Biotin-labeled Primary Antibodies with Streptavidin Products, Resources, and Tools
Fluorescently-labeled streptavidin can be used to visualize biotin-labeled primary antibodies. Protocols, top tips, and benefits of this technique can be found here, alongside general resources for choosing fluorophores and building panels.
Products
Fluorescently-labeled Streptavidin for Flow Cytometry
Highly specific streptavidin-biotin binding is utilized in many applications, for example, HRP labeled streptavidin (STAR5B) and alkaline phosphatase labeled streptavidin (STAR6B) are commonly used in ELISA, western blotting, and immunohistochemistry. We have handy protocols describing the use of streptavidin in immunostaining (frozen tissue sections and paraffin sections) and ELISA (direct and sandwich).
Fluorescently labeled streptavidin is also ideal for flow cytometry to visualize primary antibodies conjugated to biotin, also known as biotinylated primary antibodies. They are compatible with multiplexing panels, making them a great option when the ideal directly labeled fluorescent antibody is not available. We will describe their many benefits for use in flow cytometry, and how to use them successfully in your experiments.
The Biology of Streptavidin
Streptavidin is isolated from Streptomyces avidinii and has four binding sites with strong affinity to biotin, a naturally occurring vitamin. The relatively small size of biotin means that when bound to an antibody it does not hinder the antibody binding site allowing it to still bind to its target antigen.
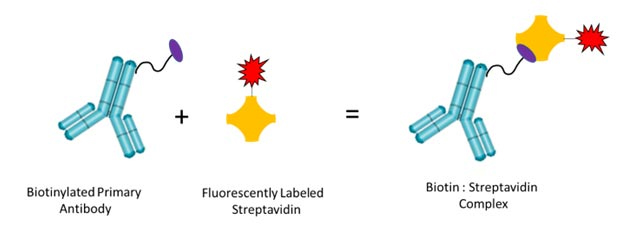
Fig. 1. Binding of biotinylated antibodies to fluorescently labeled streptavidin. Primary antibodies are biotinylated and bind fluorescently labeled streptavidin with high affinity.
Benefits when using Biotinylated Antibodies with Fluorescently Labeled Streptavidin in Flow Cytometry
There are many features and benefits when using the biotin:streptavidin system, as shown in Table 1, that not only aid multiplex panel design, but also help to achieve good quality flow data.
Table 1. Features and benefits of using biotin: streptavidin in flow cytometry.
Feature |
Benefit |
|---|---|
|
Low background staining |
Streptavidin has a high affinity to biotin, with negligible binding to non-biotinylated macromolecules, and therefore gives very low background. |
|
Stable |
The strong biotin-streptavidin bond can withstand extremes in pH and temperature therefore their interaction remains very stable during assays. |
|
Signal amplification |
Biotinylated antibodies may be conjugated to more than one biotin molecule, each biotin will bind a labeled streptavidin resulting in signal amplification. This makes them ideal for detecting low-expressing antigens. |
|
Expands antibody choice |
When there is no directly conjugated antibody available or not one in your preferred format using the biotin:streptavidin system can provide the solution. |
|
Suitable for use with all isotypes |
In flow cytometry secondary antibodies need to be carefully chosen to match the isotype of your primary antibody, while also ensuring there is no unwanted binding to other primary antibodies in your experiment. As the biotin:streptavidin system is isotype independent, it overcomes any problems of isotype compatibility. |
|
Large range of biotinylated antibodies |
Many antibodies validated for flow cytometry are available biotinylated. |
|
Simple to conjugate purified antibodies to biotin |
If an antibody of interest is not available biotinylated, it’s easy to conjugate a purified version of the antibody. For example, Biotin (Type 1) Conjugation Kit from Bio-Rad is simple to use, adding biotin to an antibody in minutes. |
|
Large choice of fluorophores |
Bio-Rad’s labeled streptavidin conjugates are available in a range of fluorophores across all the common laser lines, including new StarBright™ Dyes. This gives a wide choice of fluorophores to easily incorporate into new or existing flow panels. |
How to Use Fluorescently Labeled Streptavidin in Flow Cytometry
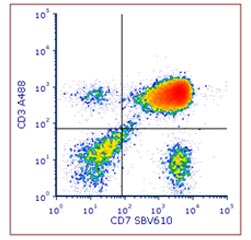
Fig. 2. Staining of human peripheral blood with anti-Human CD7 biotin: StarBright Violet 610 streptavidin and Anti-CD3 Alexa Fluor 488. Red cell lysed human blood was stained with Alexa Fluor 488 conjugated Mouse anti-Human CD3 (MCA463A488) and biotin conjugated Mouse anti-Human CD7 (MCA1197B) detected with streptavidin StarBright Violet 610 (STAR210SBV610). Cells were acquired on the ZE5 Cell Analyzer and gated on live single cell lymphocytes.
