CD4 antibody | CVS4
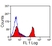
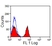
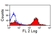





Mouse anti Horse CD4
- Product Type
- Monoclonal Antibody
- Clone
- CVS4
- Isotype
- IgG1
- Specificity
- CD4
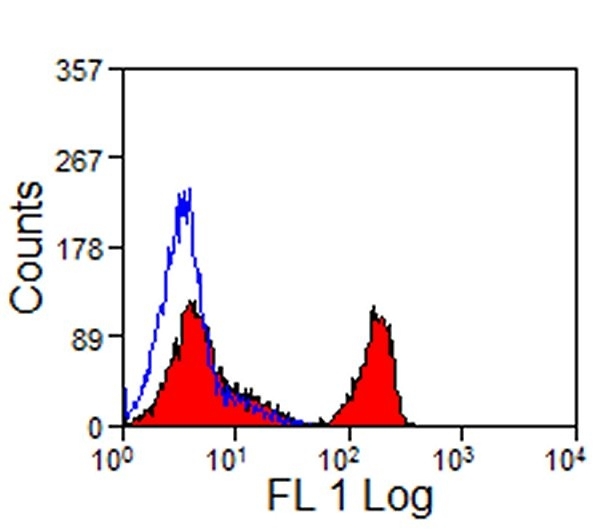
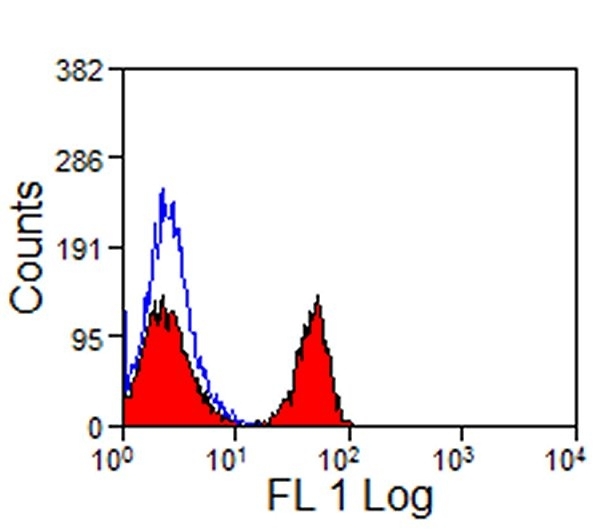
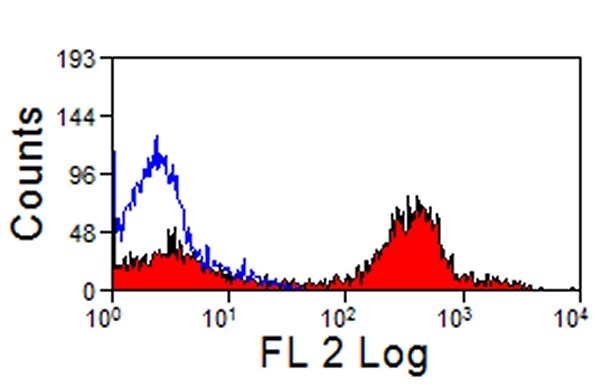
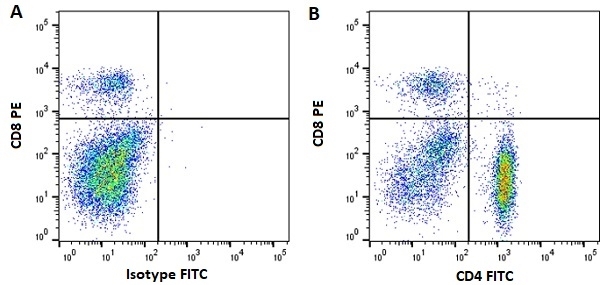
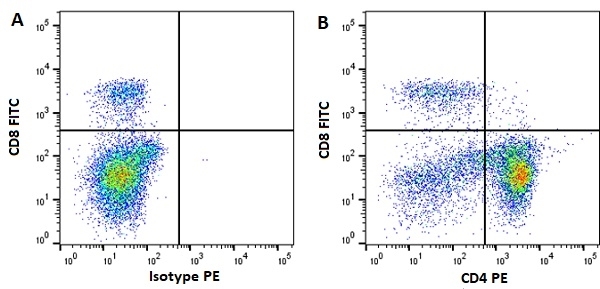
| Mouse anti Horse CD4 antibody, clone CVS4 recognizes equine CD4, a ~58 kDa cell surface glycoprotein that is primarily expressed on a subpopulation of T lymphocytes. As in humans, equine CD4 expression is mutually exclusive with CD8 expression on mature T-cells A study undertaken using Mouse anti Horse CD4, clone CVS4 to identify CD4 on several wild african equid species indicates that the CVS4 clone recognizes Somali wild ass (Equus asinus) but not Grévy's Zebra (E. grevyi) or Hartmann's Mountain Zebra (E. zebra). In addition to the CVS4 clone, other CVS clones recognizing equine cell surface and MHC antigen are available from Bio-Rad. |
- Target Species
- Horse
- Product Form
- Purified IgG - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% sodium azide (NaN3)
- Carrier Free
- Yes
- Immunogen
- Equine thymocytes.
