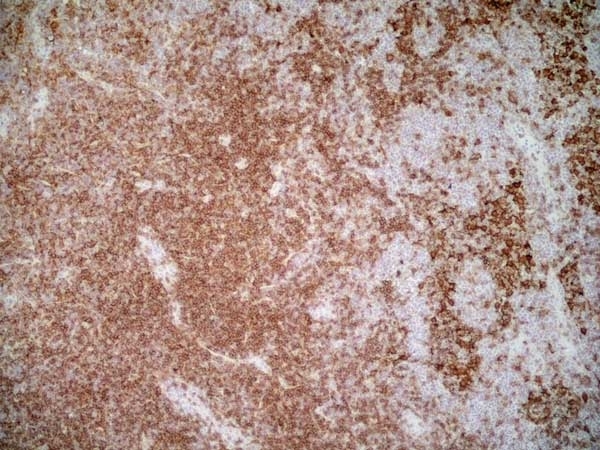
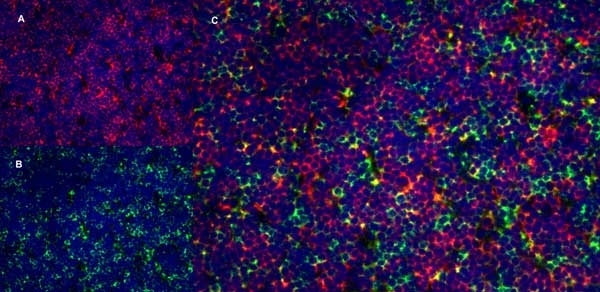
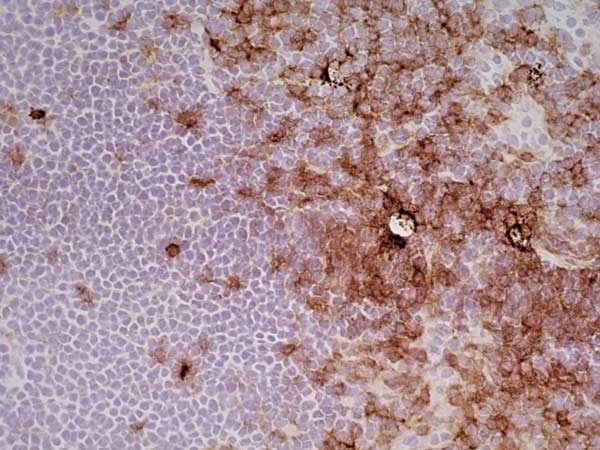
CD4 antibody | OX-35





Mouse anti Rat CD4 (Domain 2)
- Product Type
- Monoclonal Antibody
- Clone
- OX-35
- Isotype
- IgG2a
- Specificity
- CD4
- Region
- (DOMAIN 2)
| Mouse anti Rat CD4 (domain 2) antibody, clone OX-35 recognizes the rat CD4 cell surface antigen, a ~55kDa glycoprotein expressed by helper T cells and weakly by monocytes. Mouse anti Rat CD4 (Domain 2) antibody, clone OX-35 recognizes a different epitope on the CD4 molecule to Mouse anti Rat CD4 antibody, clone W3/25. |
- Target Species
- Rat
- Product Form
- Purified IgG - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
0.09% Sodium Azide - Carrier Free
- Yes
- Immunogen
- MLR generated rat T helper lymphocytes.
- Approx. Protein Concentrations
- IgG concentration 1.0 mg/ml
- Fusion Partners
- Spleen cells from immunised BALB/c mice were fused with cells of the mouse NS1 myeloma cell line.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | 1/10 | 1/50 | |
| Immunofluorescence | |||
| Immunohistology - Frozen | |||
| Immunoprecipitation |
- Flow Cytometry
- Use 10ul of the suggested working dilution to label 106 cells in 100ul.
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG2a Negative Control | MCA1210 | F | 100 Tests |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG2a Negative Control | ||||||
References for CD4 antibody
-
Wang, C.C. et al. (1996) Immunohistochemical study of amoeboid microglial cells in fetal rat brain.
J Anat. 189 ( Pt 3): 567-74. -
Jefferies, W.A. et al. (1985) Authentic T helper CD4 (W3/25) antigen on rat peritoneal macrophages.
J Exp Med. 162 (1): 117-27. -
Camelo, S. et al. (2004) The distribution of antigen in lymphoid tissues following its injection into the anterior chamber of the rat eye.
J Immunol. 172: 5388-95. -
Elflein, K. et al. (2003) Rapid recovery from T lymphopenia by CD28 superagonist therapy.
Blood. 102: 1764-70. -
Scherr, M. et al. (2002) Efficient gene transfer into the CNS by lentiviral vectors purified by anion exchange chromatography.
Gene Ther. 9: 1708-14. -
Cho, K.S. et al. (2010) Mechanism analysis of long-term graft survival by monocarboxylate transporter-1 inhibition.