1. Indirect staining — common two-step method
The most common method to use the biotin:streptavidin system is a two-step staining protocol. This method is similar to using a labeled secondary antibody to detect a primary antibody.
Step 1:
- Incubate cells with a biotinylated primary antibody
- Wash to remove excess antibody
Step 2:
- Incubate cells with a fluorescently labeled streptavidin
- Wash to remove excess streptavidin and acquire samples on a flow cytometer
An example of indirect staining can be seen in Figure 2, where human blood was stained with biotinylated CD7 (MCA1197B) and detected with StarBright Violet 610 labeled streptavidin (STAR210SBV610).
2. Direct staining — premix one-step method
An alternative method is to mix a biotinylated antibody with a fluorescently labeled streptavidin. Cells can be directly stained with this premix complex using a one-step method.
Step 1:
- Incubate cells with a premixed biotinylated primary antibody: fluorescently labeled streptavidin complex
- Wash to remove excess antibody and streptavidin, and acquire samples on a flow cytometer
This method requires optimization of the ratio of biotinylated antibody to fluorescently labeled streptavidin. As shown in Figure 3, Anti-Human CD19 biotin and StarBright UltraViolet 795 labeled streptavidin (STAR210SBUV795) were premixed at three different molar ratios prior to staining. For this pair the 1:3 ratio gave the best staining, having the greatest separation of the positive and negative population and a higher stain index.
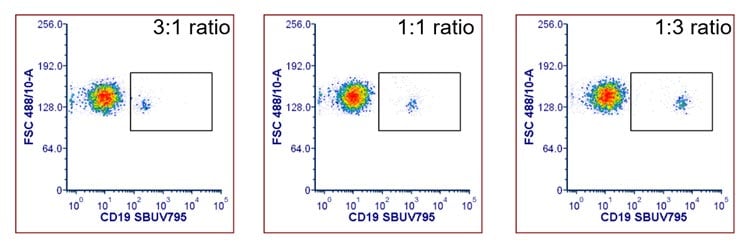
Fig. 3. Staining of human PBMCs with biotinylated Anti-CD19 premixed with streptavidin StarBright UltraViolet 795. Anti-CD19 biotin antibodies were mixed with streptavidin StarBright UltraViolet 795 (STAR210SBUV795) at different molar ratios for 30 minutes at room temperature prior to staining human PBMCs. Cells were acquired on the ZE5 Cell Analyzer and gated on single cell lymphocytes.
Optimization of the ratio only needs to be done once for each biotinylated antibody: labeled streptavidin pair. If a pairing will be used more than twice this method will save time as staining is reduced by 60-80 minutes over an indirect staining protocol. Also, as the premix is stable for at least seven days, a master premix can be made and used over the course of at least a week.
It is important to note that there may be rare cases where this method doesn’t work well. If too many biotins are attached to an antibody or biotin residues are too close to the antigen binding site, there will be steric hinderance resulting in loss of binding. In these rare cases an indirect method will be required to achieve bright staining.
A direct staining protocol will be required when more than one biotinylated antibody is incorporated into a multiplexing panel. Each biotinylated antibody needs to be premixed with a different fluorescent labeled streptavidin. In Figure 4 you can see an example of using three different premixed biotinylated antibody:streptavidin complexes together. The optimal ratio of each pairing was determined and titrated prior to use.
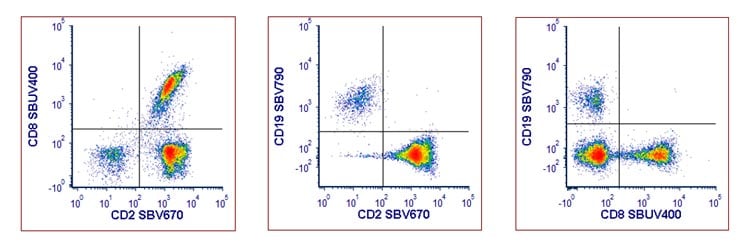
Fig. 4. Staining of human PBMCs with three premixed biotinylated antibody: fluorescently labeled streptavidin complexes. PBMCs were stained with CD2B: STAR210SBV670. CD8B: STAR210SBUV400 and CD19B :STAR210SBV790. Cells were acquired on the ZE5 Cell Analyzer and gated on single cell lymphocytes.
