IFN Gamma antibody | CC330





Mouse anti Bovine Interferon Gamma
- Product Type
- Monoclonal Antibody
- Clone
- CC330
- Isotype
- IgG1
- Specificity
- IFN Gamma
| Mouse anti Bovine interferon γ antibody, clone CC330 recognizes bovine interferon gamma, also known as immune interferon. Interferon γ is a 143 amino acid ~20 kDa immunomodulatory cytokine secreted predominantly by T lymphocytes in response to infection by invading microorganisms. Interferon γ enhances cytotoxic activities of many cell types including macrophages, NK cells and T lymphocytes (Weiss et al. 2002). |
- Target Species
- Bovine
- Species Cross-Reactivity
-
Target Species Cross Reactivity Sheep Goat - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% sodium azide (NaN3)
- Carrier Free
- Yes
- Immunogen
- Recombinant bovine IFN-gamma.
- Approx. Protein Concentrations
- IgG concentration 1.0 mg/ml
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| ELISA | 5μg/ml as capture antibody | ||
| ELISpot | |||
| Flow Cytometry 1 | 1/50 | 1/100 |
- 1 Membrane permeabilisation is required for this application. Bio-Rad recommends the use of Leucoperm (Product Code BUF09) for this purpose.
- Flow Cytometry
- Use 10μl of the suggested working dilution to label 106 cells or 100μl whole blood
- ELISA
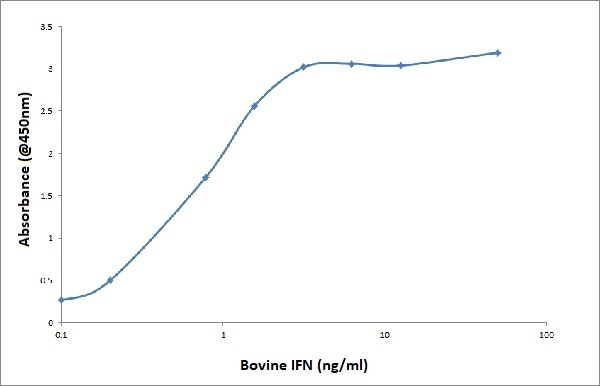
- Mouse anti Bovine interferon-γ antibody, clone CC330 may be used as a capture reagent in a sandwich ELISA in combination with Mouse anti Bovine interferon-γ antibody, clone CC302 (MCA1783B) as a detection reagent and recombinant bovine interferon-γ (PBP007A) as a protein standard.
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG1 Negative Control | MCA928 | F | 100 Tests |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG1 Negative Control | ||||||
References for IFN Gamma antibody
-
Mwangi, W. et al. (2002) DNA-encoded fetal liver tyrosine kinase 3 ligand and granulocyte macrophage-colony-stimulating factor increase dendritic cell recruitment to the inoculation site and enhance antigen-specific CD4+ T cell responses induced by DNA vaccination of outbred animals.
J Immunol. 169 (7): 3837-46. -
Baszler, T.V. et al. (2008) Bovine immune response to inoculation with Neospora caninum surface antigen SRS2 lipopeptides mimics immune response to infection with live parasites.
Clin Vaccine Immunol. 15: 659-67. -
Pascale, F. et al. (2008) Plasmacytoid dendritic cells migrate in afferent skin lymph.
J Immunol. 180: 5963-72. -
Tourais-Esteves, I. et al. (2008) Neonatal goats display a stronger TH1-type cytokine response to TLR ligands than adults
Dev Comp Immunol. 32: 1231-41. -
Begg, D.J. et al. (2009) Enzyme-linked immunospot: an alternative method for the detection of interferon gamma in Johne's disease.
J Vet Diagn Invest. 21: 187-96. -
Panadero, R. et al. (2009) Immunomodulatory effect of Hypoderma lineatum antigens: in vitro effect on bovine lymphocyte proliferation and cytokine production.
Parasite Immunol. 31: 72-7. -
Elhmouzi-Younes, J. et al. (2009) Bovine neonate natural killer cells are fully functional and highly responsive to interleukin-15 and to NKp46 receptor stimulation.
Vet Res. 2009 40: 54. -
Ferret-Bernard, S. et al. (2010) Cellular and molecular mechanisms underlying the strong neonatal IL-12 response of lamb mesenteric lymph node cells to R-848.
PLoS One. 5: e13705.
View The Latest Product References
-
Contreras, V. et al. (2010) Existence of CD8α-like dendritic cells with a conserved functional specialization and a common molecular signature in distant mammalian species.
J Immunol. 185: 3313-25. -
Elhmouzi-Younes, J. et al. (2010) Ovine CD16+/CD14- blood lymphocytes present all the major characteristics of natural killer cells.
Vet Res. 41 (1): 4. -
McLaughlin, K. et al. (2010) Hsp70 enhances presentation of FMDV antigen to bovine CD4+ T cells in vitro.