Transplantation. 90: 1299-306. -
Chang, C.J. et al (2004) The immunization site of cytokine-secreting tumor cell vaccines influences the trafficking of tumor-specific T lymphocytes and antitumor efficacy against regional tumors.
J Immunol. 173: 6025-32. -
Basiri, M. and Doucette, R. (2010) Sensorimotor cortex aspiration: a model for studying Wallerian degeneration-induced glial reactivity along the entire length of a single CNS axonal pathway.
Brain Res Bull. 81: 43-52.
View The Latest Product References
-
Esquifino, A.I. et al. (2007) Immune response after experimental allergic encephalomyelitis in rats subjected to calorie restriction.
J Neuroinflammation. 4:6. -
Zhao, S. et al. (2007) Extensive FDG uptake and its modification with corticosteroid in a granuloma rat model: an experimental study for differentiating granuloma from tumors.
Eur J Nucl Med Mol Imaging 34: 2096-105. -
Yan, Y. et al. (2003) Pathogenesis of autoimmunity after xenogeneic thymus transplantation.
J Immunol. 170: 5936-46. -
Li, Q. et al. (2019) Dendritic cell-targeted CD40 DNA vaccine suppresses Th17 and ameliorates progression of experimental autoimmune glomerulonephritis
Journal of Leukocyte Biology. 27 Feb [Epub ahead of print]. -
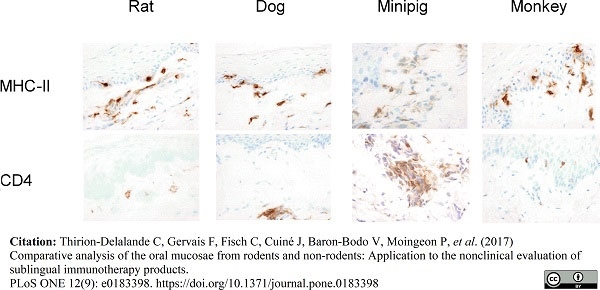
Thirion-Delalande, C. et al. (2017) Comparative analysis of the oral mucosae from rodents and non-rodents: Application to the nonclinical evaluation of sublingual immunotherapy products.
PLoS One. 12 (9): e0183398. -
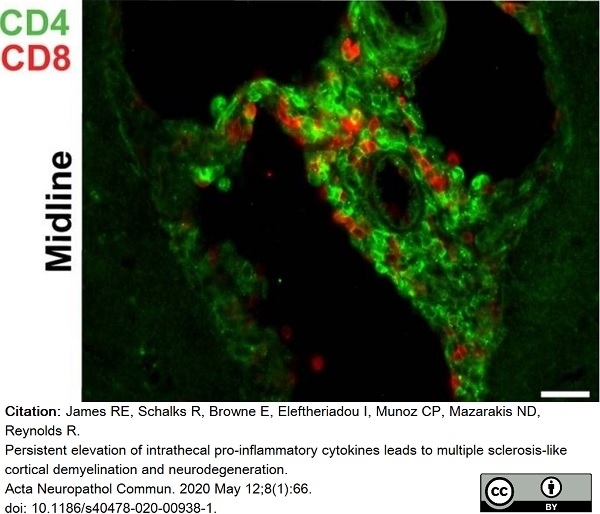
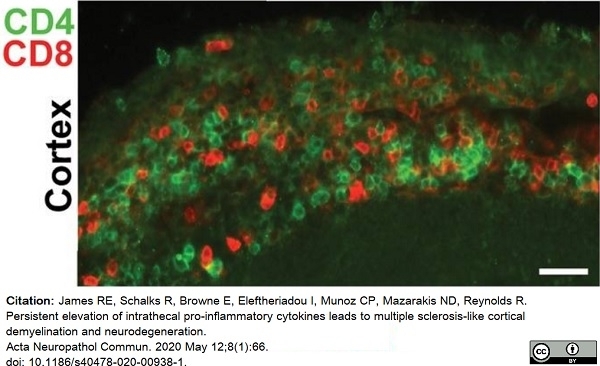
James, R.E. et al. (2020) Persistent elevation of intrathecal pro-inflammatory cytokines leads to multiple sclerosis-like cortical demyelination and neurodegeneration.
Acta Neuropathol Commun. 8 (1): 66. -
Dabrowska, S. et al. (2021) Neuroinflammation evoked by brain injury in a rat model of lacunar infarct.
Exp Neurol. 336: 113531. -
Gad, R.A. et al. (2022) Mitigating effects of Passiflora incarnata. on oxidative stress and neuroinflammation in case of pilocarpine-Induced status epilepticus model.
J King Saud Uni - Science. 34 (3): 101886.
- RRID
- AB_322831
- UniProt
- P05540
- Entrez Gene
- Cd4
- GO Terms
- GO:0007155 cell adhesion
- GO:0016021 integral to membrane
- GO:0006955 immune response
- GO:0019901 protein kinase binding
- GO:0045058 T cell selection
MCA153R
MCA153GA
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Rat ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up