Antibody Titration in Flow Cytometry
Why Is Antibody Titration Important?
To optimize staining in flow cytometry antibody titration is recommended. Whilst antibodies will bind to high affinity targets on a cell, if they are present in excess they will also bind to low affinity targets. This results in an increase in background fluorescence and consequently a reduction in your ability to resolve populations, especially if there are subtle differences. Furthermore if the antibody concentration is too high, it may result in a false negative prozone effect.
Essentials for Multicolor Panel Building Poster, incl. tips on antibody titration.

It is therefore important to use the antibodies at the right concentration. Although antibodies are sold with a recommended dilution and it is a good starting point, it may not be optimal for your cell type or protocol, therefore titration is an essential step to optimize your staining.
How to Titrate Your Antibodies
A titration experiment starts by selecting a fixed incubation time, cell type and experimental conditions. The last two should preferably match your final experiment. The cells are then stained in a series of dilutions of the antibody. It is good practice to add a viability dye even when titrating antibodies as dead cells can bind antibodies non-specifically, making your results hard to interpret. To determine the best antibody concentration, the stain index can be used as a guide (formula in Figure 1).
To obtain a stain index you must have a positive and a negative population (Figure 1). If all of your sample population is positive, for instance CD45 on human peripheral blood, then it is necessary to spike the population with some unstained cells. As with all flow cytometry experiments, don’t forget to include a single cell gate in your analysis to exclude doublets.
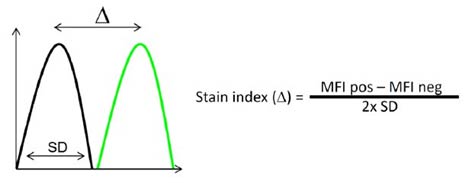
Fig. 1. Calculation of the index. The stain index is the ratio of the separation between the positive population (green) and the negative population (black), divided by two times the standard deviation of the negative population.
The dilution which gives the brightest staining with minimum background is the dilution to use. An example of how the stain index is used in a titration series can be seen in Figure 2 below, where 1 x 106 human peripheral blood was stained with CD56 with increasing dilutions of antibody, in 100 ml, and the stain index plotted. Titration of this antibody shows the optimal concentration to be 1/5.
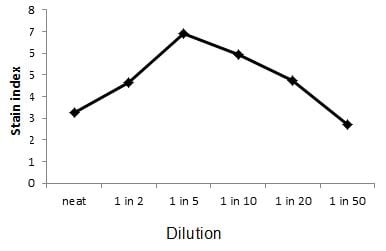
Fig. 2. Titration of CD56 on human blood. Human peripheral blood was stained with CD56 at increasing dilution and the stain index calculated and plotted as shown. The optimal separation of the positive and negative populations was observed at 1/5 dilution of the antibody stock solution.
An example of how antibody concentration may affect your staining can be seen in Figure 3. Human peripheral blood was stained for CD3 and CD55 and the cells within a mononuclear gate (panel A) assessed after gating to remove propidium iodide positive dead cells and doublets. Results from an untitrated staining (panel C) show two populations that are CD3 negative, whereas when the antibody was titrated (panel D) more populations are resolved. As the number of colors is increased, this ability to resolve populations becomes more difficult due to fluorescence spillover and spread, therefore titration is an essential starting point in panel building.
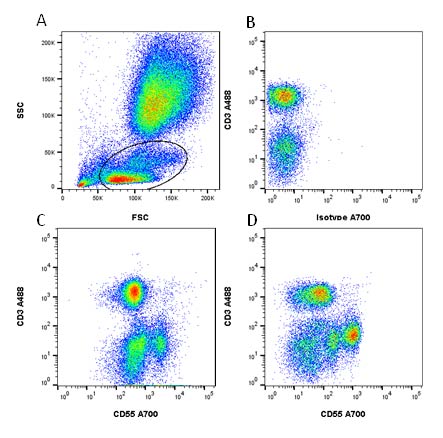
Fig. 3. Example of how titration can improve resolution. Human peripheral blood was stained for CD3 A488 (MCA463A488) and CD55 (MCA1614A700). A, mononuclear cells were identified for further analysis. B, the isotype control showed there was no non-specific binding of the antibody. C, when CD55 A700 was added neat to the cells, only two CD3 negative populations could be resolved, whereas when used at a 1/10 dilution, D, more populations were visible.
In conclusion, titration of antibodies will improve your experiments and could save you money if a lower antibody concentration is required than the one stated in the manufacturer's instructions. If you are using the same batch of antibody from a manufacturer then you should only need to titrate once. Similarly, if you increase your staining volume, simply increase the amount of antibody to keep the concentration the same avoiding re-titration. If you change batch, staining conditions or cell type it is recommended to re-titrate.
For more information and help with multicolor panel building, explore our resources below.
Resources for Multicolor Panel Building
Titration of your antibodies will not only improve your multicolor antibody panel experiments, but also any fluorescent staining in flow cytometry.
Apoptosis reagents, such as FLICA Caspase Assays and Mitochondrial Membrane Potential Kits, and cell cycle reagents, like CytoTrack™ Cell Proliferation Assays, have to be titrated to the right concentration in order to accurately measure changes that may occur. High concentrations of these reagents for instance, may mask changes and reduce the resolution. Finally antibody titration is recommended for all antibody based applications such as western blotting, immunofluorescence, immunohistochemistry and ELISA.
Bio-Rad offers over 4,000 antibodies validated for flow cytometry as well as resources to help you run and improve your flow cytometry experiments. The resources in addition to listings of antibodies, kits and controls can be accessed from our dedicated flow cytometry hub or selecting the links of interest below.