Biotinylated Antibodies
Bio-Rad has a range of biotinylated antibodies validated for use in flow cytometry.
However, if an antibody of interest is not available biotinylated it is easy to conjugate the purified version using a simple kit, such as Biotin (Type 1) Conjugation Kit. As shown in Figure 5, an antibody conjugated using this kit gave similar staining to a commercial biotinylated antibody and a directly conjugated antibody.
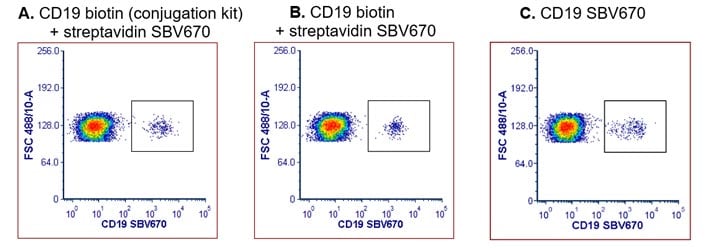
Fig. 5. CD19 staining of human peripheral blood using a biotinylated CD19 antibody detected with StarBright Violet 670 streptavidin or a directly conjugated CD19 SBV670 antibody. Red cell lysed human blood was stained using either A. Anti-CD19 (MCA1940GA) biotinylated using a conjugation kit (LNK261B) and detected with streptavidin StarBright Violet 670 (STAR210SBV670). B. A commercially available biotinylated Anti-CD19 Antibody (MCA1940B) and detected with streptavidin StarBright Violet 670 (STAR210SBV670). C. Anti-CD19 directly conjugated to StarBright Violet 670 (MCA1940SBV670). Cells were acquired on the ZE5 Cell Analyzer and gated on live single cell lymphocytes.
Top Tips When Using Biotinylated Antibodies with Fluorescently Labeled Streptavidin in Flow Cytometry
The following tips should be considered when designing flow experiments.
- Perform a titration. It is best practice to titrate your antibody to determine the best concentration of antibody and streptavidin to use. Start with a fixed amount of the biotinylated antibody, using the recommended amount from the supplier, and titrate the fluorescently labeled streptavidin. If there is high nonspecific staining, next titrate the primary antibody using the fixed amount of the fluorescently labeled streptavidin that has been determined.
- Include a blocking step. Although streptavidin gives negligible nonspecific staining it is recommended to still perform an antibody blocking step that is in most staining protocols. This is due to the presence of primary antibodies that may bind non-specifically to Fc receptors.
- Optimize premixed antibody: streptavidin ratios. The optimal molar ratio of antibody to streptavidin needs to be determined when using premixed complexes. Recommended ratios for initial testing are 3:1, 1:1, and 1:3 of antibody to streptavidin, as these cover the ratios that commonly give the highest stain index.
Streptavidin Formats
Bio-Rad offers a wide range of fluorescently labeled streptavidin including our new StarBright Dyes as well as traditional dyes, shown in Table 2 below.
Table 2. Fluorescently labeled streptavidin available from Bio-Rad.
