Secondary Antibodies
Secondary antibodies recognize an antibody as their target antigen, usually referred to as the primary antibody which in turn is bound to its antigen or protein of interest.
Unlike anti-idiotypic antibodies which bind specifically to the antigen-binding domain, secondary antibodies are specific to the conserved regions of an antibody which are constant within a particular class of antibody such as human IgG or mouse IgM.
Secondary antibodies are therefore able to bind to ranges of antibody clones making them very effective research tools.
Like primary antibodies, secondary antibodies can be monoclonal and polyclonal. They are typically produced by immunizing a host animal (such as a goat, rabbit, or donkey) with antibodies from another species (for example, mouse or human IgG). The host animal’s immune system recognizes these foreign antibodies and generates an immune response, producing polyclonal secondary antibodies. Whereas monoclonal secondary antibodies are produced by creating hybridomas from antibody-producing cells and myeloma cells, resulting in a single antibody type.
Alternatively, recombinant monoclonal secondary antibodies are made by cloning antibody genes into expression vectors introduced into host cells. These host cells produce consistent antibodies in vitro, which are then purified—eliminating the need for animal immunization.
For more specific information on secondary antibodies, you can select from the topics below, those listed in the navigation bar on the left, or alternatively, download our secondary antibody guide.
Secondary Antibody Selection Tool
Flow Cytometry Secondary Antibodies
Western Blot Secondary Antibodies
Immunofluorescence Secondary Antibodies
Immunohistochemistry Secondary Antibodies
Here we describe the applications of secondary antibodies and the considerations needed in their selection.
Applications of Secondary Antibodies
1. Detection of a primary antibody:
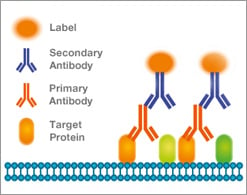 Secondary antibodies are commonly used to detect and visualize the presence of a primary antibody in applications like western blotting or immunofluorescent histology. In these applications the secondary antibody is labeled with a reporter molecule, which may be an enzyme like HRP or a fluorophore such as FITC. Multiple secondary antibodies can bind to a single primary antibody, increasing the sensitivity and amplifying the signal.
Secondary antibodies are commonly used to detect and visualize the presence of a primary antibody in applications like western blotting or immunofluorescent histology. In these applications the secondary antibody is labeled with a reporter molecule, which may be an enzyme like HRP or a fluorophore such as FITC. Multiple secondary antibodies can bind to a single primary antibody, increasing the sensitivity and amplifying the signal.
Further signal amplification can be achieved by using an unlabeled secondary and a labeled tertiary antibody if required.
Read our secondary antibodies optimization guide for essential information on experimental design.
2. Antibody capture:
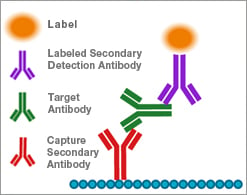 Unlabeled secondary antibodies can be used to capture antibodies of interest from biological solutions by quantitative ELISA. For example, an unlabeled secondary mouse anti-human IgG may be used as the capture antibody to bind human IgG from a patient serum sample. This is then detected with a labeled secondary mouse anti-human IgG which binds to the captured IgG.
Unlabeled secondary antibodies can be used to capture antibodies of interest from biological solutions by quantitative ELISA. For example, an unlabeled secondary mouse anti-human IgG may be used as the capture antibody to bind human IgG from a patient serum sample. This is then detected with a labeled secondary mouse anti-human IgG which binds to the captured IgG.
The use of a calibration curve of known standards allows this signal to be quantified.
3. Detection and quantification of recombinant proteins:
Secondary antibodies can be used to detect and quantify recombinant proteins that have been engineered to contain antibody domains, for example, for ease of expression, detection, stability, or increased in vivo half-life.
Read more about how secondary antibodies specifically targeted to the CH2 and CH3 domains of immunoglobulins enable the study of Fc fragments in the development of new therapeutic antibody fragments.
Secondary Antibodies at a Glance
The secondary reagents have been carefully selected to provide optimum quality and flexibility for your experimental design:
- Choose monoclonal or polyclonal
- IgG molecules or F(ab’)2 fragments
- Available in many formats
- Suitable for a wide range of applications such as flow cytometry, western blotting, and immunocytochemistry
- Cross-absorbed antibodies
- Fluorescent StarBright™ Dye Secondary Antibodies. Exceptionally bright and ideal for multiplex western blotting
Target |
Host |
Class and Chain |
Conjugates |
|---|---|---|---|
|
Ig IgA, IgA A1, IgA A2, IgA H, IgA secretory chain IgD, IgD H, IgD D IgE IgG, IgG1, IgG2, IgG2a,IgG2b, IgG2c, IgG3, IgG4, IgG H/L, IgG Fc, IgG Fc CH2 domain, IgG CH2 domain, IgG CH3 domain, IgG gamma, IgG Fab, IgG F(ab’)2 IgM, IgM Mu, IgM H IgG/A/M Lamba light chain Kappa light chain J chain |