Macrophages/Monocytes/Granulocytes antibody | MAC387
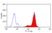




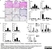

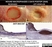


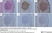









Mouse anti Human Macrophages
- Product Type
- Monoclonal Antibody
- Clone
- MAC387
- Isotype
- IgG1
- Specificity
- Macrophages/Monocytes/Granulocytes
| Mouse anti Human macrophages, clone MAC387 recognizes the L1 or Calprotectin molecule, an intracytoplasmic antigen comprised of a 12 kDa alpha chain and a 14 kDa beta chain. Although originally described as binding to epitopes common to both the alpha and beta chains (Flavell et al. 1987) subsequent evidence indicates that the antibody detects an epitope exclusively expressed on the beta chain (Goebeler et al. 1994) demonstrated by immunofluorescent and western blotting on both naturally expressing and transfected targets. In addition, Mouse anti Human macrophages, clone MAC387 detects the beta chain in complex with the alpha. The antigen recognized by Mouse anti Human macrophages, clone MAC387 is expressed by granulocytes, monocytes and by tissue macrophages. Variable results have been reported for staining brain macrophages and microglia. The epitope recognized appears to be well conserved and the antibody is routinely used for the detection of myeloid cells in a wide range of species. |
- Target Species
- Human
- Species Cross-Reactivity
-
Target Species Cross Reactivity Horse Pig Dog Rabbit Baboon Bovine Guinea Pig Rat Cat Cynomolgus monkey Rhesus Monkey Goat Fallow deer Pygmy hippopotamus Mink Marmoset - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG - liquid
- Preparation
- MCA874GT, MCA874G: Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- MCA874GA: Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant.
- Buffer Solution
- MCA874GT, MCA874G: Phosphate buffered saline
- MCA874GA: Phosphate buffered saline.
- Preservative Stabilisers
0.09% Sodium Azide - Carrier Free
- Yes
- Immunogen
- Human monocytes.
- Approx. Protein Concentrations
- IgG concentration 1.0 mg/ml
- Fusion Partners
- Spleen cells from immunized BALB/c mice were fused with cells of the mouse NS1 myeloma cell line.
- Regulatory
- For research purposes only
- Guarantee
- MCA874GT, MCA874G: 12 months from date of despatch
- MCA874GA: 12 months from date of despatch.
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry 1 | 1/50 | 1/100 | |
| Immunohistology - Frozen | 1/100 | 1/200 | |
| Immunohistology - Paraffin 2 | 1/100 | 1/200 |
- 1 Membrane permeabilization is required for this application. The use of Leucoperm (Product Code BUF09) is recommended for this purpose.
- 2This product requires protein digestion pre-treatment of paraffin sections e.g. trypsin or pronase.
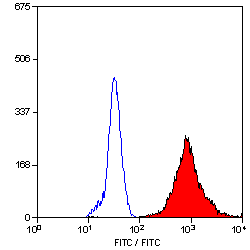
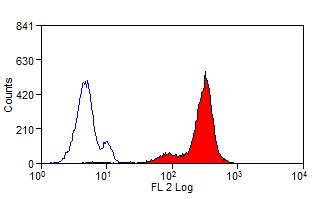
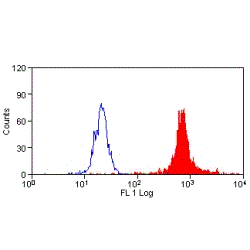
- Flow Cytometry
- Use 10ul of the suggested working dilution to label 1x106 cells in 100ul.
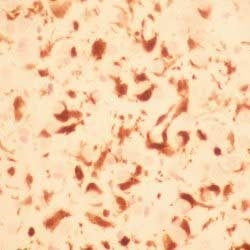
- Histology Positive Control Tissue
- Human Spleen
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG1 Negative Control | MCA928 | F | 100 Tests |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG1 Negative Control | ||||||
Source Reference
-
Flavell, D.J. et al. (1987) Identification of tissue histiocytes on paraffin sections by a new monoclonal antibody.
J Histochem Cytochem. 35 (11): 1217-26.
Antibody Characterization Reference
-
Brandtzaeg, P. et al. (1988) Mac 387 antibody and detection of formalin resistant myelomonocytic L1 antigen.
J Clin Pathol. 41 (9): 963-70.
