
Recombinant Fully-Human Immunoglobulin
Isotype Controls
Controls for antibody drug discovery and diagnostic assays
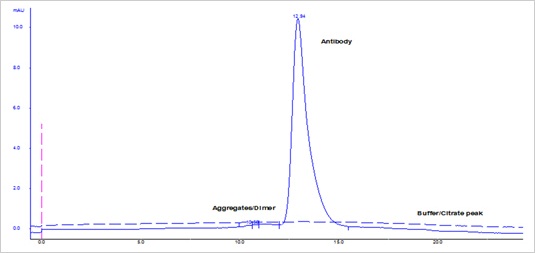
Fig. 1. Quality check by size exclusion chromatography of recombinant human IgG2/kappa.
Our fully-human recombinant immunoglobulin monoclonal antibodies are widely used as negative controls in antibody drug discovery and development, and in vitro diagnostic assays. Below, view our range of controls for all IgG isotypes, IgA, IgE, and IgM. Preservative free and low endotoxin formats for in vivo studies are also available on request.
These recombinant antibodies are expressed in a mammalian cell line. The production method ensures continuity of supply and consistent quality from batch to batch, overcoming the challenges of sourcing control immunoglobulins from human sera.
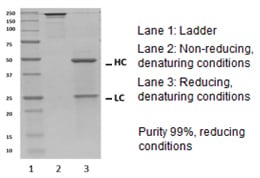
Fig. 2. Quality check of recombinant human IgG2/kappa by SDS-PAGE.
Special features of our recombinant isotype controls
- Sequence of heavy and light chain known, full allotype information available
- Specific for Green Fluorescent Protein (GFP), not found in mammalian cells
- IgG4 available as wildtype and point mutant S228P
- IgG1 available in allotypes G1m3 and G1m17,1
- IgG1 in allotypes G1m3 available as wildtype and point mutant LALA, N297Q or YTE
- Preservative free and low endotoxin format on request
Isotype Control Selection
Selecting an appropriate isotype control is crucial in the antibody drug discovery process, as well as for diagnostic assays measuring levels of human antibodies. Negative isotype controls are used for controlled validation of antibody functions. They serve to differentiate the nonspecific background signals initiated by the antibody Fc domain or other cellular protein interactions, from the target-specific antibody signal.
The ideal negative control matches the species and antibody isotype (including light chain) and subclass of the primary antibody used in the assays; it must not bind to any cellular target present in the sample.
For a recombinant fully-human control antibody targeting a specific antigen of your choice, see our custom HuCAL® monoclonal antibody generation service.
Immunoglobulin Isotype Controls
| Description | Specificity | Target | Format | Host | Isotype | Clone | Applications | Citations | Product Type | Code | Validation Types |
|---|
Related Reading
Discover the technology behind our fully human isotype control antibodies: