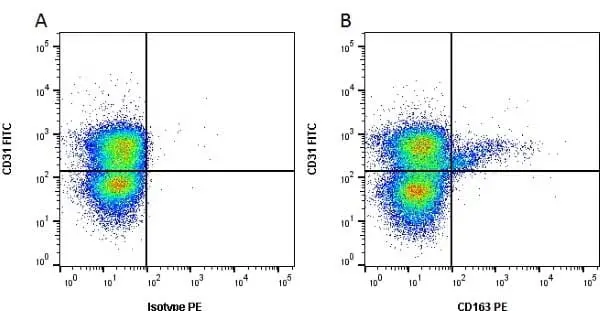
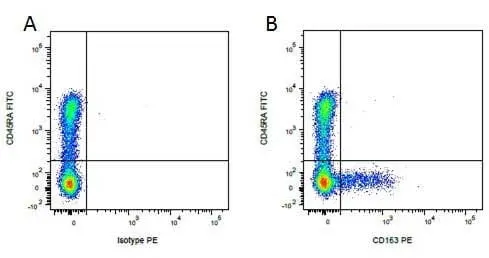
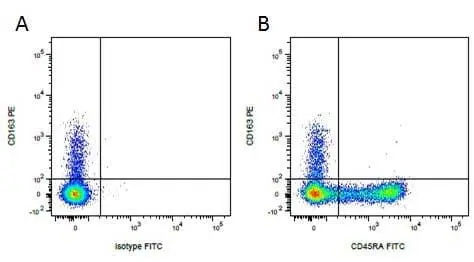
CD163 antibody | 2A10/11
Mouse anti Pig CD163
- Product Type
- Monoclonal Antibody
- Clone
- 2A10/11
- Isotype
- IgG1
- Specificity
- CD163
Filter by Application:
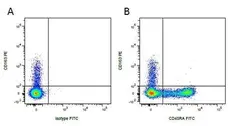
F IF WB C P Reset| Mouse anti Pig CD163 antibody, clone 2A10/11 recognises porcine CD163, a ~120 kDa single pass type 1 transmembrane cell surface glycoprotein expressed on cells of the monocyte/macrophage lineage. The expression levels of CD163 vary during the course of macrophage differentiation. The highest levels of CD163 expression are found on tissue macrophages but bone marrow derived cells are CD163 negative. Expression of CD163 on peripheral blood monocytes varies between about 5% and 50% depending on the donor (Sanchez et al. 1999). Mouse anti Pig CD163, clone 2A10/11 is reported to inhibit both African swine fever infection and viral particle binding to alveolar macrophages in a dose-dependent manner (Sanchez-Torres et al. 2003). |
- Target Species
- Pig
- Product Form
- Purified IgG - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% sodium azide (NaN3)
- Carrier Free
- Yes
- Immunogen
- Porcine alveolar macrophages.
- Approx. Protein Concentrations
- IgG concentration 1.0 mg/ml
- Fusion Partners
- Spleen cells from immunized BALB/c mice were fused with cells of the X63-Ag.8.653 myeloma cell line.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | 1/50 | 1/200 | |
| Immunofluorescence | |||
| Immunohistology - Frozen | |||
| Immunoprecipitation | |||
| Western Blotting 1 |
- 1Clone 2A10/11 recognizes porcine CD163 under non-reducing conditions.
- Flow Cytometry
- Use 10μl of the suggested working dilution to 1x106 cells in 100μl
- Western Blotting
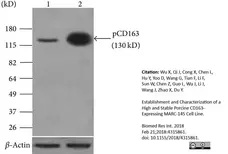
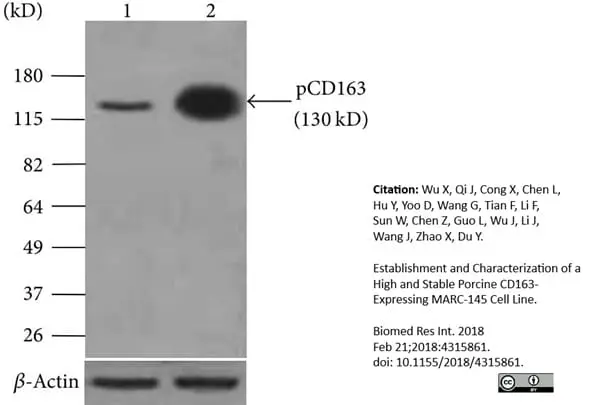
- Mouse anti Pig CD163 antibody, clone 2A10/11 detects a band of approximately 120 kDa in alveolar macrophage cell lysates under non-reducing conditions.