References for Macrophages/Monocytes/Granulocytes antibody
-
Burudi, E.M. et al. (2002) Regulation of indoleamine 2,3-dioxygenase expression in simian immunodeficiency virus-infected monkey brains.
J Virol. 76: 12233-41. -
Ueland, T. et al. (2009) Dickkopf-1 enhances inflammatory interaction between platelets and endothelial cells and shows increased expression in atherosclerosis.
Arterioscler Thromb Vasc Biol. 29: 1228-34 -
Brandtzaeg, P. et al. (1992) The leucocyte protein L1 (calprotectin): usefulness as an immunohistochemical marker antigen and putative biological function.
Histopathology. 21: 191-6. -
Gutierrez, M. et al. (1999) The detection of CD2+, CD4+, CD8+, and WC1+ T lymphocytes, B cells and macrophages in fixed and paraffin embedded bovine tissue using a range of antigen recovery and signal amplification techniques.
Vet Immunol Immunopathol. 71 (3-4): 321-34. -
Ramsay, A.D. et al. (1991) Phenotypic analysis of malignant lymphoma in simian immunodeficiency virus infection using anti-human antibodies.
J Pathol. 164 (4): 321-8. -
Christgau, M. et al. (1998) Characterization of immunocompetent cells in the diseased canine periodontium.
J Histochem Cytochem. 46 (12): 1443-54. -
Pérez, J. et al. (1999) Immunohistochemical study of the inflammatory infiltrate associated with equine squamous cell carcinoma.
J Comp Pathol. 121 (4): 385-97. -
Nanney, L.B. et al. (2008) Calreticulin enhances porcine wound repair by diverse biological effects.
Am J Pathol. 173: 610-30.
View The Latest Product References
-
Poncelet, L. et al. (2008) Detection of antigenic heterogeneity in feline coronavirus nucleocapsid in feline pyogranulomatous meningoencephalitis.
Vet Pathol. 45: 140-53. -
Sethi, R.S. et al. (2010) Immunolocalization of pulmonary intravascular macrophages, TLR4, TLR9 and IL-8 in normal and Pasteurella multocida-infected lungs of water buffalo (Bubalus bubalis).
J Comp Pathol. 144: 135-44. -
Sanchez, J. et al. (2011) Microscopical and immunological features of tuberculoid granulomata and cavitary pulmonary tuberculosis in naturally infected goats.
J Comp Pathol. 145 (2-3): 107-17. -
Isling, L.K. et al. (2010) Pyelonephritis in slaughter pigs and sows: morphological characterization and aspects of pathogenesis and aetiology.
Acta Vet Scand. 52: 48. -
Vranckx, K. et al. (2012) Vaccination reduces macrophage infiltration in bronchus-associated lymphoid tissue in pigs infected with a highly virulent Mycoplasma hyopneumoniae strain.
BMC Vet Res. 8: 24. -
Campuzano, O. et al. (2012) Arrhythmogenic right ventricular cardiomyopathy: severe structural alterations are associated with inflammation.
J Clin Pathol. 65 (12): 1077-83. -
García-Jiménez, W.L. (2012) Histological and immunohistochemical characterisation of Mycobacterium bovis induced granulomas in naturally infected fallow deer (Dama dama).
Vet Immunol Immunopathol. 149: 66-75. -
Santana, C.H. et al. (2016) Relationship between the inflammatory infiltrate and the degree of differentiation of the canine cutaneous squamous cell carcinoma.
Vet Anim Sci. 1-2: 4-8. -
Masure, D. et al. (2013) A Role for Eosinophils in the Intestinal Immunity against Infective Ascaris suum Larvae.
PLoS Negl Trop Dis. 2013 Mar;7(3): e2138. -
Tellez, A. et al. (2014) Experimental evaluation of efficacy and healing response of everolimus-eluting stents in the familial hypercholesterolemic swine model: a comparative study of bioabsorbable versus durable polymer stent platforms.