- Approx. Protein Concentrations
- IgG concentration 1.0mg/ml
- Fusion Partners
- Spleen cells from immunized BALB/c mice were fused with cells of the X63-Ag 8.653 mouse myeloma cell line.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | 1/25 | 1/200 | |
| Immunohistology - Frozen | |||
| Immunoprecipitation |
- Flow Cytometry
- Use 10μl of the suggested working dilution to label 106 cells in 100μl
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse anti Horse CD8:FITC | MCA2385F | F | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse anti Horse CD8:FITC | ||||||
| Mouse anti Horse CD8:RPE | MCA2385PE | F | 100 Tests | Log in | |||
| List Price | Your Price | ||||||
| Log in | |||||||
| Description | Mouse anti Horse CD8:RPE | ||||||
References for CD4 antibody
-
Lunn, D.P. et al. (1991) Three monoclonal antibodies identifying antigens on all equine T lymphocytes, and two mutually exclusive T-lymphocyte subsets.
Immunology. 74 (2): 251-7. -
Kydd, J. et al. (1994) Report of the First International Workshop on Equine Leucocyte Antigens, Cambridge, UK, July 1991.
Vet Immunol Immunopathol. 42 (1): 3-60. -
Lunn, D.P. et al. (1998) Report of the Second Equine Leucocyte Antigen Workshop, Squaw valley, California, July 1995.
Vet Immunol Immunopathol. 62: 101-143 -
Roberto Da Costa, R.P. et al. (2003) Peripheral blood neutrophil function and lymphocyte subpopulations in cycling mares.
Reprod Domest Anim. 38 (6): 464-9. -
Deeg,C.A. et al. (2004) The uveitogenic potential of retinal S-antigen in horses.
Invest Ophthalmol Vis Sci. 45: 2286-92 -
Ferreira-Dias, G. et al. (2005) Seasonal reproduction in the mare: possible role of plasma leptin, body weight and immune status.
Domest Anim Endocrinol. 29 (1): 203-13. -
Garcia-Tapia, D. et al. (2006) Replication of West Nile virus in equine peripheral blood mononuclear cells.
Vet Immunol Immunopathol. 110 (3-4): 229-44. -
Goodman, L.B. et al. (2007) A point mutation in a herpesvirus polymerase determines neuropathogenicity.
PLoS Pathog. 3(11):e160.
View The Latest Product References
-
de Bruijn, C.M. et al. (2007) Clinical, histopathological and immunophenotypical findings in five horses with cutaneous malignant lymphoma.
Res Vet Sci. 83 (1): 63-72. -
Pearson, W. et al. (2007) Low-dose ginseng (Panax quinquefolium) modulates the course and magnitude of the antibody response to vaccination against equid herpesvirus I in horses.
Can J Vet Res. 71: 213-7. -
Lai SW et al. (2004) Influence of Ganoderma lucidum on blood biochemistry and immunocompetence in horses.
Am J Chin Med. 32 (6): 931-40. -
Ibrahim, S. et al. (2007) Screening of anti-human leukocyte monoclonal antibodies for reactivity with equine leukocytes.
Vet Immunol Immunopathol. 119 (1-2): 63-80. -
Agrícola, R. et al. (2008) Blood lymphocyte subpopulations, neutrophil phagocytosis and proteinogram during late pregnancy and postpartum in mares.
