Western Blot: Gel Electrophoresis
Overview
After the samples have been prepared, they are separated by size using SDS-PAGE (sodium dodecyl sulfate polyacrylamide gel electrophoresis). Since the samples have been denatured in gel loading buffer containing SDS detergent, the protein is uniformly negatively charged and will now migrate in an electric field through the gel and towards the positive electrode. Since the charge to mass ratio is equalized by the binding of SDS consistently along the length of the proteins, and higher structure has been removed, the proteins will be separated primarily by size. The key is to effect a separation such that the target protein will be properly resolved from the other components of the mixture. This makes it possible to clearly identify the target protein later through immunodetection with a specific antibody.
SDS-PAGE gels (commercially supplied or made in-house) usually consist of a main gel, which is poured between two glass or plastic plates, and which is sometimes topped by a short stacking gel. Gels can be made with a uniform acrylamide percentage, or with a continuously varying gradient that yields improved resolution over a broader range of molecular weights. See the table below for some common gel percentages and their separation ranges.
Polyacrylamide Gel Percentage Separation Ranges
| Acrylamide Concentration (%) | Separation Range (kDa) |
|---|---|
| 5 | 60-210 |
| 7.5 | 35-95 |
| 10 | 15-70 |
| 15 | 4-45 |
| 4-12 gradient | 5-200 |
| 4-20 gradient | 4-200 |
| 10-20 gradient | 3.5-110 |
The percentage and the thickness of the gel will impact the transfer of proteins out of the gel in the blotting phase, so using a thinner gel, or a lower percentage of acrylamide, may improve transfer results
Once the gel sets, it is placed into the running apparatus. Small volumes of protein (5-20 ml) dissolved in gel loading buffer are added to each individual well. The gel is then connected to a power supply and allowed to run for a few hours in a buffer tank to separate the proteins. If the gel is run at too high a voltage it will overheat and distort the bands. The accompanying gel shows cellular lysates which have been well-separated on a gradient gel, and stained with Coomassie dye to visualize all the separated protein bands.
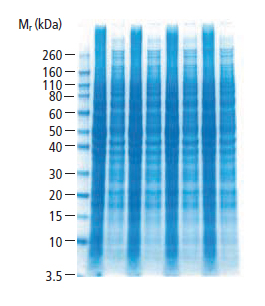
Figure 7: HeLa Cell Lysates (LYS001) Separated on a 4-12% Gradient Gel
| Sample Preparation | Blotting |