Coron Artery Dis. 25 (3): 198-207. -
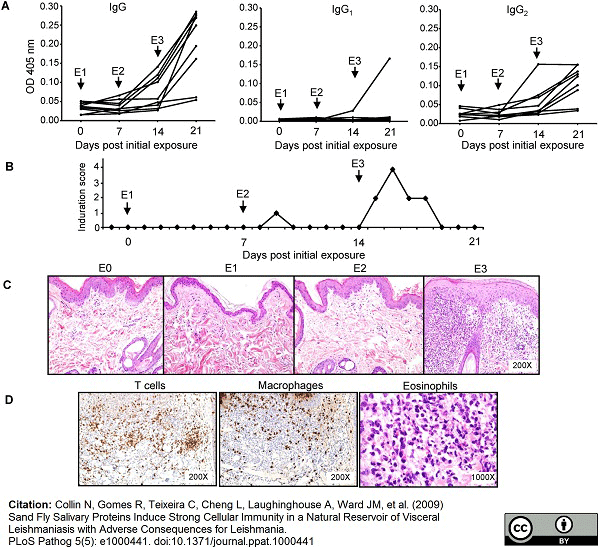
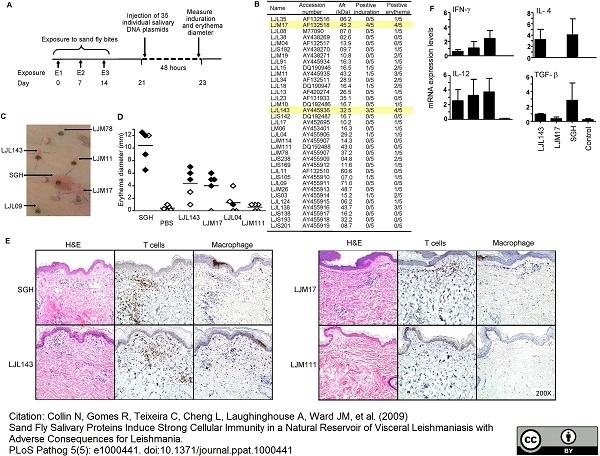
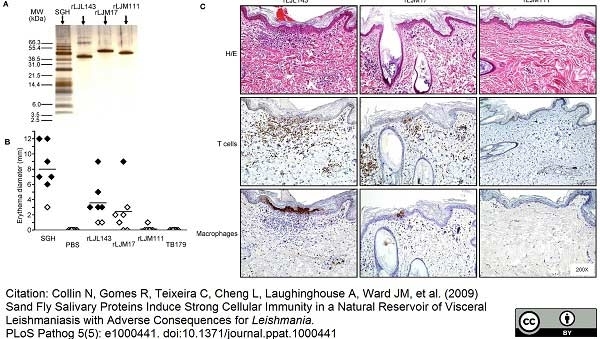
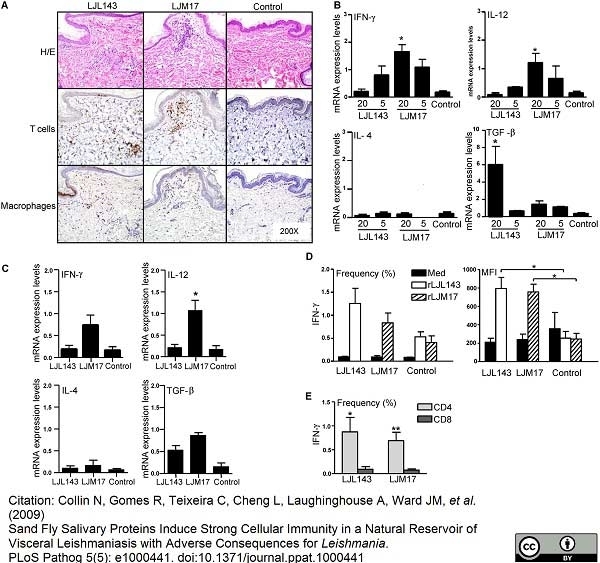
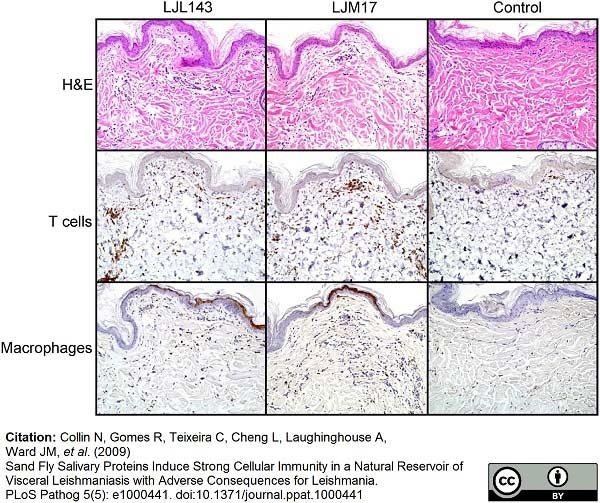
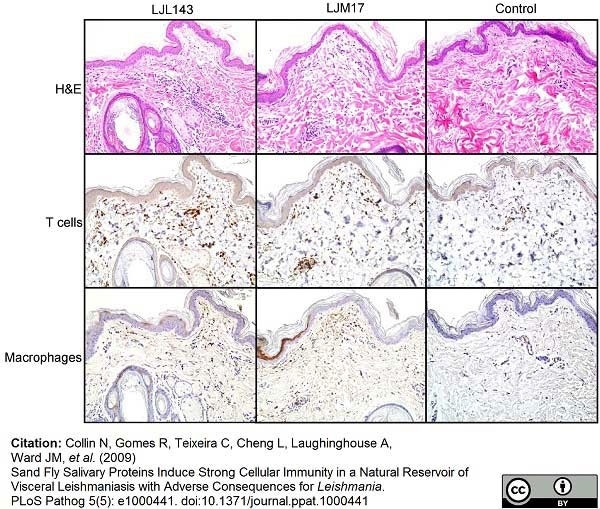
Collin, N. et al. (2009) Sand fly salivary proteins induce strong cellular immunity in a natural reservoir of visceral leishmaniasis with adverse consequences for Leishmania.
PLoS Pathog. 5(5):e1000441. -
McCurdy, P. et al. (2014) Acute lymphoblastic leukemia in a pygmy hippopotamus (Hexaprotodon liberiensis).
