Cell Surface Products and Resources for Flow Cytometry

- On This Page
- Products
- Antibody Catalog
- Cell Surface Flow Cytometry Antibodies
- Resources
- Protocol
Cell surface staining is the simplest staining method as antibodies against surface markers don’t need to enter the cells. We have provided a protocol and resources to help you get the best results when using this staining method.
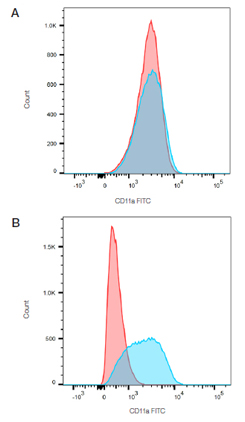
Fig. 1. Fc blocking. THP-1 cells stained with mouse anti-Human CD11a (blue) or mouse IgG2a isotype control (MCA929F) (red) in the absence (A) and presence (B) of human Seroblock (BUF070A).
Products
Flow Cytometry Blocking Controls
Fc receptors are found on monocytes, macrophages, dendritic cells, and B cells. As the name suggests they bind antibodies via their constant Fc domain rather than the antigen specific Fab domain.
This type of binding can lead to false positives and meaningless data. In order to prevent this type of binding, Fc blocking reagents (e.g. Human Fc Seroblock and Murine Fc Seroblock) have been developed which, when added to a staining protocol, can ensure that only antigen specific binding is observed.
Antibody Catalog
Bio-Rad offers over 4000 antibodies, kits, and reagents to support flow cytometry for a wide range of research areas such as cancer, immunology, cell biology, and veterinary research.
Cell Surface Flow Cytometry Antibodies
| Description | Target | Format | Clone | Applications | Citations | Code |
|---|

Updated Flow Cytometry Basics Guide
Practical advice to get you started in flow cytometry.
Resources
Introduction to Flow Cytometry Basics
This flow cytometry guide aims to give you a basic overview of all the important aspects of flow cytometry.
With chapters on instrumentation, useful reagents, controls, experimental set up, and much more, this guide enables best practice to be followed and gives practical advice on building multicolor panels with example protocols.
This guide is invaluable to beginners wanting to start flow cytometry and as a handy tool for teaching others about this powerful application.
To further assist learning we have recently added a flow cytometry glossary of terms to this guide.
Protocol
Direct Immunofluorescence Staining Surface Epitopes of Cells and Blood
Applicable where the fluorophore is directly linked to the primary antibody e.g. RPE, FITC, and Alexa Fluor® conjugates. RPE conjugates should always be handled in the dark.
This method provides a general procedure for use with the majority of Bio-Rad reagents. In some cases specific recommendations are provided on product datasheets, and these methods should always be used in conjunction with the product and batch specific information provided with each vial. A certain level of technical skill and immunological knowledge is required for the successful design and implementation of these techniques; these are guidelines only and may need to be adjusted for particular applications. Specific methodology for blood appears in [ ] brackets.
Reagents
- Phosphate buffered saline (BUF036A) containing 1% bovine serum albumin (PBS/BSA).
- Erythrolyse red blood cell lysing buffer (BUF04).
- Anticoagulant (Note: for basic staining any appropriate anticoagulant, such as heparin, EDTA, or acid citrate dextrose, may be used. In some instances specific anticoagulants may be required)
- Optional: 0.5% (w/v) paraformaldehyde in PBS (Note: dissolve on heated stirrer and cool before use)
Method
-
Prepare cells appropriately; refer to protocol FC1. 'Preparation of cells for flow cytometry' for further information. Adjust the cell suspension to a concentration of 1 x 107 cells/ml with cold (4oC) PBS/BSA buffer.
[Whole blood samples may be used undiluted unless the cell count is high, e.g. as in leukemia. Use appropriate anticoagulants].
- Aliquot 100 μl of the cell suspension [or whole blood] into as many test tubes as required.
- Add antibody at the vendor-recommended dilution. Mix well and incubate at 4oC for at least 30 min, avoiding direct light.
-
Wash cells with 2 ml cold (4oC) PBS/BSA, centrifuge at 300-400 g for 5 min at 4oC, and discard the resulting supernatant.
[To the blood suspension add 2ml freshly prepared erythrolyse red cell lysing buffer and mix well. Incubate for 10 minutes at room temperature. Centrifuge at 300-400 g for 5 min at room temperature and discard the supernatant. Wash with 2 ml room temperature PBS/BSA, centrifuge at 300-400 g for 5 min, and discard the supernatant. Proceed to step 5].
- Re-suspend cells in 200 μl cold (4oC) PBS or with 200 μl of 0.5% paraformaldehyde in PBS if required.
- Acquire data by flow cytometry. Analyze fixed cells within 24 hours.
Notes
-
The following should be considered when designing your flow cytometry experiments:
-
To avoid nonspecific binding, you also need to block Fc receptors on cell types such as spleen cells, with FcR blocking reagents e.g. Bio-Rad’s Mouse Seroblock reagent (BUF041).
-
Appropriate controls should always be carried out, for flow cytometry the following should be considered for inclusion;
-
Isotype controls used to determine if the staining is specific
-
Unstained cells should always be included in the experimental set up to monitor autofluorescence.
-
-
-
For all multicolor flow cytometry experiments it is advisable to include compensation controls and fluorescence minus one (FMO) controls, which assist with identifying gating boundaries.