Fluorophore |
Optimal excitation laser |
Excitation Max, nm |
Emission Max, nm |
Product code |
|---|---|---|---|---|
|
StarBright UltraViolet 400 |
355 |
347 |
394 |
|
|
StarBright UltraViolet 445 |
355 |
347 |
440 |
|
|
StarBright UltraViolet 510 |
355 |
340 |
513 |
|
|
StarBright UltraViolet 575 |
355 |
340 |
569 |
|
|
StarBright UltraViolet 605 |
355 |
340 |
609 |
|
|
StarBright UltraViolet 665 |
355 |
340 |
684 |
|
|
StarBright UltraViolet 740 |
355 |
344 |
742 |
|
|
StarBright UltraViolet 795 |
355 |
340 |
792 |
|
|
StarBright Violet 440 |
405 |
385 |
438 |
|
|
StarBright Violet 475 |
405 |
405 |
479 |
|
|
StarBright Violet 515 |
405 |
401 |
516 |
|
|
StarBright Violet 570 |
405 |
402 |
570 |
|
|
StarBright Violet 610 |
405 |
402 |
607 |
|
|
StarBright Violet 670 |
405 |
400 |
667 |
|
|
StarBright Violet 760 |
405 |
403 |
754 |
|
|
StarBright Violet 790 |
405 |
401 |
782 |
|
|
DyLight 405 |
405 |
400 |
420 |
|
|
StarBright Blue 580 |
488 |
475 |
582 |
|
|
StarBright Blue 615 |
488 |
475 |
612 |
|
|
StarBright Blue 675 |
488 |
476 |
675 |
|
|
StarBright Blue 700 |
488 |
470 |
700 |
|
|
StarBright Blue 765 |
488 |
476 |
764 |
|
|
StarBright Blue 810 |
488 |
477 |
802 |
|
|
FITC |
488 |
490 |
525 |
|
|
DyLight 488 |
488 |
493 |
518 |
|
|
StarBright Yellow 575 |
561 |
548 |
579 |
|
|
StarBright Yellow 605 |
561 |
572 |
606 |
|
|
StarBright Yellow 665 |
561 |
554 |
670 |
|
|
StarBright Yellow 720 |
561 |
548 |
719 |
|
|
StarBright Yellow 800 |
561 |
549 |
788 |
|
|
RPE |
561 |
496 |
578 |
|
|
DyLight 550 |
561 |
562 |
576 |
|
|
DyLight 650 |
561 |
564 |
673 |
|
|
StarBright Red 670 |
640 |
653 |
666 |
|
|
StarBright Red 715 |
640 |
638 |
712 |
Coming soon |
|
StarBright Red 775 |
640 |
653 |
778 |
Coming soon |
|
StarBright Red 815 |
640 |
654 |
811 |
Coming soon |
|
APC |
640 |
650 |
661 |
Protocol
Indirect Immunofluorescence Staining of Cells and Blood for Flow Cytometry
This technique is applicable when using unconjugated or biotin-conjugated monoclonal and polyclonal antibodies recognizing cell surface antigens. A conjugated secondary reagent must be used to visualize the primary antibody, for example streptavidin in the case of biotin.
This method provides a general procedure for use with the majority of Bio-Rad reagents. In some cases specific recommendations are provided on product datasheets, and these methods should always be used in conjunction with the product and batch specific information provided with each vial. A certain level of technical skill and immunological knowledge is required for the successful design and implementation of these techniques; these are guidelines only and may need to be adjusted for particular applications. Specific methodology for blood appears in [ ] brackets.
Reagents
- Phosphate buffered saline (BUF036A) containing 1% bovine serum albumin (PBS/BSA).
- Erythrolyse red blood cell lysing buffer (BUF04).
- Anticoagulant (Note: for basic staining any appropriate anticoagulant, such as heparin, EDTA, or acid citrate dextrose, may be used. In some instances specific anticoagulants may be required)
- Optional: 0.5% (w/v) paraformaldehyde in PBS (Note: dissolve on heated stirrer and cool before use)
Method
-
Prepare cells appropriately; refer to protocol FC1. 'Preparation of cells for flow cytometry' for further information. Adjust the cell suspension to a concentration of 1 x 107 cells/ml with cold (4oC) PBS/BSA buffer.
[Whole blood samples may be used undiluted unless the cell count is high, e.g. as in leukemia. Use appropriate anticoagulant].
- Aliquot 100 μl of the cell suspension [or whole blood] into as many test tubes as required.
- Add primary antibody at the vendor-recommended dilution. Mix well and incubate at 4oC for at least 30 min.
-
Wash cells with 2 ml of cold (4oC) PBS/BSA, centrifuge at 300-400 g for 5 min, and discard the supernatant.
[To the blood suspension add 2 ml freshly prepared red cell lysis buffer and mix well. Incubate for 10 minutes at room temperature. Centrifuge at 300-400 g for 5 minutes and discard the supernatant. Wash with 2 ml of PBS/BSA, centrifuge at 300-400 g for 5 minutes, and discard the supernatant].
- Add an appropriate secondary reagent at the vendor-recommended dilution. Mix well and incubate at 4oC for at least 30 min, avoiding direct light.
- Centrifuge at 300-400 g for 5 min at room temperature and discard the supernatant.
- Re-suspend cells in 200 μl of cold (4oC) PBS or with 200 μl of 0.5% paraformaldehyde in PBS if required.
- Acquire data by flow cytometry. Analyze fixed cells within 24 hours.