J Zoo Wildl Med. 45 (4): 906-10. -
Marcaccini, A. et al. (2008) Pseudorabies virus infection in mink: a host-specific pathogenesis.
Vet Immunol Immunopathol. 124 (3-4): 264-73. -
Romero-Palomo, F. et al. (2017) Immunopathologic Changes in the Thymus of Calves Pre-infected with BVDV and Challenged with BHV-1.
Transbound Emerg Dis. 64 (2): 574-84. -
Rossi, C.N. et al. (2016) In situ Cutaneous cellular immune response in dogs naturally infected by visceral leishmaniasis.
Rev Inst Med Trop Sao Paulo. 58: . -
Vrolyk, V. et al. (2017) Lung Inflammation Associated With Clinical Acute Necrotizing Pancreatitis in Dogs.
Vet Pathol. 54 (1): 129-40. -
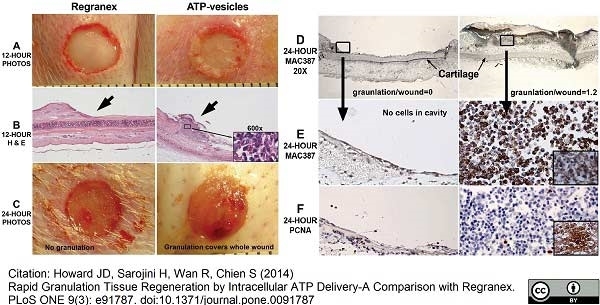
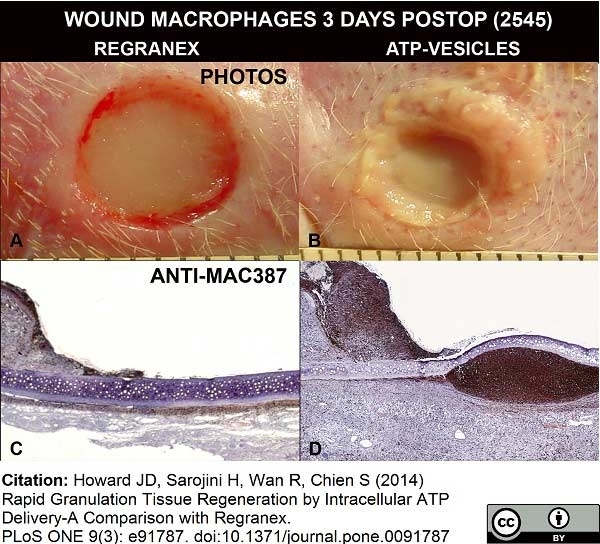
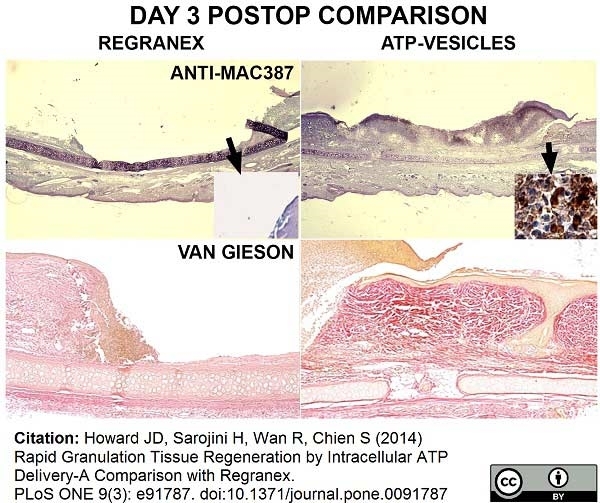
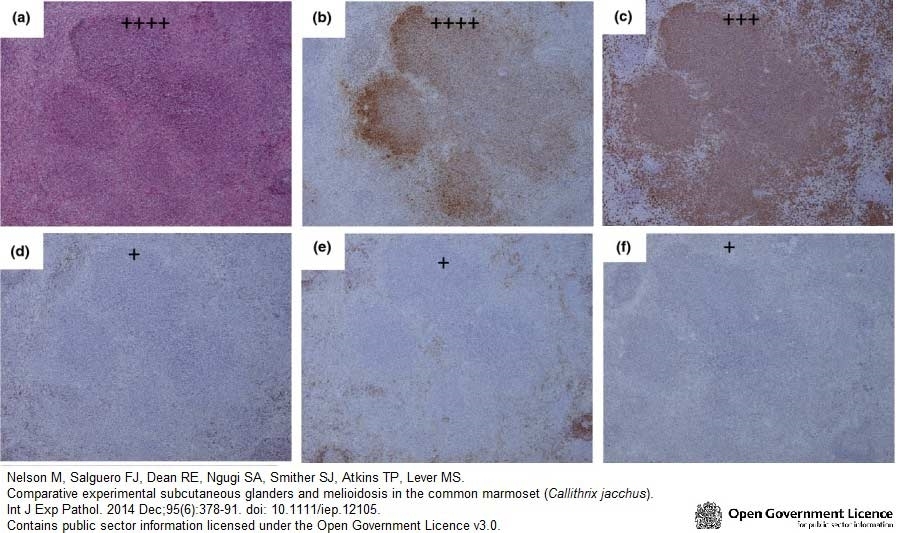
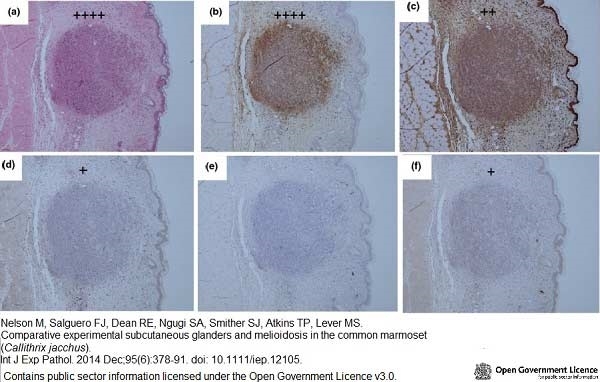
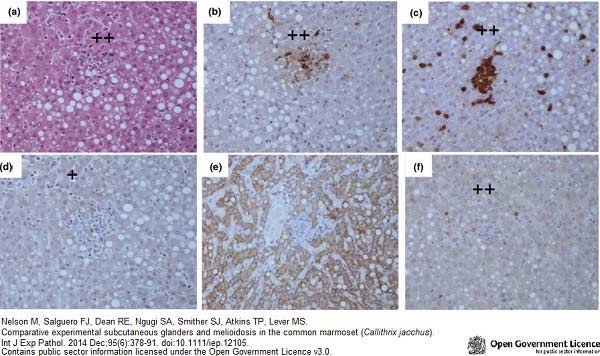
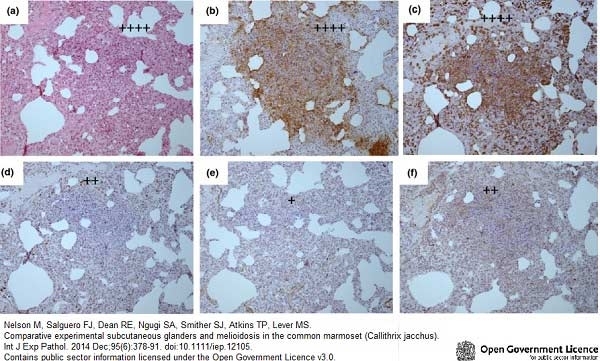
Nelson, M. et al. (2014) Comparative experimental subcutaneous glanders and melioidosis in the common marmoset (Callithrix jacchus).
