ED Clone Antibodies for Studying Rat Macrophage Subtype Markers and Related Molecules

- On This Page
- Overview
- Clone ranges
- Further reading
Overview
ED clone anti-rat antibodies have been specifically designed to identify macrophages. However a number of ED clones are also specific for other cell types such as myeloid cells and reticular elements in secondary lymphoid tissue. Bio-Rad is the primary manufacturer of the complete range of ED clone antibodies.
ED1 (CD68)
Monoclonal Antibody (MCA341) clone ED1, is the widely used pan-macrophage marker recognizing CD68, a single chain glycoprotein of 90-110 kDa. CD68 staining is predominantly found on the lysosomal membrane of myeloid cells, where there is also weak cell surface expression. The majority of tissue macrophages express CD68, while peripheral granulocytes only stain weakly. Expression levels of CD68 correlate with the phagocytic activity of macrophages. Refer to Table 1 for a list of all formats available for MCA341 and the validated applications.
ED2 (CD163)
Monoclonal Antibody (MCA342) clone ED2, recognizes CD163, a 175 kDa cell surface glycoprotein present on resident rat macrophages. This antibody is a useful tool for staining synovial lining and Kupffer cells in the liver. It can also be used to discriminate between thymic cortical macrophages (ED2+ve) and thymic medullary macrophages (ED2-ve). However, CD163 is not expressed by monocytes, alveolar macrophages, or microglial cells. Refer to Table 1 for a list of all formats available for MCA342 and the validated applications.
ED3 (CD169)
Monoclonal Antibody (MCA343) clone ED3, recognizes the rat CD169 cell surface antigen, a 185 kDa molecule expressed by macrophages predominately confined to lymphoid organs. CD169 is a receptor for glycoconjugates containing sialic acid. The ED3 antibody stains marginal zone macrophages and marginal metallophils in the spleen very strongly. CD169 is also expressed in macrophages in auto-immune diseased tissues, such as those associated with multiple sclerosis. Refer to Table 1 for a list of the formats available for MCA343 and the validated applications.
ED7 and ED8 (CD11b)
The ED7 clone antibody (MCA618R) recognizes a membrane antigen, CD11b, on rat macrophages, monocytes, dendritic cells, and granulocytes. The recognized antigen is a 160 kDa/95 kDa heterodimer belonging to the CD11b/CD18 family of adhesion molecules and is also known as Mac-1 or CR3. ED8 (MCA619R) is believed to recognize a second epitope from the same molecule.
ED7 and ED8 clone antibodies have been shown to detect small ramified microglia in the central nervous system as well as cilia of the bronchus epithelium. No other cell types are positive for staining. Both ED7 and ED8 can be used to induce homotypic aggregation of granulocytes. Refer to Table 1 for a list of all the formats available for MCA618R and MCA619 and the validated applications.
Rat markers CD11b and CD106 have been used in flow cytometry (Figure 1), showing myeloid cell populations. Below this immunofluorescence staining of CD11b alongside CD4 in rat spleen is shown, showing macrophages and T cells.
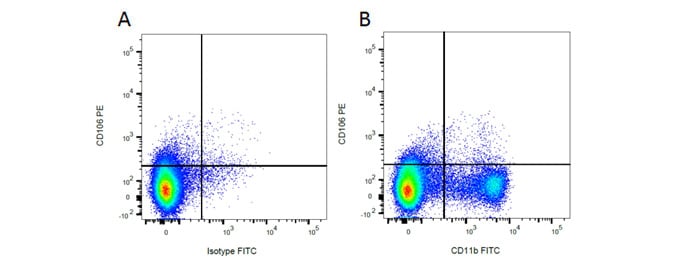
Fig. 1. Flow cytometry analysis of rat makers CD11b and CD106. A, PE conjugated Mouse Anti-Rat CD106 (MCA4633PE) and FITC conjugated Mouse IgG2a Isotype Control (MCA1210F). B, PE conjugated Mouse Anti-Rat CD106 (MCA4633PE) and FITC conjugated Mouse Anti-Rat CD11b (MCA619F). All experiments performed on red cell lysed rat bone marrow gated on mononuclear cells.
