CD4 antibody | CC30




Mouse anti Bovine CD4
- Product Type
- Monoclonal Antibody
- Clone
- CC30
- Isotype
- IgG1
- Specificity
- CD4
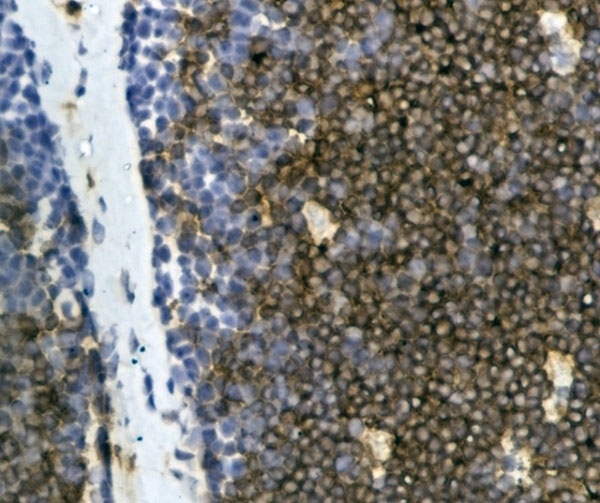
| Mouse anti Bovine CD4 antibody, clone CC30 recognizes a ~50 kDa transmembrane molecule considered to be the bovine homologue of human CD4. The phenotype, tissue distribution and function of T-cells expressing the bovine CD4 antigen are similar to those in other species. However, expression on macrophages has not yet been detected. Mouse anti Bovine CD4, clone CC30 has successfully been used for immunohistochemical localization of CD4 on paraffin embedded material using zinc salt fixation (Cantón et al. 2013). Additionally, clone CC30 has been reported as being suitable for use on formal dichromate (FD5) fixed paraffin embedded tissue with amplification and antigen retrieval techniques (Gutierrez et al. 1999). |
- Target Species
- Bovine
- Species Cross-Reactivity
-
Target Species Cross Reactivity Bison - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% Sodium Azide (NaN3)
- Carrier Free
- Yes
- Immunogen
- Bovine thymocytes.
- Approx. Protein Concentrations
- IgG concentration 1.0 mg/ml
- Fusion Partners
- Spleen cells from an immunized mouse were fused with cells of the mouse NS1 myeloma cell line.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | 1/25 | 1/200 | |
| Immunohistology - Frozen | 1/100 | ||
| Immunohistology - Paraffin 1 | |||
| Immunoprecipitation |
- 1
Clone CC30 has been reported as being suitable for use on formal dichromate (FD5) fixed paraffin embedded tissue with amplification and antigen retrieval techniques, see Gutierrez et al. for details.
- Flow Cytometry
- Use 10ul of the suggested working dilution to label 106 cells in 100ul.
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG1 Negative Control | MCA928 | F | 100 Tests |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG1 Negative Control | ||||||
References for CD4 antibody
-
Bensaid, A. & Hadam, M. (1991) Individual antigens of cattle. Bovine CD4 (BoCD4).
Vet Immunol Immunopathol. 27 (1-3): 51-4. -
Eskra, L. et al. (1991) Effect of monoclonal antibodies on in vitro. function of T-cell subsets.
Vet Immunol Immunopathol. 27 (1-3): 215-22. -
Gutierrez, M. et al. (1999) The detection of CD2+, CD4+, CD8+, and WC1+ T lymphocytes, B cells and macrophages in fixed and paraffin embedded bovine tissue using a range of antigen recovery and signal amplification techniques.
Vet Immunol Immunopathol. 71 (3-4): 321-34. -
Winkler, M.T. et al. (1999) Bovine herpesvirus 1 can infect CD4(+) T lymphocytes and induce programmed cell death during acute infection of cattle.
J Virol. 73 (10): 8657-68. -
Winkler, M.T. et al. (2000) Persistence and reactivation of bovine herpesvirus 1 in the tonsils of latently infected calves.