Int J Exp Pathol. 95 (6): 378-91. -
Amarilla, S.P. et al. (2016) Thymic depletion of lymphocytes is associated with the virulence of PRRSV-1 strains.
Vet Microbiol. 188: 47-58. -
García-Jiménez, W.L. et al. (2013) Immunopathology of granulomas produced by Mycobacterium bovis in naturally infected wild boar.
Vet Immunol Immunopathol. 156 (1-2): 54-63. -
Zhao, L. et al. (2020) Reducing macrophage numbers alleviates temporomandibular joint ankylosis.
Cell Tissue Res. 379 (3): 521-36. -
Lai, H.Y. et al. (2017) CCAAT/enhancer-binding protein delta promotes intracellular lipid accumulation in M1 macrophages of vascular lesions.
Cardiovasc Res. 113 (11): 1376-88. -
Wacinski, P. et al. (2021) Anti-Inflammatory Effect of Very High Dose Local Vessel Wall Statin Administration: Poly(L,L-Lactide) Biodegradable Microspheres with Simvastatin for Drug Delivery System (DDS).
Int J Mol Sci. 22 (14): 7486. -
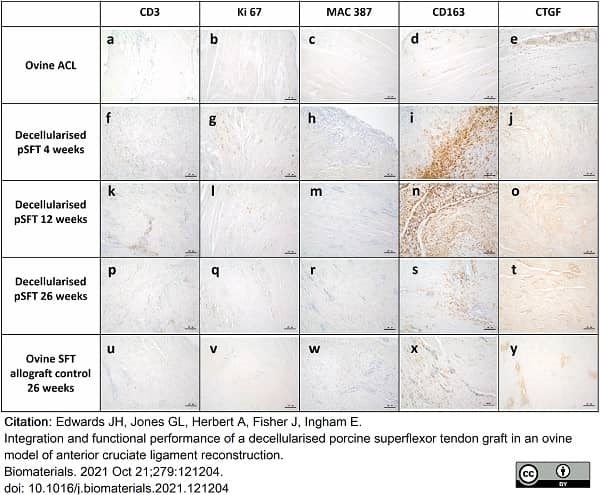
Edwards, J.H. et al. (2021) Integration and functional performance of a decellularised porcine superflexor tendon graft in an ovine model of anterior cruciate ligament reconstruction.
Biomaterials. 279: 121204. -
Bertolo, P.H.L. et al. (2022) Influence of serum progesterone levels on the inflammatory response of female dogs with visceral leishmaniosis.
Vet Parasitol. 302: 109658. -
do Prado Duzanski, A. et al. (2022) Cell-mediated immunity and expression of MHC class I and class II molecules in dogs naturally infected by canine transmissible venereal tumor: Is there complete spontaneous regression outside the experimental CTVT?
