Western Blotting: Introduction

- On This Page
- Overview
- All chapters
Overview
Western blotting is a core technique in cell and molecular biology, which is used to detect the presence of a specific protein in a complex mixture extracted from cells. As highlighted by Figure 1 below, the Western blotting procedure relies upon three key elements to accomplish this task: the separation of protein mixtures by size using gel electrophoresis; the efficient transfer of separated proteins to a solid support; and the specific detection of a target protein by appropriately matched antibodies. Once detected, the target protein will be visualized as a band on a blotting membrane, X-ray film, or an imaging system.
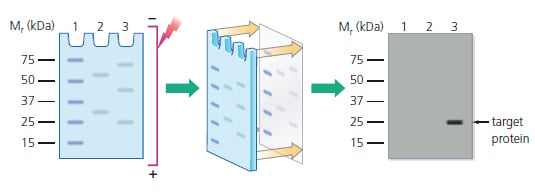
Figure 1: Separation of protein mixtures by electrophoresis; transfer to a blotting membrane; and detection of target protein, which only becomes visible in the final stage as a band similar to that shown in lane 3. Lane 1: Prestained molecular weight standards. Lanes 2 & 3: Protein mixtures
Since Western blotting is accomplished rapidly, using simple equipment and inexpensive reagents, it is one of the most common laboratory techniques. The results achieved are also easy to interpret, unique, and unambiguous. Therefore, it is routinely used on its own, or along with other immunoassays, in research and clinical settings.
Chapter 2
- Samples, Gels and Blotting
- Sample Preparation (Cell Lysates, Tissue Samples, Purified or Semi-purified Extracts, Determining Protein Concentration, Loading Buffer, Controls & Standards)
- Gel Electrophoresis
- Blotting (Blotting Membranes, Setting up the Transfer, Size of the Target Protein, Confirming the Transfer)
Chapter 3
Chapter 4
Chapter 5
Chapter 6