Reprod Domest Anim. 43 (2): 212-7. -
Brault, S.A. et al. (2010) The immune response of foals to natural infection with equid herpesvirus-2 and its association with febrile illness.
Vet Immunol Immunopathol. 137: 136-41. -
Go, Y.Y. et al. (2010) Complex interactions between the major and minor envelope proteins of equine arteritis virus determine its tropism for equine CD3+ T lymphocytes and CD14+ monocytes.
J Virol. 84: 4898-911 -
Hamza, E. et al. (2011) Equine CD4(+) CD25(high) T cells exhibit regulatory activity by close contact and cytokine-dependent mechanisms in vitro.
Immunology. 134 (3): 292-304. -
Hamza, E.et al. (2012) CD4+CD25+ T cells expressing FoxP3 in Icelandic horses affected with insect bite hypersensitivity.
Vet Immunol Immunopathol. 148 (1-2): 139-44. -
Khol-Parisini, A. et al. (2012) Highly deoxynivalenol contaminated oats and immune function in horses.
Arch Anim Nutr. 66 (2): 149-61. -
Uner, A. G. et al. (2013) Blood Levels of Selected Metabolic Factors, Cytokines, and Lymphocyte Subpopulations in Arabian and Thoroughbred Horses During the Longest and Shortest Days of the Year
J Equine Vet Sci. 33 (11): 969-976. -
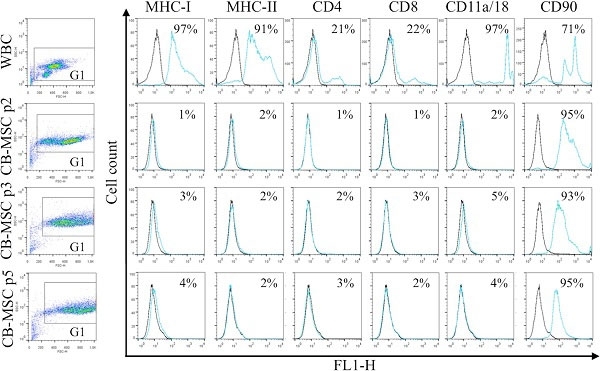
Tessier, L. et al. (2015) Phenotypic and immunomodulatory properties of equine cord blood-derived mesenchymal stromal cells.
PLoS One. 10 (4): e0122954. -
Behrens, N.E. & Gershwin, L.J. (2015) Immune modulation of T regulatory cells and IgE responses in horses vaccinated with West Nile virus vaccine combined with a CpG ODN.
Vaccine. 33 (43): 5764-71. -
Ziegler, A. et al. (2016) Identification and characterization of equine blood plasmacytoid dendritic cells.
Dev Comp Immunol. 65: 352-7. -
Krakowski. L. et al. (2017) Changes in Blood Lymphocyte Subpopulations and Expression of MHC-II Molecules in Wild Mares Before and After Parturition.
J Vet Res. 61 (2): 217-21. -
Degroote, R.L. et al. (2017) Formin like 1 expression is increased on CD4+ T lymphocytes in spontaneous autoimmune uveitis.
J Proteomics. 154: 102-108. -
Schauer, M. et al. (2018) Interaction of septin 7 and DOCK8 in equine lymphocytes reveals novel insights into signaling pathways associated with autoimmunity.
Sci Rep. 8 (1): 12332. -
Tomlinson, J.E. et al. (2018) Multispectral fluorescence-activated cell sorting of B and T cell subpopulations from equine peripheral blood.
Vet Immunol Immunopathol. 199: 22-31. -
Witonsky, S. et al. (2019) Can levamisole upregulate the equine cell-mediated macrophage (M1) dendritic cell (DC1) T-helper 1 (CD4 Th1) T-cytotoxic (CD8) immune response in vitro.?
J Vet Intern Med. 33 (2): 889-96. -
Marteles, D. et al. (2019) Effects of allergen-specific immunotherapy on peripheral blood regulatory T cells and serum concentrations of cytokines and immunoglobulins in horses with allergic dermatitis.
Int Immunopharmacol. 74: 105674. -
Hillmann, A. et al. (2019) A novel direct co-culture assay analyzed by multicolor flow cytometry reveals context- and cell type-specific immunomodulatory effects of equine mesenchymal stromal cells.
