Cancer Associated Fibroblasts: An Introductory Guide
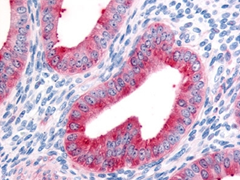
Fig. 1. Staining of formalin fixed, paraffin embedded human uterus with Rabbit Anti-Human FAP Alpha (AHP1322).
The Stromal Cells of the Tumor Microenvironment
The contribution of the tumor microenvironment to the development, progression and resistance to treatment in cancer is increasingly becoming clear. In breast cancer, tumors present with significant alterations to the surrounding tissue or microenvironment. Depending on the type of breast cancer, these environmental changes can include increased production of fibrous tissue (desmoplasia) as well as transformation of many cell types including fibroblasts and adipocytes.
In addition to neoplastic cells, the breast tumor microenvironment consists of extracellular matrix (ECM) and numerous stromal cell types, including endothelial cells, immune cells, fibroblasts, and adipocytes. Of the cell types, the most abundant and perhaps most significant are the cancer associated fibroblasts (CAFs). CAFs are identified using several markers including fibroblast activating protein (FAP) which is also present in the uterus and other organs (Figure 1).
Cancer Associated Fibroblasts
CAF is a general term that describes activated fibroblasts of different origin which express differing markers depending on their location (Sugimoto et al. 2006). Among the potential sources of CAFs are local activated fibroblasts, mesenchymal stem cells and cells which have undergone epithelial to mesenchymal transition (EMT). Fibroblasts are the primary cell type of connective tissue and form the extracellular matrix. Local non activated fibroblasts can undergo activation by tumor cells which secrete TGF-beta and SDF-1 (Kojima et al. 2010)(Figure 2).

Fig. 2. Development of cancer associated fibroblasts.
CAFs express smooth muscle actin alpha (actin alpha), PDGF receptor alpha (CD140a), PDGF receptor beta (CD140b), fibroblast specific protein 1(FSP-1/S100A4) and fibroblast activation protein (FAP), all of which have been used as markers to identify CAFs.
Role of CAFs
CAFs influence tumor development and progression by secreting many growth factors, interleukins and matrix metalloproteases (MMPs). Several of the markers, secreted factors and relevant pathways are listed in Table 1.
Table 1. CAF markers, secreted factors, MMPs and associated pathways.
| Markers | Factors Secreted | MMPs | Associated Pathways | |
|---|---|---|---|---|
| Cancer-associated fibroblasts |
Smooth Muscle Actin Alpha |
HGF |
MMP9 |
TGF/SMAD |
Once transformed to their activated phenotype, CAFs promote tumor growth and metastasis. This is in contrast to disease free fibroblasts which exert an anti-tumorigenic effect (Olumi et al. 1999). The tumor promotion occurs through the release of factors that promote cell growth and recruit new cells into the tumor microenvironment. SDF-1 (CXCL12) promotes survival of cancer cells and is a chemoattractant for other stromal cells (Orimo et al. 2005). IL-6 is a pro-inflammatory cytokine which is produced in substantial amounts by CAFs. IL-6 produced by CAFs also promoted tumor development in a cell model system (Osuala et al. 2015).
In addition to promoting tumor development by enhancing proliferation and cell recruitment, CAFs also release pro-angiogenic factors. Release of VEGF and PDGF cause recruitment and proliferation of endothelial cells leading to new blood vessel development (Taddei et al. 2014).
CAFs also help the tumor cells to metastasize to secondary sites. One method of achieving this is through their release of TGF-beta. TGF-beta signaling is capable of transforming stationary endothelial cells into mobile mesenchymal cells by EMT (Massague 2008). Similarly hepatocyte growth factor (HGF) can also induce EMT enabling cells to migrate more efficiently. In addition to creating a more mobile cell, CAFs can also edit the ECM to further facilitate invasion and metastasis. Release and activation of MMPs (MMP9 and MMP14) degrade type IV and V collagens and other extracellular matrix proteins enabling cancer cell migration.