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG1 Negative Control | MCA928 | F | 100 Tests |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG1 Negative Control | ||||||
Source Reference
-
Bullido, R. et al. (1997) Monoclonal antibodies specific for porcine monocytes/macrophages: macrophage heterogeneity in the pig evidenced by the expression of surface antigens.
Tissue Antigens. 49 (4): 403-13.
Antibody Characterization Reference
-
Sánchez, C. et al. (1999) The porcine 2A10 antigen is homologous to human CD163 and related to macrophage differentiation.
J Immunol. 162 (9): 5230-7.
References for CD163 antibody
-
Gómez del Moral M et al. (1999) African swine fever virus infection induces tumor necrosis factor alpha production: implications in pathogenesis.
J Virol. 73 (3): 2173-80. -
Thacker, E. et al. (2001) Summary of workshop findings for porcine myelomonocytic markers.
Vet Immunol Immunopathol. 80 (1-2): 93-109. -
Yang, P. et al. (2002) Immune cells in the porcine retina: distribution, characterization and morphological features.
Invest Ophthalmol Vis Sci. 43 (5): 1488-92. -
Sánchez-Torres, C. et al. (2003) Expression of porcine CD163 on monocytes/macrophages correlates with permissiveness to African swine fever infection.
Arch Virol. 148 (12): 2307-23. -
Katchman, H. et al. (2008) Embryonic porcine liver as a source for transplantation: advantage of intact liver implants over isolated hepatoblasts in overcoming homeostatic inhibition by the quiescent host liver.
Stem Cells. 26: 1347-55. -
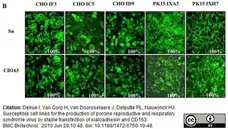
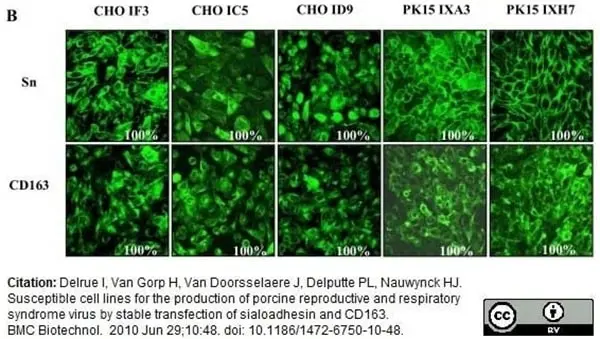
Delrue, I. et al. (2010) Susceptible cell lines for the production of porcine reproductive and respiratory syndrome virus by stable transfection of sialoadhesin and CD163.
BMC Biotechnol. 10: 48. -
Moreno, S.et al. (2010) Porcine monocyte subsets differ in the expression of CCR2 and in their responsiveness to CCL2.
Vet Res. 41: 76. -
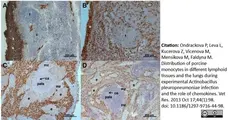
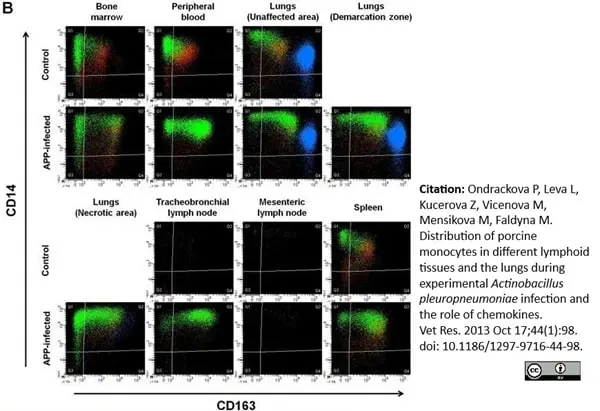
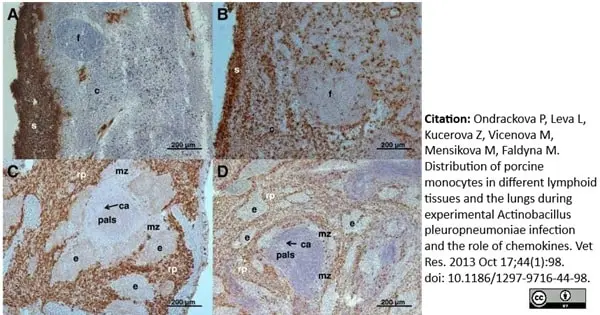
Ondrackova, P. et al. (2010) Porcine mononuclear phagocyte subpopulations in the lung, blood and bone marrow: dynamics during inflammation induced by Actinobacillus pleuropneumoniae.