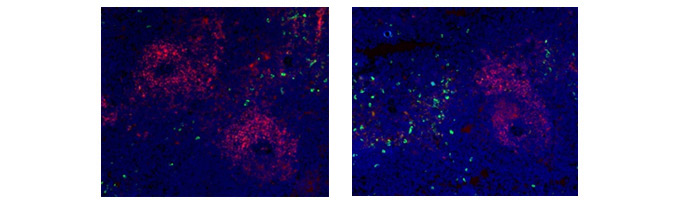
Fig. 2. Immunofluorescence analysis of rat marker CD11b and CD4. CD4+ T cells (red) stained with (MCA153) and CD11b positive macrophages (green) stained with (MCA619) in rat spleen (cryosection).
ED9 (CD172a)
Monoclonal Antibody (MCA620) clone ED9, recognizes rat CD172a, also known as signal regulatory protein (SIRP). CD172a is expressed by myeloid cells and neurons. The ED9 clone antibody binds to a different epitope on SIRP than the CD172a OX-41 (MCA274) antibody and has been reported to block the interaction of CD172a with CD47. Refer to Table 1 for a list of all the formats available for MCA620 and the validated applications.
Table 1. ED clone antibody formats and validated applications.
Antibody (catalog #) |
Formats |
Applications |
|---|---|---|
|
Pur., A488, A647, A700, B., FITC, PE |
FC, IF/ICC, IHC-F, IHC-P, IP, WB |
|
|
Pur., FITC, LE |
IF/ICC, IHC-F, IHC-P, IP, WB |
|
|
Pur. |
FC, IF/ICC, IHC-F, IP |
|
|
Pur. |
FC, FN, IHC-F |
|
|
Pur., FITC, PE |
FC, IHC-F |
|
|
Pur., FITC, PE |
FC, IHC-F, IP, WB |
Abbreviations: B., Biotin; E, ELISA; FC, flow cytometry; FN, functional assay; IF/ICC, immunofluorescence/immunocytochemistry; IHC-F, immunohistochemistry-frozen; IHC-P, immunohistochemistry-paraffin; IP, immunoprecipitation; LE, low endotoxin; Pur., purified; WB, western blotting.
Further Reading
Note more references are available on the individual ED clone antibody datasheets.
ED1, ED2, and ED3
- Dijkstra CD et al. (1985). The heterogeneity of mononuclear phagocytes in lymphoid organs: distinct macrophage subpopulations in the rat recognized by monoclonal antibodies ED1, ED2 and ED3. Immunology 54, 589-599.
This reference has been cited in more than 200 publications.
ED5 and ED6
- Jeurissen SHM and Dijkstra CD (1986). Characteristics and functional aspects of non lymphoid cells in rat germinal centres, recognized by two monoclonal antibodies ED5 and ED6. Eur. J. Immunol 16, 562-568.
ED7, ED8, and ED9
- Damoiseaux JG et al. (1989). Heterogeneity of macrophages in the rat evidenced by variability in determinants: two new anti-rat macrophage antibodies against a heterodimer of 160 and 95 kd (CD11/CD18). J. Leukoc. Biol 46: 556-564.
- Damoiseaux JG et al. (1989). Rat bone marrow and monocyte cultures: influence of culture time and lymphokines on the expression of macrophage differentiation antigens. J Leukoc Biol 46:246-53.
ED10 and ED11
- Van den Berg TK et al. (1989). The heterogeneity of the reticulum of rat peripheral lymphoid organs identified by monoclonal antibodies. Eur. J. Immunol 19, 1747-1756.
- Van den Berg TK et al. (1992). Selective inhibition of immune complex trapping by follicular dendritic cells with monoclonal antibodies against rat C3. Eur. J. Immunol 22, 957-962.







