Western Blot: Alternate Forms of a Protein - Prion Disease

- On This Page
- Overview
- Confirmation of HIV
Overview
Prion disease is a rare, progressive, neurodegenerative disorder that affects humans (vCJD, CJD), cows (BSE), sheep (Scrapie), and elk/deer (CWD). It leads to characteristic brain lesions and a rapid loss in neurological functions after a long latency period. The accumulation of an altered isoform of a normal cellular protein appears to be instrumental in the development of prion disease.
There are several ways to identify the presence of the abnormal prion protein - a bioassay, immunohistochemistry on diseased tissue samples, and with a faster, well-characterized, and sensitive Western blot.
The Western blot below shows Scrapie infected sheep brain lysates (Lanes 3&4) compared to normal sheep brain lysates (Lanes 1&2). The key to this particular Western assay is the use of Proteinase (PK) treatment on parallel sets of samples. Since the Scrapie associated form of the prion protein is resistant to digestion, it is possible to distinguish between normal cellular forms and abnormal prion protein in a sample based upon their sensitivity to PK.
Thus, it is possible to discriminate diseased samples from normal ones quickly and unambiguously on a Western blot. The absence of bands in lane 2 indicates that the normal form is present as compared to the bands visible in lane 4, which indicate the presence of the pathogenic form.
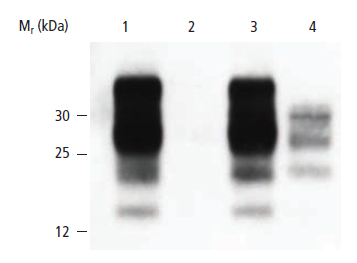
Figure 18: Detection of Scrapie Infected Sheep Brain. Bands in lane 4 indicate that the protease resistant form of the prion protein is present in the sample as compared to normal tissue in lane 2. Lanes 1&2: Uninfected sheep brain homogenate. Lanes 3&4: Scrapie-infected sheep brain homogenate. Lanes 1&3: No PK added. Lanes 2&4: Digested with PK.
Confirmation of HIV
In the diagnosis of HIV infection in patients, an ELISA is used first because it demonstrates 99 .5% specificity and is quick and easy to perform on a large number of samples. Western blotting is used as a supplementary assay because the ELISA is subject to false positives.
In order to perform this assay, patient serum samples are used as the source of antibodies for immunodetection, and blots containing HIV antigens, proteins, viral lysates, or peptides are used as the source of target antigen on the Western blot. If the HIV target proteins on the blot are detected by the patient serum then it indicates that the patient sample is a true positive for HIV infection, since an immune response has been mounted by the patient. A similar strategy is also used in tests for Lyme disease and autoimmune disease.
| Western Blot Example: Demonstrating Antibody Specificity. | Chapter 5: Western Blot Buffers |