Vet Res. 41: 64.
View The Latest Product References
-
Das, P.B. et al. (2010) The minor envelope glycoproteins GP2a and GP4 of porcine reproductive and respiratory syndrome virus interact with the receptor CD163.
J Virol. 84: 1731-40. -
Urbieta Caceres, V.H. et al. (2011) Early experimental hypertension preserves the myocardial microvasculature but aggravates cardiac injury distal to chronic coronary artery obstruction.
Am J Physiol Heart Circ Physiol. 300: H693-701. -
Gimeno, M. et al. (2011) Cytokine profiles and phenotype regulation of antigen presenting cells by genotype-I porcine reproductive and respiratory syndrome virus isolates.
Vet Res. 42: 9. -
Kapetanovic, R. et al. (2012) Pig bone marrow-derived macrophages resemble human macrophages in their response to bacterial lipopolysaccharide.
J Immunol. 188: 3382-94. -
De Baere, M.I. et al. (2012) Interaction of the European genotype porcine reproductive and respiratory syndrome virus (PRRSV) with sialoadhesin (CD169/Siglec-1) inhibits alveolar macrophage phagocytosis.
Vet Res. 43: 47. -
Prather, R.S. et al. (2013) An Intact Sialoadhesin (Sn/SIGLEC1/CD169) Is Not Required for Attachment/Internalization of the Porcine Reproductive and Respiratory Syndrome Virus.
J Virol. 87: 9538-46. -
Costa-Hurtado, M. et al. (2013) Changes in macrophage phenotype after infection of pigs with Haemophilus parasuis strains with different levels of virulence.
Infect Immun. 81 (7): 2327-33. -
Kyrova K et al. (2014) The response of porcine monocyte derived macrophages and dendritic cells to Salmonella typhimurium and lipopolysaccharide.
BMC Vet Res. 10: 244. -
Stenfeldt, C. et al. (2014) Morphologic and phenotypic characteristics of myocarditis in two pigs infected by foot-and mouth disease virus strains of serotypes O or A.
Acta Vet Scand. 56: 42. -
Sang, Y. et al. (2014) Antiviral Regulation in Porcine Monocytic Cells at Different Activation States.
J Virol. pii: JVI.01714-14. -
Haslauer, C.M. et al. (2014) Gene expression of catabolic inflammatory cytokines peak before anabolic inflammatory cytokines after ACL injury in a preclinical model.
J Inflamm (Lond). 11 (1): 34. -
Li, H. et al. (2015) Function of CD163 fragments in porcine reproductive and respiratory syndrome virus infection.
Int J Clin Exp Med. 8 (9): 15373-82. -
Le Luduec, J.B. et al. (2016) Intradermal vaccination with un-adjuvanted sub-unit vaccines triggers skin innate immunity and confers protective respiratory immunity in domestic swine.
Vaccine. 34 (7): 914-22. -
Deloizy, C. et al. (2016) Expanding the tools for identifying mononuclear phagocyte subsets in swine: Reagents to porcine CD11c and XCR1.
Dev Comp Immunol. 65: 31-40. -
Gu, M.J. et al. (2016) Barrier protection via Toll-like receptor 2 signaling in porcine intestinal epithelial cells damaged by deoxynivalnol.
Vet Res. 47: 25. -
Ma, H. et al. (2017) The Crystal Structure of the Fifth Scavenger Receptor Cysteine-Rich Domain of Porcine CD163 Reveals an Important Residue Involved in Porcine Reproductive and Respiratory Syndrome Virus Infection.
J Virol. ;91(3):e01897-16. -
Zhang, L. et al. (2016) Developing a Triple Transgenic Cell Line for High-Efficiency Porcine Reproductive and Respiratory Syndrome Virus Infection.
PLoS One. 11 (5): e0154238. -
Westover, A.J. et al. (2016) An Immunomodulatory Device Improves Insulin Resistance in Obese Porcine Model of Metabolic Syndrome.
J Diabetes Res. 2016: 3486727. -
Contreras, G.A. et al. (2016) Adipose tissue remodeling in late-lactation dairy cows during feed-restriction-induced negative energy balance.