Vet Res. 41: 36. -
Ferret-Bernard, S. et al. (2011) Mesenteric lymph node cells from neonates present a prominent IL-12 response to CpG oligodeoxynucleotide via an IL-15 feedback loop of amplification.
Vet Res. 42:19. -
Sow, F.B. et al. (2011) Respiratory syncytial virus is associated with an inflammatory response in lungs and architectural remodeling of lung-draining lymph nodes of newborn lambs.
Am J Physiol Lung Cell Mol Physiol. 300 (1): L12-24. -
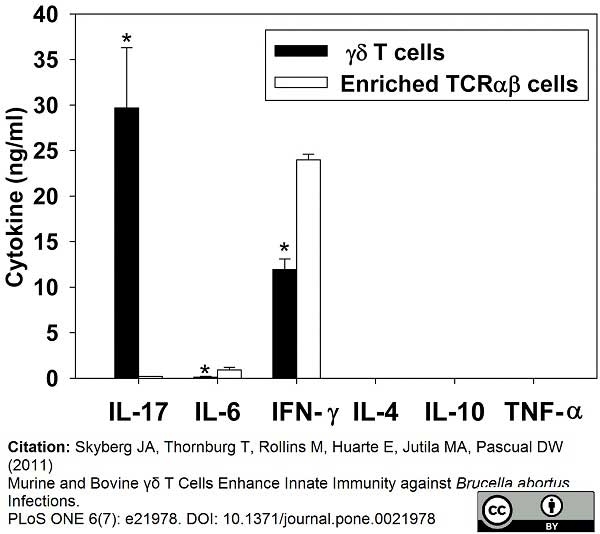
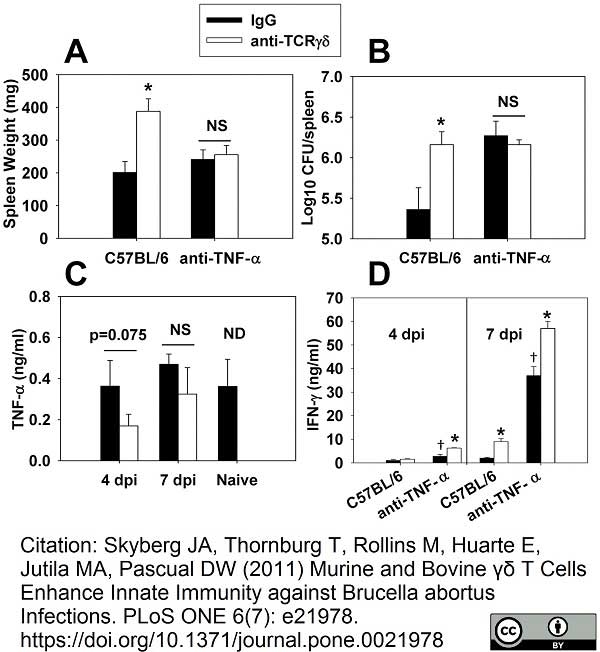
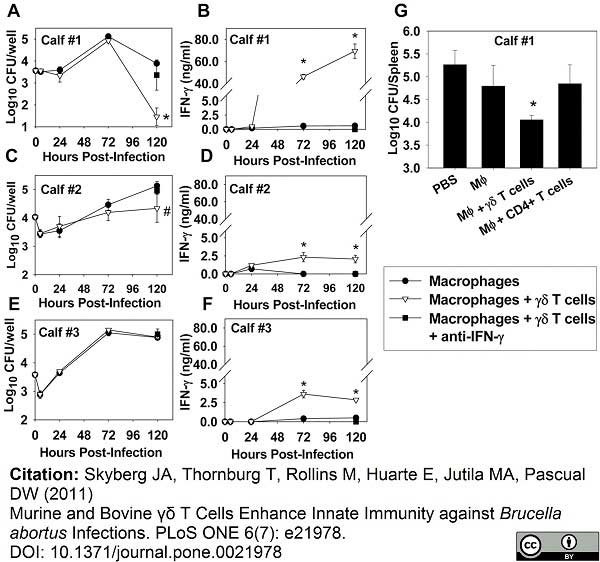
Skyberg, J.A. et al. (2011) Murine and bovine γδ T cells enhance innate immunity against Brucella abortus infections.
PLoS One. 6:e21978. -
Appana, G. et al. (2013) Antemortem and postmortem examinations of the cattle calf naturally infected with Mycobacterium avium subsp. paratuberculosis.