PLoS One. 14 (6): e0218949. -
Placci, M. et al. (2020) Natural Horse Boarding Vs Traditional Stable: A Comparison of Hormonal, Hematological and Immunological Parameters.
J Appl Anim Welf Sci. 23 (3): 366-77. -
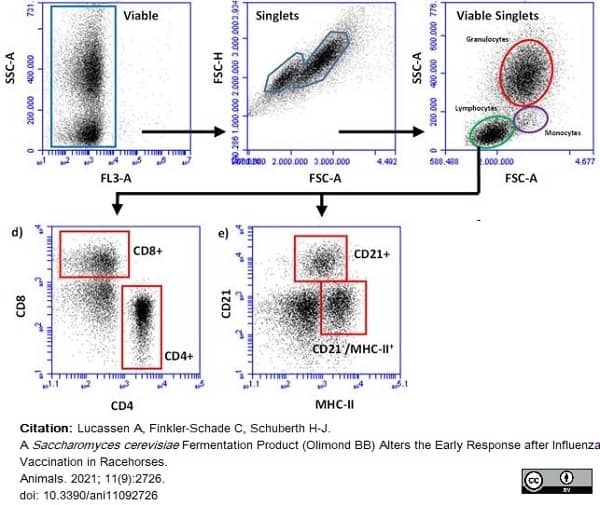
Lucassen, A. et al. (2021) A Saccharomyces cerevisiae Fermentation Product (Olimond BB) Alters the Early Response after Influenza Vaccination in Racehorses.
Animals (Basel). 11(9):2726. -
Cequier, A. et al. (2022) Equine Mesenchymal Stem Cells Influence the Proliferative Response of Lymphocytes: Effect of Inflammation, Differentiation and MHC-Compatibility.
Animals (Basel). 12 (8): 984. -
Townsend, K.S. et al. (2023) Concurrent chronic lymphocytic leukemia and primary hyperparathyroidism in a mule.
J Vet Intern Med. 37 (3): 1250-5. -
Hartmann, C. et al. (2018) Influences of intrauterine semen administration on regulatory T lymphocytes in the oestrous mare (Equus caballus).
Theriogenology. 118: 119-25. -
Siqueira, R.F. & Fernandes, R.L. (2018) Cryopreservation of lymphocytes for immunological studies in horses
Pesquisa Veterinária Brasileira. 38 (11): 2019-22. -
Bourebaba, L. et al. (2023) The PTP1B inhibitor MSI-1436 ameliorates liver insulin sensitivity by modulating autophagy, ER stress and systemic inflammation in Equine metabolic syndrome affected horses.
Front Endocrinol (Lausanne). 14: 1149610. -
Cabezas, J. et al. (2020) In vitro preconditioning of equine adipose mesenchymal stem cells with prostaglandin E(2), substance P and their combination changes the cellular protein secretomics and improves their immunomodulatory competence without compromising stemness.
Vet Immunol Immunopathol. 228: 110100. -
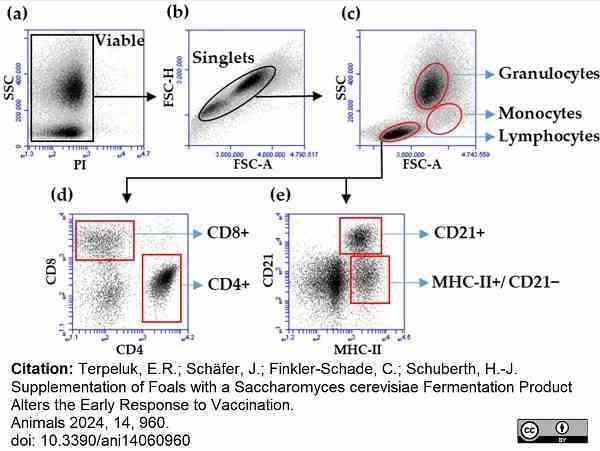
Terpeluk, R.E. et al. (2024) Supplementation of Foals with a Saccharomyces cerevisiae Fermentation Product Alters the Early Response to Vaccination
Animals. 14 (6): 960.
- UniProt
- F6Y6X8
MCA1078GA
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Horse ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up