Notes
-
To avoid nonspecific binding, you also need to block Fc receptors on cell types such as spleen cells, with FcR blocking reagents e.g. Bio-Rad’s Mouse Seroblock reagent (BUF041).
-
Appropriate controls should always be carried out, for flow cytometry the following should be considered for inclusion;
-
Isotype controls used to determine if the staining is specific.
-
Unstained cells should always be included in the experimental set up to monitor autofluorescence.
-
- For all multicolor flow cytometry experiments it is advisable to include compensation controls and fluorescence minus one (FMO) controls, which assist with identifying gating boundaries.
Resources
Choosing Fluorophores for Flow Cytometry

Fluorophores and Viability Dyes for Flow Cytometry
Reference Postcard
Flow cytometry generally requires bright fluorophores that are excitable by one laser with a narrow emission wavelength to allow multicolor panel building. Below is a handy guide to the range of fluorophores Bio-Rad offers, including our new StarBright Dyes, that are suitable for flow cytometry, grouped by the laser used to excite them.
Additional fluorophore information can be found on specific webpages organized by the laser that excites them. This includes the maximal emission wavelength, relative brightness, ZE5 Cell Analyzer filter used for detection, similar or incompatible fluorophores, and our antibodies available conjugated to them, helping you select the right fluorophore for your experiment.
For more information on which fluorophores are the most suitable for your experiment, try our fluorescent spectraviewer or download our poster showing common fluorophores used in flow cytometry.
Fluorophores excited by the 355 nm (Ultraviolet) Laser
- StarBright UltraViolet 400
- StarBright UltraViolet 445
- StarBright UltraViolet 510
- StarBright UltraViolet 575
- StarBright UltraViolet 605
- StarBright UltraViolet 665
- StarBright UltraViolet 740
-
StarBright UltraViolet 795
Fluorophores Excited by the 405 nm (Violet) Laser
- Pacific Blue
- StarBright Violet 440
- StarBright Violet 475
- StarBright Violet 515
- Amethyst Orange
- StarBright Violet 570
- StarBright Violet 610
- StarBright Violet 670
- StarBright Violet 710
- StarBright Violet 760
-
StarBright Violet 790
Fluorophores Excited by the 488 nm (Blue) Laser
- FITC
- Alexa Fluor 488
- PE
- PE-Alexa Fluor647
- PE-Cy5
- PE-Cy5.5
- PE-Alexa Fluor750
- PE-Cy7
- PerCP
- StarBright Blue 580
- StarBright Blue 615
- StarBright Blue 675
- StarBright Blue 700
- StarBright Blue 765
-
StarBright Blue 810
Fluorophores Excited by the 561 nm (Yellow) Laser
- PE
- PE-Alexa Fluor647
- PE-Cy5
- PE-Cy5.5
- PE-Alexa Fluor750
- PE-Cy7
- StarBright Yellow 575
- StarBright Yellow 605
- StarBright Yellow 665
- StarBright Yellow 720
-
StarBright Yellow 800
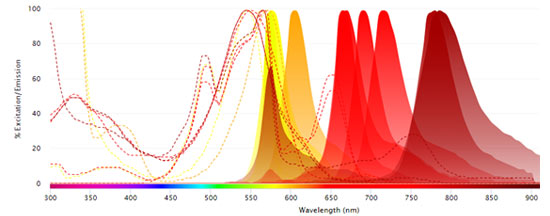
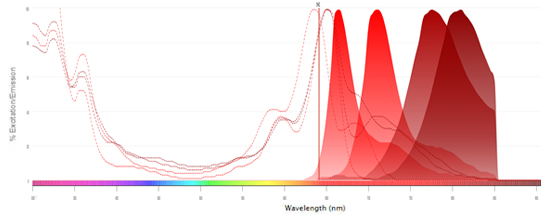
Fluorophores Excited by the 640 nm (Red) Laser
- Alexa Fluor 647
- APC
- Alexa Fluor 700
- StarBright Red 670
- StarBright Red 715
- StarBright Red 775
-
StarBright Red 815
Antigen Density of Common Human and Murine Markers
Quick Guide
Antigen Density
Tutorials for building multicolor flow cytometry panels always highlight the importance of antigen density — But why is it so important?