J Dairy Sci. 99 (12): 10009-21. -
Singleton, H. et al. (2016) Establishing Porcine Monocyte-Derived Macrophage and Dendritic Cell Systems for Studying the Interaction with PRRSV-1.
Front Microbiol. 7: 832. -
Bacou, E. et al. (2017) β2-adrenoreceptor stimulation dampens the LPS-induced M1 polarization in pig macrophages.
Dev Comp Immunol. 76: 169-76. -
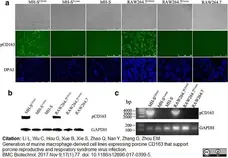
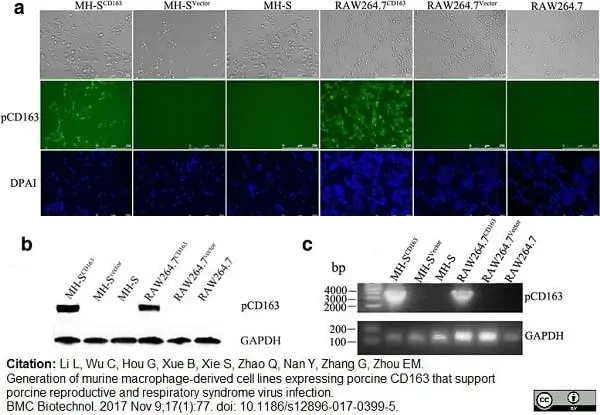
Li, L. et al. (2017) Generation of murine macrophage-derived cell lines expressing porcine CD163 that support porcine reproductive and respiratory syndrome virus infection.
BMC Biotechnol. 17 (1): 77. -
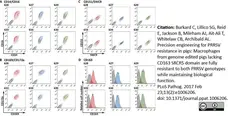
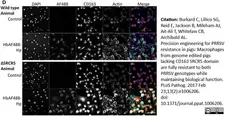
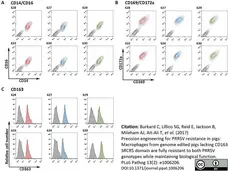
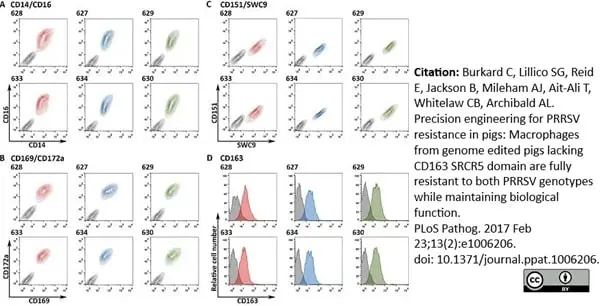
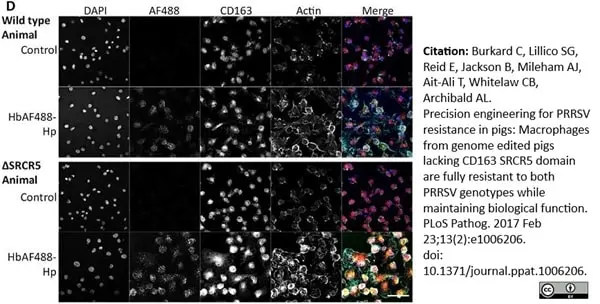
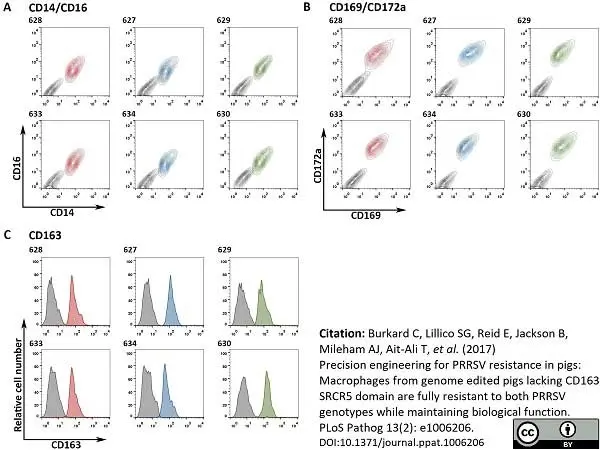
Burkard, C. et al. (2017) Precision engineering for PRRSV resistance in pigs: Macrophages from genome edited pigs lacking CD163 SRCR5 domain are fully resistant to both PRRSV genotypes while maintaining biological function.
