Anti-Idiotypic Antibodies
What is an Anti-Idiotypic Antibody?
When one antibody binds to an idiotope of another antibody it is referred to as an anti-idiotypic antibody. The variable part of an antibody including the unique antigen binding site is known as the idiotype. The combination of epitopes within the idiotype (i.e. the idiotopes) is unique for each antibody, figure 1.
Because most therapeutic monoclonal antibodies developed today are human or humanized, the most likely immunogenic epitopes for the induction of anti-drug antibodies (ADA) lie within the hypervariable complementarity determining regions (CDR) that provide the majority of the binding contacts.
Anti-idiotypic antibodies can be generated to bind specifically to one monoclonal antibody drug.
These highly specialized antibodies can be used to set up pharmacokinetic (PK) assays in different formats to measure free or total drug levels in preclinical and clinical samples, or as positive controls in ADA assays.
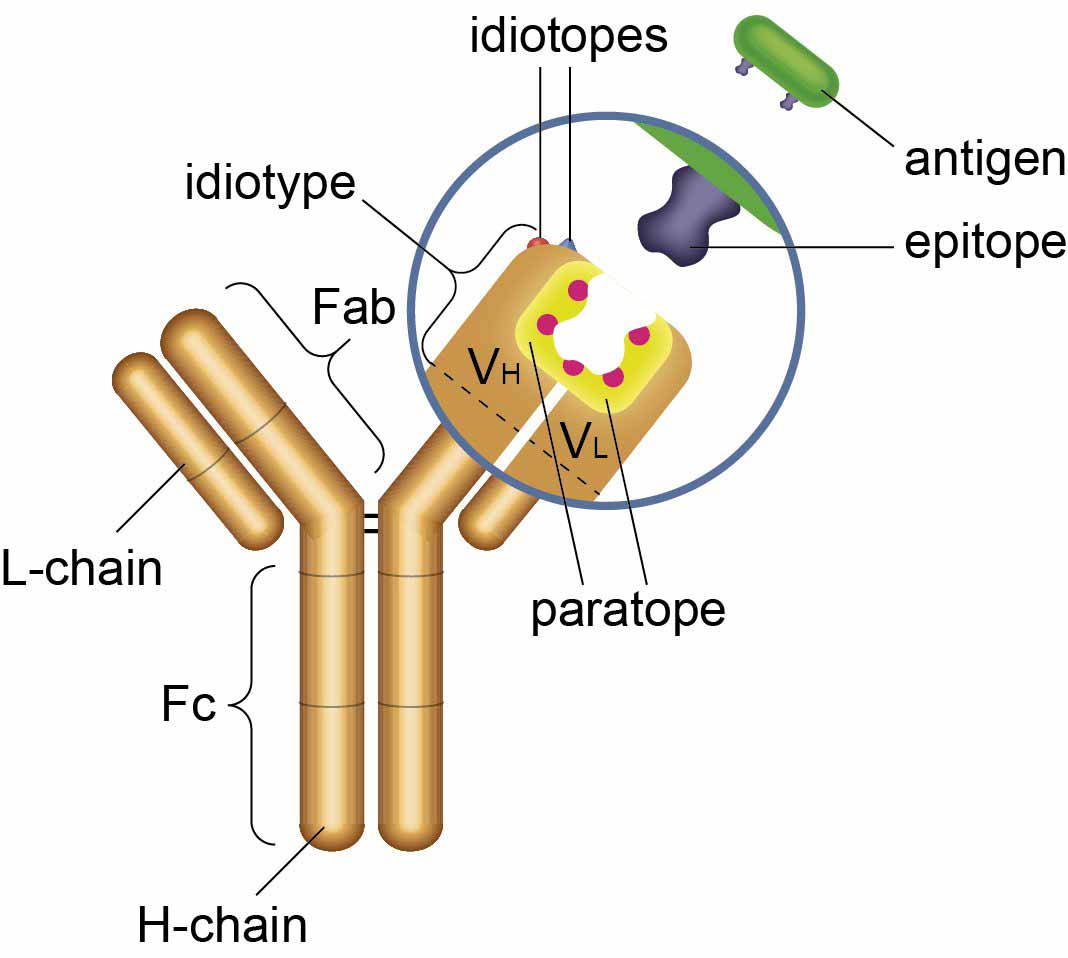
Fig 1. Antibody idiotope - the unique set of antigenic determinants of the variable region of an antibody
Anti-Idiotypic Antibodies Support the Generation of Human Antibody Drugs
Bio-Rad is an expert manufacturer of highly specific, high affinity anti-idiotypic antibodies to support preclinical research, clinical trials and patient monitoring for innovator and biosimilar products.