Research in Veterinary Science. 145: 193-204. -
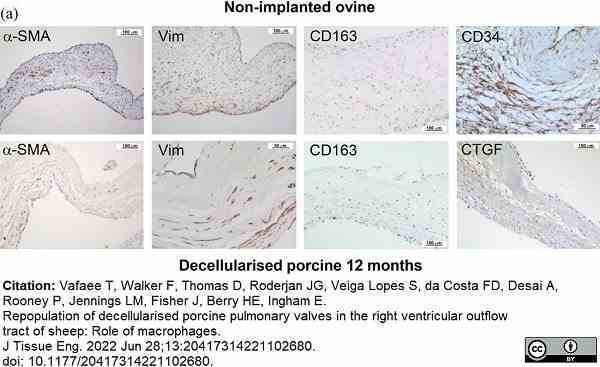
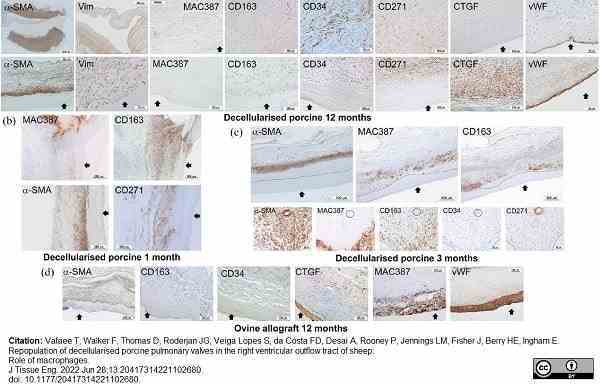
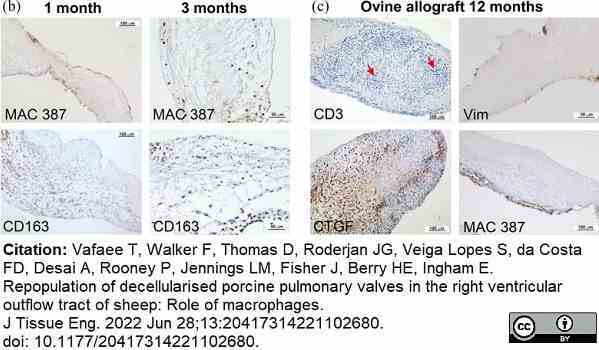
Vafaee, T. et al. (2022) Repopulation of decellularised porcine pulmonary valves in the right ventricular outflow tract of sheep: Role of macrophages.
J Tissue Eng. 13: 20417314221102680. -
Roux, H.M. et al. (2023) DNA ultra-sensitive quantification, a technology for studying HIV unintegrated linear DNA.
Cell Rep Methods. 3 (4): 100443. -
Agerholm, J.S. et al. (2023) Actinobacillus lignieresii‐associated myocellulitis of the nasal planum in a Jersey cow
Veterinary Record Case Reports. 11 (4) [Epub ahead of print]. -
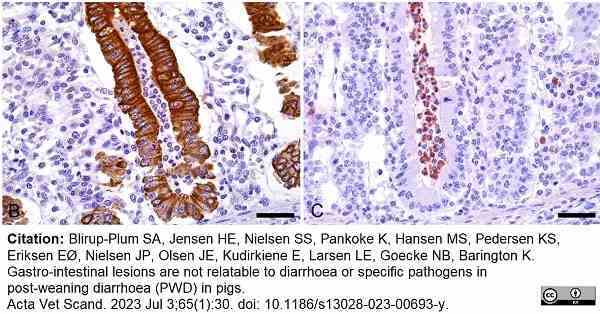
Blirup-Plum, S.A. et al. (2023) Gastro-intestinal lesions are not relatable to diarrhoea or specific pathogens in post-weaning diarrhoea (PWD) in pigs.
Acta Vet Scand. 65 (1): 30. -
Anderson, S.L. et al. (2021) Depletion of pulmonary intravascular macrophages rescues inflammation-induced delayed neutrophil apoptosis in horses.
Am J Physiol Lung Cell Mol Physiol. 320 (1): L126-L136.
Further Reading
-
Burk, J. et al. (2013) Equine cellular therapy--from stall to bench to bedside?
Cytometry A. 83 (1): 103-13. -
Piriou-Guzylack, L. (2008) Membrane markers of the immune cells in swine: an update.
Vet Res. 39: 54.
- Synonyms
- Calprotectin
- RRID
- AB_321963
- UniProt
- P06702
- Entrez Gene
- S100A9
- GO Terms
- GO:0005886 plasma membrane
- GO:0005515 protein binding
- GO:0005509 calcium ion binding
- GO:0005576 extracellular region
- GO:0004871 signal transducer activity
- GO:0005634 nucleus
- GO:0005737 cytoplasm
- GO:0005856 cytoskeleton
- GO:0006935 chemotaxis
- View More GO Terms
- GO:0006954 inflammatory response
- GO:0007267 cell-cell signaling
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Human ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up