CAFs and Treatment Resistance
Recent evidence has shown that CAFs are capable of shielding tumors from the effect of various treatments. Type I collagen secreted by CAFs creates a physical barrier between the tumor cells and the drugs diffusing from the blood stream. CAFs also mediated treatment resistance in response to gefitinib through activation of Met and EGF R/Met pathway crosstalk (Mueller et al. 2012). Two further studies have shown that CAFs mediated resistance to anti-angiogenic therapy (Pietras et al. 2008) and Cetuximab (Johansson et al. 2012).
CAF Related Products
CAFs are primarily identified by immunohistochemistry on either paraffin on frozen sections of human tissue. Table 2 shows the IHC validated antibodies against CAF markers.
Table 2. IHC/IF validated antibodies against CAF markers.
Target |
Applications |
Catalog # |
|---|---|---|
|
Alpha Smooth Muscle Actin |
IHC-F, E, IF, IHC-P, WB |
|
|
FAP Alpha |
IHC-P |
|
|
FSP-1 (S100A4) |
IF, WB |
Abbreviations: FC, Flow Cytometry; E, ELISA; IF, immunofluorescence; IHC-F, immunohistochemistry frozen; IHC-P, immunohistochemistry paraffin ; WB, western blotting.
CAFs can also be identified by flow cytometry. Combining the detectable expression of a CAF marker such as CD140b (Table 3) with the lack of other cell specific markers (for instance CD3, and CD45) may be required to ensure detection of CAFs.
Table 3. FC validated antibodies against CAF markers.
Target |
Applications |
Catalog # |
|---|---|---|
|
CD36 |
IHC-F, FC, FN |
Abbreviations: FC, Flow Cytometry; E, ELISA; FN, functional assay; IF, immunofluorescence; IHC-F, immunohistochemistry frozen; IHC-P, immunohistochemistry paraffin ; WB, western blotting.
Many of the signaling pathways that affect activation of CAFs and their subsequent behavior remain to be elucidated. A systems biology approach requires western blot validated antibodies against whole proteins and phosphorylated residues (Table 4).
Table 4. WB validated antibodies against CAF related products.
Target |
Applications |
Catalog # |
|---|---|---|
|
TGF BETA |
IHC-F, E, FC, IHC-P, WB |
|
|
SDF-1 alpha |
E, FN, IHC-P, WB |
Abbreviations: FC, Flow Cytometry; E, ELISA; FN, functional assay; IF, immunofluorescence; IHC-F, immunohistochemistry frozen; IHC-P, immunohistochemistry paraffin ; WB, western blotting.
References
- Johansson AC et al. (2012). Cancer-associated fibroblasts induce matrix metalloproteinase-mediated cetuximab resistance in head and neck squamous cell carcinoma cells. Mol Cancer Res. 10, 1158-1168.
- Kojima Y et al. (2010). Autocrine TGF-beta and stromal cell-derived factor-1 (SDF-1) signaling drives the evolution of tumor-promoting mammary stromal fibroblasts. PNAS 107, 20009-20014.
- Massague J (2008). TGFbeta in Cancer. Cell 134, 215-230.
- Mueller KL et al. (2012). Fibroblast-secreted hepatocyte growth factor mediates epidermal receptor tyrosine kinase inhibitor resistance in triple negative breast cancers through paracrine activation of Met. Breast Cancer Res. 14, 104.
- Olumi AF et al. (1999). Carcinoma-associated fibroblasts direct tumor progression of initiated human prostatic epithelium. Cancer Res. 59, 5002-11.
- Ormio A et al. (2005). Stromal fibroblasts present in invasive breast carcinomas promote tumor growth and angiogenesis through elevated CXCL12/SDF-1. Cell 121, 435-440.
- Osuala K et al. (2015). IL-6 signaling between ductal carcinoma in situ cells and carcinoma associated fibroblasts mediates tumor cell growth and migration. BMC Cancer 584.
- Pietras K et al. (2008). Functions of paracrine PDGF signaling in the proangiogenic tumor stroma revealed by pharmacological targeting. PLoS Med. 5, e19.
- Sugimoto H et al. (2006). Identification of fibroblast heterogeneity in the tumor microenvironment. Cancer Bio. Ther. 5, 1640-1646.
- Taddei ML et al. (2014). Senescent stroma promotes prostate cancer progression: the role of miR-210. Mol Oncol 8, 1729-1746.




