2005, Roberts et al. 2007, Recio et al. 2007). Furthermore, the archetypal auto-immune disease, systemic lupus erythematosus (SLE) is associated with a deficiency in the CD3 ζ polypeptide chain (Takeuchi et al. 2012).
References
- Alarcon B et al. (1988) Assembly of the human T cell receptor CD3 complex takes place in the endoplasmic reticulum and involves intermediary complexes between the CD3γ, δ, ε core and single T-cell receptor a or b chains. Journal of Biological Chemistry 263, 2953-2961.
- Chetty R and Gatter K (1994) CD3: Structure, function and the role of immunostaining in clinical practice. Journal of Pathology 173, 303-307.
- Clayton LK et al. (1991) CD3 eta and CD3 zeta are alternatively spliced products of a common genetic locus and are transcriptionally and/or post-translationally regulated during T-cell development. Proceedings of the National Academy of Sciences of the USA 88, 5202-5206.
- Clevers H et al. (1988) The T cell receptor/CD3 complex: A dynamic protein ensemble. Annual Review of Immunology 6, 629-662.
- de Saint Basile G et al. (2004) Severe combined immunodeficiency caused by deficiency in either the delta or the epsilon subunit of CD3. Journal of Clinical Investigation 114, 1512-1517.
- Fischer A et al. (2005) CD3 deficiencies. Current Opinion in Allergy and Clinical Immunology 5, 491-495.
- Gao GF et al. (2002) Molecular coordination of the alphabeta T-cell receptors and co-receptors CD8 and CD4 in their recognition of peptide MHC ligands. Trends in Immunology 23, 408-413.
- Guy CS and Vignali DA (2009) Organisation of proximal signal initiation at the TCR:CD3 complex. Immunological Reviews 232, 7-21.
- Horejsi V et al. (1988) Monoclonal antibodies against human leucocyte antigens. II. Antibodies against CD45 (T200), CD3 (T3), CD43, CD10 (CALLA), transferring receptor (T9), a novel broadly expressed 18 kDa antigen (MEM-43) and a novel antigen of restricted expression (MEM-74). Folia Biologica (Prague) 34, 23-34.
- Jones M et al. (1993) Detection of T and B cells in many animal species using cross-reactive anti-peptide antibodies. Journal of Immunology 150, 5429-5435.
- Leo O et al. (1987) Identification of a monoclonal antibody specific for a murine T3 polypeptide. Proceedings of the National Academy of Sciences of the USA 84, 1374-1378.
- Leon F (2011) Flow cytometry of intestinal intraepithelial lymphocytes in celiac disease. Journal of Immunological Methods 363, 177-186.
- Malissen B (2008) CD3 ITAMs count! Nature Immunology 9, 583-584.
- Mosnier JF et al. (1996) Lymphocytic and collagenous colitis: an immunohistochemical study. American Journal of Gastroenterology 91, 709-713.
- Neisig A et al. (1993). Assembly of the T-cell antigen receptor. Participation of the CD3 omega chain. J Immunol 151, 870-879.
- Nicolls MR et al. (1993) Induction of long-term specific tolerance to allografts in rats by therapy with an anti-CD3 like monoclonal antibody. Transplantation 55, 459-468.
- Norman DJ (1995) Mechanisms of action and overview of OKT3. Therapeutic Drug Monitoring 17, 615-620.
- Orloff DG et al. (1989) Biochemical characterization of the eta chain of the T-cell receptor. A unique subunit related to zeta. Journal of Biological Chemistry 264, 14812-14817.
- Pettey CL et al. (1987) T3-p28 is a protein associated with the delta and epsilon chains of the T cell receptor-T3 antigen complex during biosynthesis. Journal of Biological Chemistry 262, 4854-4859.
- Pitcher LA and van Oers NSC (2003) T-cell receptor signal transmission: who gives an ITAM TRENDS in Immunology 24, 554-560.
- Recio MJ et al. (2007) Differential biological role of CD3 chains revealed by human immunodeficiencies. Journal of Immunology 178, 2556-2564.
- Roberts JL et al. (2007) T-B+NK+ severe combined immunodeficiency caused by complete deficiency of the CD3 zeta subunit of the T-cell antigen receptor complex. Blood 109, 3198-3206.
- Rudd CE (1990) CD4, CD8 and the TCR-CD3 complex: a novel class of protein tyrosine kinase receptor. Immunology Today 11, 400-406.
- Salvadori S et al. (1994) Abnormal signal transduction by T cells of mice with parental tumors is not seen in mice bearing Il-2 secreting tumors. Journal of Immunology 153, 5176-5182.
- Sapp H et al. (2002) The terminal ileum is affected in patients with lymphocytic or collagenous colitis. The American Journal of Surgical Pathology 26, 1484-1492.
- Satake Y et al. (2000) Characterization of rat OX40 ligand by monoclonal antibody. Biochemical and Biophysical Research Communications 270, 1041-1048.
- Smith-Garvin JE et al. (2009) T cell Activation. Annual Review of Immunology 27, 591-619.
- Sprangers B et al. (2011) Otelixizumab in the treatment of type 1 diabetes mellitus. Immunotherapy 11, 1303-1316.
- Takeuchi T et al. (2012) CD3 defects in systemic lupus erythematosus. Annals of Rheumatic diseases 71, 78-81.
- Tanaka T et al. (1989) Characterization of a CD-3 like rat T cell surface antigen recognized by a monoclonal antibody. Journal of Immunology 142, 2791-2795.
- Tomonari K (1988) A rat antibody against a structure functionally related to the mouse T-cell receptor/T3 complex. Immunogenetics 28, 455-458.
- Vernau W and Moore PF (1999) An immunophenotypic study of canine leukemias and preliminary assessment of clonality by polymerase chain reaction. Veterinary Immunology and Immunopathology 69, 145-164.
- Weissman AM et al. (1988) Molecular cloning and chromosomal localization of the human T-cell receptor zeta chain: distinction from the molecular CD3 complex. Proceedings of the National Academy of Sciences of the USA 85, 9707-9713.
- Wong S et al. (1991) Susceptibility to type I diabetes in women is associated with the CD3 epsilon locus on chromosome 11. Clinical and Experimental Immunology 83, 69-73.