The number of antigens, or target molecules that a cell carries directly correlates to the intensity of the positive population and will determine the optimal fluorophore you should use for each marker. As a general rule you should pair bright fluorophores (e.g. PE) with low expressing markers and dimmer fluorophores (e.g. Pacific Blue) with highly expressed markers. Careful choice of fluorophores will help with the resolution of your cell populations. It is possible for bright fluorophores to be paired with highly expressed antigens, but better to avoid pairing dim fluorophores with low abundance antigens.
The guide provides advice on how to measure antigen density and lists the antigen density of some common markers.
Basic Multicolor Panel Design Rules
Before you build your panels it is critically important to have as clear an idea as possible of what is required to identify your population of interest. For example, are you looking at one cell type or is it a subset, are you looking at an activation marker or a change in cell frequency, are the markers co-expressed or do you even know the expression pattern?
Ideally, when building multicolor panels, it is best to separate fluorophore excitation across lasers, and where possible, the emission across the detectors. This will minimize the amount of spillover and therefore compensation you will need to do. It will also reduce the effect that fluorescence spread will have on your data. However, as you increase the number of fluorophores in your panel, this will not always be possible. Therefore other considerations need to be included in your design.
Webinar: Multicolor Panel Building in Flow Cytometry
Presented by: Dr Sebastian Hedlund, Flow Cytometry Specialist at Bio-Rad Laboratories
Multicolor flow cytometry is the analysis of multiple fluorescent parameters in one sample. Building large flow cytometry panels can be daunting because each additional fluorophore you add to your panel has the potential to influence another fluorophore.
Join our 45 minute webinar to learn which fluorophores are compatible with each other, how they interact and can affect your staining and how to obtain optimal resolution of signal. Furthermore, we will show you how dump channels, fluorophore brightness, antigen density, marker expression patterns and instrument configuration can all contribute to improving your panel design.
This webinar is recommended for both novices and researchers with some experience of flow cytometry.
Speaker Biography
 Sebastian obtained his degree from the Institute of Technology, Linköping University, Sweden, followed by an MSc in Medical Biology from the same institute. He then moved to Freiburg, Germany where he obtained his PhD in Immunology and Molecular Biology at the Max Planck Institute of Immunobiology and Epigenetics.
Sebastian obtained his degree from the Institute of Technology, Linköping University, Sweden, followed by an MSc in Medical Biology from the same institute. He then moved to Freiburg, Germany where he obtained his PhD in Immunology and Molecular Biology at the Max Planck Institute of Immunobiology and Epigenetics.
The focus of his thesis was the role of microRNA in innate immunity and immunopathology. He then left academia and started work as a dedicated flow cytometry specialist, before joining Bio-Rad in 2016 as the flow cytometry specialist for Scandinavia, Eastern Europe, Israel, and South Africa.
Multicolor Panel Builder
Build multicolor flow cytometry panels in just a few simple steps
- Start your design by first selecting your instrument from the drop down menu, or create a customized instrument, to build a personalized panel
- Select the target and species you want to identify. Keep adding until you have all the markers you want
- Choose antibodies by clone, isotype and format. If there is not an antibody available in the format you require, select the fluorophore as a placeholder and continue
- Include a viability dye or dump channel for more complexity
The ability to select antigen density and fluorophores based on brightness, combined with automatic exclusion of incompatible fluorophores, will help you build larger panels with confidence.
Interactive Flow Cytometry, Spectral Analyzer, Microscopy, and Western Blot Fluorescent Spectraviewer
Find the right fluorophore for your application using our new interactive, fluorescent spectraviewer with hundreds of fluorophores to choose from.
Bio-Rad’s new spectraviewer is the only spectraviewer to allow a multi-laser view and support three applications in a single tool.
- Supports flow cytometry, fluorescence microscopy, and western blotting
- Displays the excitation and emission profiles of the fluorophores available to you, essential when building multicolor panels for flow cytometry
- Allows full customization: select any fluorophore, laser, and filter combination
- Provides more information than any other spectraviewer: the traditional view, showing the fluorescence spectra, is complemented by the multi-laser view visualizing the spillover and cross-laser excitation and emission
- Saves time with the pre-loaded Bio-Rad instrument settings
The ability to view in a multi-laser format allows you to check fluorophore compatibility, including potential issues due to cross-laser excitation, and predict compensation.
Bio-Rad has over 4,000 antibodies validated for flow cytometry as well as helpful resources to guide you through your flow cytometry experiments. These resources as well as listings of antibodies, kits, and controls can be accessed from our dedicated flow cytometry page.