PLoS Pathog. 13 (2): e1006206. -
Popescu, L. et al. (2017) Genetically edited pigs lacking CD163 show no resistance following infection with the African swine fever virus isolate, Georgia 2007/1.
Virology. 501: 102-6. -
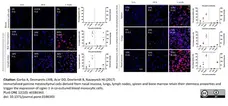
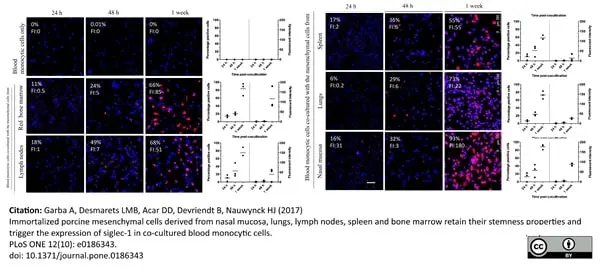
Garba, A. et al. (2017) Immortalized porcine mesenchymal cells derived from nasal mucosa, lungs, lymph nodes, spleen and bone marrow retain their stemness properties and trigger the expression of siglec-1 in co-cultured blood monocytic cells
PLOS ONE. 12 (10): e0186343. -
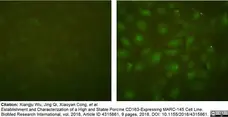
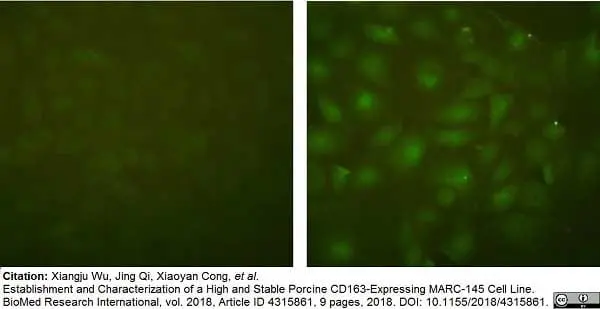
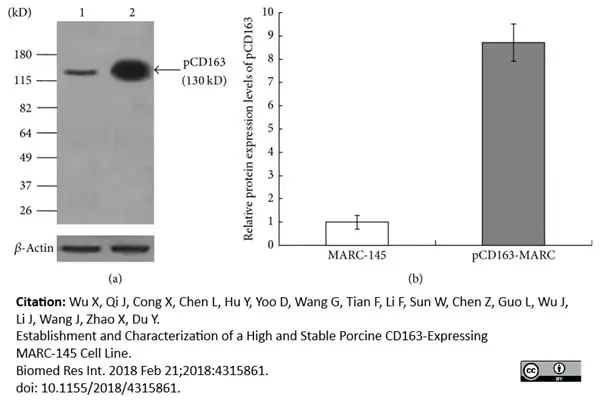
Wu, X. et al. (2018) Establishment and Characterization of a High and Stable Porcine CD163-Expressing MARC-145 Cell Line.
Biomed Res Int. 2018: 4315861. -
Sautter, C.A. et al. (2018) Phenotypic and functional modulations of porcine macrophages by interferons and interleukin-4.
Dev Comp Immunol. 84: 181-92. -
Waddell, L.A. et al. (2018) ADGRE1 (EMR1, F4/80) Is a Rapidly-Evolving Gene Expressed in Mammalian Monocyte-Macrophages.
Front Immunol. 9: 2246. -
Pasternak, J.A. et al. (2019) Development and application of a porcine specific ELISA for the quantification of soluble CD163.
Vet Immunol Immunopathol. 210: 60-7. -
Li, P. et al. (2020) Susceptibility of porcine pulmonary microvascular endothelial cells to porcine reproductive and respiratory syndrome virus.
J Vet Med Sci. 82 (9): 1404-9. -
Ma, H. et al. (2021) Structural comparison of CD163 SRCR5 from different species sheds some light on its involvement in porcine reproductive and respiratory syndrome virus-2 infection in vitro.
Vet Res. 52 (1): 97. -
Jarosova, R. et al. (2022) Cytokine expression by CD163+ monocytes in healthy and Actinobacillus pleuropneumoniae-infected pigs.
Res Vet Sci. 152: 1-9. -
Franzoni, G. et al. (2022) Analyses of the Impact of Immunosuppressive Cytokines on Porcine Macrophage Responses and Susceptibility to Infection to African Swine Fever Viruses.