Eur J Microbiol and Immunol. 3: 241–51. -
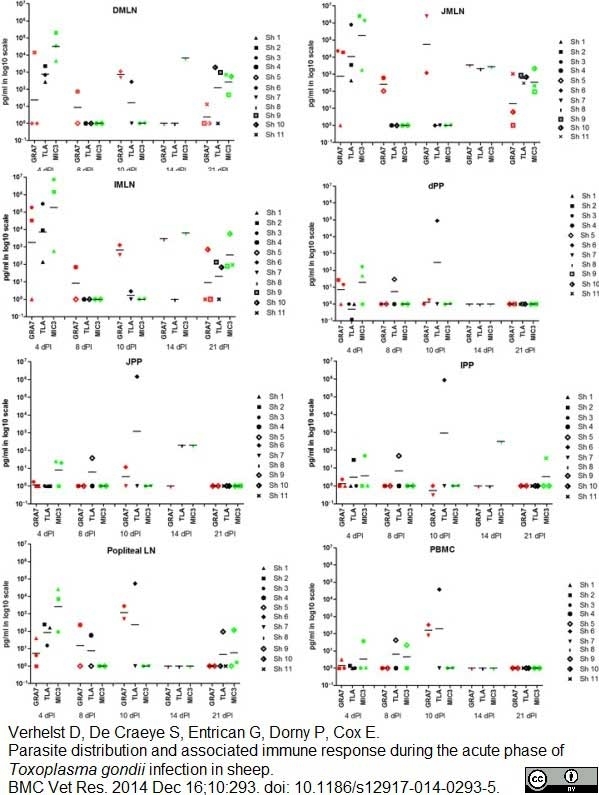
Verhelst, D. et al. (2014) Parasite distribution and associated immune response during the acute phase of Toxoplasma gondii infection in sheep.
BMC Vet Res. 10: 293. -
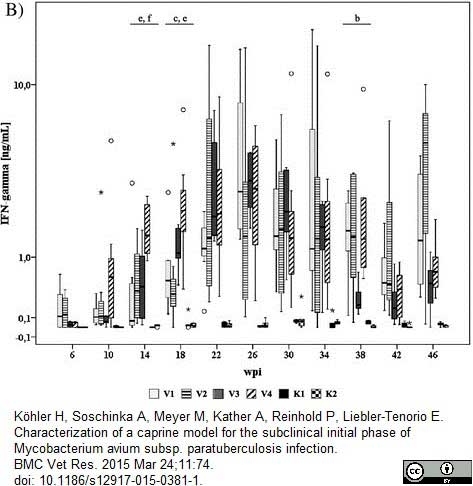
Köhler H et al. (2015) Characterization of a caprine model for the subclinical initial phase of Mycobacterium avium subsp. paratuberculosis infection.
BMC Vet Res. 11 (1): 74. -
Bergmann, A. et al. (2015) In Vivo Volatile Organic Compound Signatures of Mycobacterium avium subsp. paratuberculosis.
PLoS One. 10 (4): e0123980. -
Cassady-Cain, R.L. et al. (2017) Inhibition of Antigen-Specific and Nonspecific Stimulation of Bovine T and B Cells by Lymphostatin from Attaching and Effacing Escherichia coli.
Infect Immun. 85(2):e00845-16. -
Rodrigues, V. et al. (2017) Development of a bead-based multiplexed assay for simultaneous quantification of five bovine cytokines by flow cytometry.
Cytometry A. 91 (9): 901-7. -
Villa-Mancera, A. et al. (2021) Phage display-based vaccine with cathepsin L and excretory-secretory products mimotopes of Fasciola hepatica. induces protective cellular and humoral immune responses in sheep.
Vet Parasitol. 289: 109340. -
Risalde, M.Á. et al. (2017) Development and evaluation of an interferon gamma assay for the diagnosis of tuberculosis in red deer experimentally infected with Mycobacterium bovis..
BMC Vet Res. 13 (1): 341. -
Ciliberti, M.G. et al. (2022) Green extraction of bioactive compounds from wine lees and their bio-responses on immune modulation using in vitro sheep model.
J Dairy Sci. 105 (5): 4335-53. -
Santillo, A. et al. (2022) Feeding tannins to dairy cows in different seasons improves the oxidative status of blood plasma and the antioxidant capacity of cheese.
J Dairy Sci. 105 (11): 8609-20. -
Bouroutzika, E. et al. (2023) Melatonin Administration to Pregnant Ewes for Coccidiosis Control in Their Offspring.
Animals (Basel). 13 (14): 2381. -
Tulu, B. et al. (2020) Cellular and Cytokine Responses in the Granulomas of Asymptomatic Cattle Naturally Infected with Mycobacterium bovis in Ethiopia.
Infect Immun. 88 (12): e00507-20.
- Synonyms
- Immune Interferon
- RRID
- AB_2123456
- UniProt
- P07353
- Entrez Gene
- IFNG
- GO Terms
- GO:0009615 response to virus
- GO:0005125 cytokine activity
- GO:0005133 interferon-gamma receptor binding
- GO:0005615 extracellular space
- GO:0006955 immune response
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Bovine ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up