IgA antibody | K84 2F9

Mouse anti Bovine/Ovine IgA
- Product Type
- Monoclonal Antibody
- Clone
- K84 2F9
- Isotype
- IgG1
- Specificity
- IgA
| Mouse anti Bovine IgA antibody, clone K84 2F9 recognizes bovine and ovine IgA. No cross reactivity is observed with Bovine or Ovine IgG and IgM, |
- Target Species
- Bovine
- Species Cross-Reactivity
-
Target Species Cross Reactivity Sheep - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% Sodium Azide (NaN3)
- Carrier Free
- Yes
- Immunogen
- Bovine IgA.
- Approx. Protein Concentrations
- IgG concentration 1.0 mg/ml
- Fusion Partners
- Spleen cells from immunized mice were fused with cells of the P3.X63.Ag8.653 myeloma cell line.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
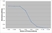
| ELISA | 1/5000 | 1/80,000 |
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse anti Bovine IgA | MCA2438GA | E IP | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse anti Bovine IgA | ||||||
| Mouse anti Bovine IgA:HRP | MCA2438P | E | 0.25 mg | Log in | |||
| List Price | Your Price | ||||||
| Log in | |||||||
| Description | Mouse anti Bovine IgA:HRP | ||||||
| Mouse anti Bovine IgG | MCA2439GA | E IP | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse anti Bovine IgG | ||||||
| Mouse anti Bovine IgG:HRP | MCA2439P | E | 0.25 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse anti Bovine IgG:HRP | ||||||
| Mouse anti Bovine IgG1 | MCA2440GA | C E F IP | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse anti Bovine IgG1 | ||||||
| Mouse anti Bovine IgG1:HRP | MCA2440P | E | 0.25 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse anti Bovine IgG1:HRP | ||||||
| Mouse anti Bovine IgG2 | MCA2441GA | E F IP | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse anti Bovine IgG2 | ||||||
| Mouse anti Bovine IgG2:HRP | MCA2441P | E | 0.25 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse anti Bovine IgG2:HRP | ||||||
| Mouse anti Bovine IgM | MCA2443GA | E F IP R | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse anti Bovine IgM | ||||||
| Mouse anti Sheep IgE | MCA5941GA | C E IF WB | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse anti Sheep IgE | ||||||
| Mouse anti Bovine IgG1 | MCA627GA | E | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse anti Bovine IgG1 | ||||||
References for IgA antibody
-
Estes, D.M. et al. (1998) Effects of type I/type II interferons and transforming growth factor-beta on B-cell differentiation and proliferation. Definition of costimulation and cytokine requirements for immunoglobulin synthesis and expression.
Immunology. 95 (4): 604-11. -
Suraud, V. et al. (2008) Acute infection by conjunctival route with Brucella melitensis induces IgG+ cells and IFN-gamma producing cells in peripheral and mucosal lymph nodes in sheep.
Microbes Infect. 10: 1370-8. -
Parreño, V.et al. (2004) Modulation by colostrum-acquired maternal antibodies of systemic and mucosal antibody responses to rotavirus in calves experimentally challenged with bovine rotavirus.
Vet Immunol Immunopathol. 100 (1-2): 7-24. -
Hassan, M. et al. (2011) The dynamic influence of the DRB1*1101 allele on the resistance of sheep to experimental Teladorsagia circumcincta infection.
Vet Res. 42: 46. -
Stanley AC et al. (2004) Intranasal immunisation with Toxoplasma gondii tachyzoite antigen encapsulated into PLG microspheres induces humoral and cell-mediated immunity in sheep.
Vaccine. 22 (29-30): 3929-41. -
Tzelos, T. et al. (2016) A preliminary proteomic characterisation of extracellular vesicles released by the ovine parasitic nematode, Teladorsagia circumcincta..
Vet Parasitol. 221: 84-92. -
Mahajan, A. et al. (2005) Phenotypic and functional characterisation of follicle-associated epithelium of rectal lymphoid tissue.
Cell Tissue Res. 321 (3): 365-74. -
Suraud, V. et al. (2007) Differential expression of homing receptors and vascular addressins in tonsils and draining lymph nodes: Effect of Brucella infection in sheep.
Vet Immunol Immunopathol. 115 (3-4): 239-50.
View The Latest Product References
-
McNeilly, T.N. et al. (2007) Simple methods for measurement of bovine mucosal antibody responses in vivo.
Vet Immunol Immunopathol. 118 (1-2): 160-7. -
McNeilly, T.N. et al. (2010) IgA and IgG antibody responses following systemic immunization of cattle with native H7 flagellin differ in epitope recognition and capacity to neutralise TLR5 signalling.
Vaccine. 28 (5): 1412-21. -
McNeilly, T.N. et al. (2010) Immunization of cattle with a combination of purified intimin-531, EspA and Tir significantly reduces shedding of Escherichia coli O157:H7 following oral challenge.
Vaccine. 28 (5): 1422-8. -
Mahajan, A. et al. (2009) An investigation of the expression and adhesin function of H7 flagella in the interaction of Escherichia coli O157 : H7 with bovine intestinal epithelium.
Cell Microbiol. 11 (1): 121-37. -
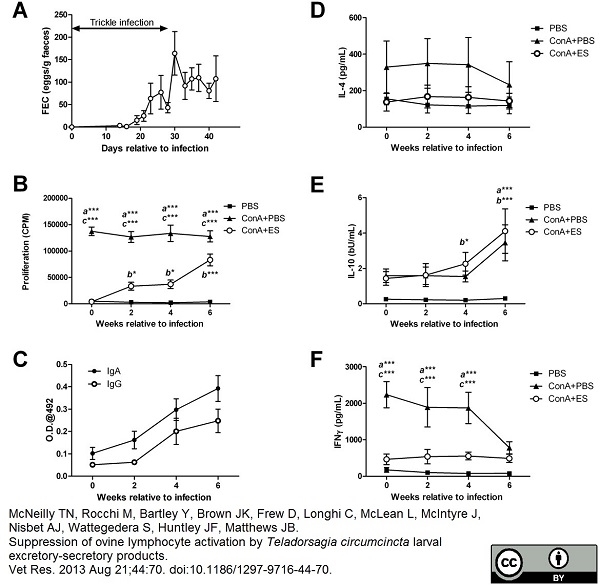
McNeilly, T.N. et al. (2013) Suppression of ovine lymphocyte activation by Teladorsagia circumcincta larval excretory-secretory products.
Vet Res. 44: 70. -
Nisbet, A.J. et al. (2016) Protection of ewes against Teladorsagia circumcincta. infection in the periparturient period by vaccination with recombinant antigens.
Vet Parasitol. 228: 130-6. -
Queiroga, M.C. (2018) Local and systemic humoral response to ovine mastitis caused by Staphylococcus epidermidis.
SAGE Open Med. 6: 2050312118801466. -
McBean, D. et al. (2023) Immune markers in goats selected for reduced gastrointestinal nematode egg count under artificial infection conditions.
Vet Parasitol. 322: 110004. -
Palarea-Albaladejo, J. et al. (2024) A curated multivariate approach to study efficacy and optimisation of a prototype vaccine against teladorsagiasis in sheep.
Vet Res Commun. 48 (1): 367-79.
MCA628GA
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Bovine ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up