Pathogens. 11 (2): 166. -
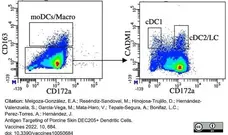
Melgoza-González, A.E. et al. (2022) Antigen Targeting of Porcine Skin DEC205+ Dendritic Cells
Vaccines. 10 (5): 684. -
Zhou, L. et al. (2022) Clinical improvement of sepsis by extracorporeal centrifugal leukocyte apheresis in a porcine model.
J Transl Med. 20 (1): 538. -
Štěpánová, H. et al. (2022) Characterization of Porcine Monocyte-Derived Macrophages Cultured in Serum-Reduced Medium.
Biology (Basel). 11(10):1457. -
Skirecki, T. et al. (2022) Compartment-Specific Differences in the Activation of Monocyte Subpopulations Are Not Affected by Nitric Oxide and Glucocorticoid Treatment in a Model of Resuscitated Porcine Endotoxemic Shock.
J Clin Med. 11 (9): 2641. -
Álvarez, B. et al. (2023) Porcine Macrophage Markers and Populations: An Update.
Cells. 12 (16): 2103. -
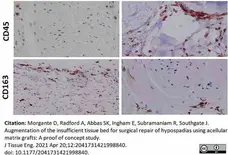
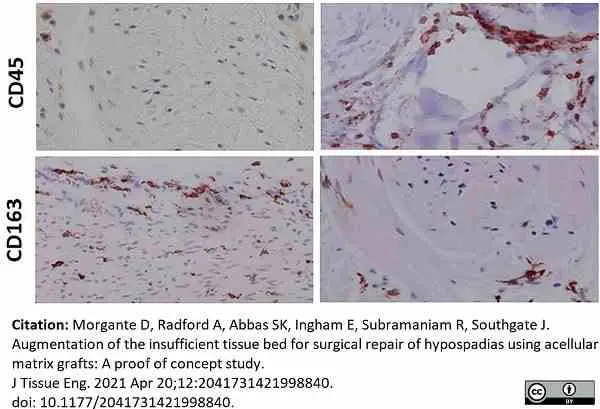
Morgante, D. et al. (2021) Augmentation of the insufficient tissue bed for surgical repair of hypospadias using acellular matrix grafts: A proof of concept study.
J Tissue Eng. 12: 2041731421998840. -
Kim, H.K.W. et al. (2021) Minimally Invasive Necrotic Bone Washing Improves Bone Healing After Femoral Head Ischemic Osteonecrosis: An Experimental Investigation in Immature Pigs.
J Bone Joint Surg Am. 103 (13): 1193-202. -
Chen, Y. et al. (2025) ASFV activates STAT3 to induce proviral M2 macrophage polarization.
Vet Microbiol. 311: 110733. -
Jaudas, F. et al. (2025) Perinatal dysfunction of innate immunity in cystic fibrosis.
Sci Transl Med. 17 (782): eadk9145. -
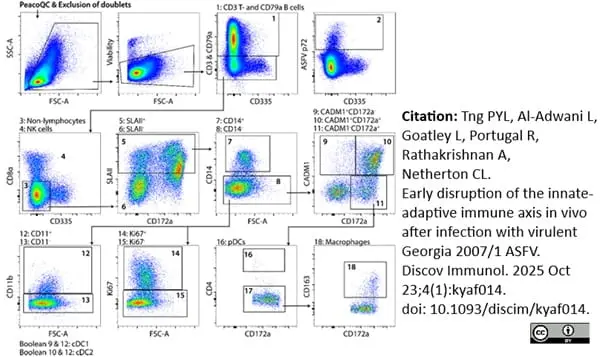
Tng, P.Y.L. et al. (2025) Early disruption of the innate-adaptive immune axis in vivo after infection with virulent Georgia 2007/1 ASFV.
Discov Immunol. 4 (1): kyaf014.
Further Reading
-
Piriou-Guzylack, L. (2008) Membrane markers of the immune cells in swine: an update.
Vet Res. 39: 54.
- UniProt
- Q2VL90
- Entrez Gene
- CD163
- GO Terms
- GO:0005886 plasma membrane
- GO:0006953 acute-phase response
- GO:0016021 integral to membrane
- GO:0005576 extracellular region
- GO:0005044 scavenger receptor activity
MCA2311GA
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Pig ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up