These antibodies are made using the HuCAL® recombinant monoclonal antibody library and a novel and improved method of phage display to generate antibodies in fully human Fab and immunoglobulin formats.
Ready-made anti-idiotypic antibodies against monoclonal antibody drugsTechnical articles featuring applications of anti-idiotypic antibodies
How to overcome assay challenges using custom recombinant antibodies - Watch Now
Custom Anti-Idiotypic Antibody Generation
At Bio-Rad we have over 15 years of experience generating recombinant monoclonal antibodies for our clients’ individual requirements using the HuCAL technology.
Recombinant monoclonal antibodies are comparable to traditional monoclonal antibodies in their functionality. However, since they are generated using fully in vitro processes they offer greater flexibility during their production and more opportunities for optimization, such as affinity maturation and conversion to different formats.
High Specificity Anti-Idiotypic Antibodies
For anti-idiotypic antibody generation using HuCAL technology, selection of the antibody is carried out on the drug in the presence of isotype sub-class matched antibodies as blockers, to avoid enrichment of specificities that bind to other regions of the antibody drug and to ensure idiotope specificity. Additionally, selection performed in the presence of human serum avoids matrix effects in the final assay.
The selection method can be guided to generate anti-idiotypic antibodies with different binding modes and properties, figure 2. Inhibitory antibodies (Type 1) are ideal for cell based assays and ELISA. Selection of a non-inhibitory antibody that binds to an idiotope outside the antigen binding site of the drug results in an antibody that can be used to detect both free and bound drug in the sample (Type 2). Unique to Bio-Rad is the capability to isolate rare specificities, such as drug-target complex binders (Type 3) that can be used to quantify bound drug, as opposed to free drug levels.
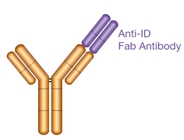 Type 1
Type 1
- Anti-idiotypic (anti-ID) antibody
- Paratope-specific
- Inhibitory
- Neutralizing
- Detects free drug
 Type 2
Type 2
- Anti-idiotypic antibody
- Not paratope-specific
- Not inhibitory
- Detects total drug (free, partially bound, fully bound)
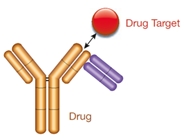
 Type 3
Type 3
- Drug-target complex specific
- Not inhibitory
- Detects bound drug exclusively
Fig 2. Three different types of anti-biotherapeutic antibody. Type 1, inhibitory anti-idiotypic antibodies for measuring free drug; type 2 non-inhibitory antibodies detect total drug; type 3 drug-target complex-specific antibodies detect bound drug exclusively
Using a combination of the different types of drug-specific antibodies offers the assay developer enhanced flexibility and better overall information about the availability and state of the drug antibody.
Our experts can design a strategy to make Type 1, Type 2 and Type 3 antibodies to your biotherapeutic target. Contact us for a discussion.
Reference
- Harth and Frisch (2021). Recombinant anti-idiotypic antibodies in ligand binding assays for antibody drug development. Methods Mol Biol. 2261:291-306.
Custom Antibody Development - Useful Reading
Related Topics
- More about our custom anti-idiotypic service
- Characterization of critical reagents for ligand binding assays
- Webinar: How to improve your antibody drug development assays with anti-idiotypic antibodies
- HuCAL® custom monoclonal antibody generation
- Ready-made antibodies for drug discovery and development
- Anti-biotherapeutic antibody quality control and characterization
Why Choose Bio-Rad
Anti-Idiotypic Antibodies
Discover the ten key reasons our anti-IDs are market leaders
With over 350 products, Bio-Rad offers one of the largest ranges of anti-IDs available on the market.
This downloadable infographic allows you to explore the top reasons to choose from our expansive range of ready-made antibodies, including stringent quality control, specificity testing, and performance guarantee.