J Virol. 74 (11): 5337-46. -
Riondato, F. et al. (2008) Effects of road transportation on lymphocyte subsets in calves
Vet J. 175: 364-8. -
Collins, R.A. et al. (1999) Bovine interleukin-12 and modulation of IFNgamma production.
Vet Immunol Immunopathol. 68: 193-207. -
Liebana, E. et al. (2007) Distribution and activation of T-lymphocyte subsets in tuberculous bovine lymph-node granulomas.
Vet Pathol. 44: 366-72.
View The Latest Product References
-
Sühwold, A. et al. (2010) T cell reactions of Eimeria bovis primary and challenge-infected calves.
Parasitol Res. 106: 595-605. -
Rhodes, S.G. et al. (2000) Bovine tuberculosis: immune responses in the peripheral blood and at the site of active disease.
Immunology. 99: 195-202. -
Machugh, N.D. et al. (1997) Identification of two distinct subsets of bovine gamma delta T cells with unique cell surface phenotype and tissue distribution.
Immunology. 92: 340-5. -
Childerstone, A.J. et al. (1999) Demonstration of bovine CD8+ T-cell responses to foot-and-mouth disease virus.
J Gen Virol. 80: 663-9. -
Rhodes, S.G. et al. (2001) Antigen recognition and immunomodulation by gamma delta T cells in bovine tuberculosis.
J Immunol. 166: 5604-10. -
Knowles, G. et al. (1996) Phenotypical characterization of lymphocytes infiltrating regressing papillomas.
J Virol. 70: 8451-8. -
Smyth, A.J. et al. (2001) In vitro responsiveness of gammadelta T cells from Mycobacterium bovis-infected cattle to mycobacterial antigens: predominant involvement of WC1(+) cells.
Infect Immun. 69: 89-96. -
Hein, W.R. and Dudler, L. (1997) TCR gamma delta+ cells are prominent in normal bovine skin and express a diverse repertoire of antigen receptors.
Immunology. 91: 58-64. -
Juleff, N. et al. (2009) Foot-and-mouth disease virus can induce a specific and rapid CD4+ T-cell-independent neutralizing and isotype class-switched antibody response in naïve cattle.
J Virol. 83: 3626-36. -
Hope, J.C. et al. (2005) Exposure to Mycobacterium avium induces low-level protection from Mycobacterium bovis infection but compromises diagnosis of disease in cattle.
Clin Exp Immunol. 141: 432-9. -
Sopp, P. et al. (2006) Flow cytometric detection of gamma interferon can effectively discriminate Mycobacterium bovis BCG-vaccinated cattle from M. bovis-infected cattle.
Clin Vaccine Immunol. 13: 1343-8. -
Sacchini, F. et al. (2011) A minor role of CD4+ T lymphocytes in the control of a primary infection of cattle with Mycoplasma mycoides subsp. mycoides.
Vet Res. 42: 77. -
Skyberg, J.A. et al. (2011) Murine and bovine γδ T cells enhance innate immunity against Brucella abortus infections.
PLoS One. 6(7): e21978. -
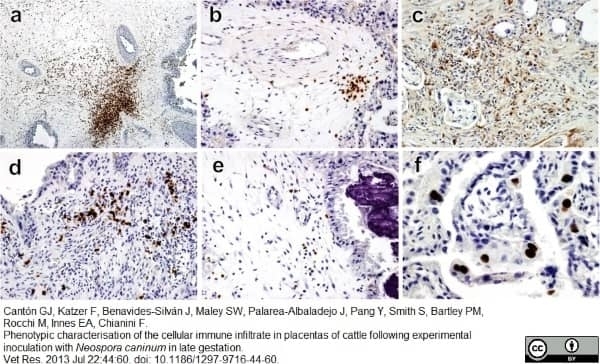
Cantón, G.J. et al. (2013) Phenotypic characterisation of the cellular immune infiltrate in placentas of cattle following experimental inoculation with Neospora caninum in late gestation.
Vet Res. 44: 60. -
Reid, E. et al. (2011) Bovine plasmacytoid dendritic cells are the major source of type I interferon in response to foot-and-mouth disease virus in vitro and in vivo.
J Virol. 85: 4297-308. -
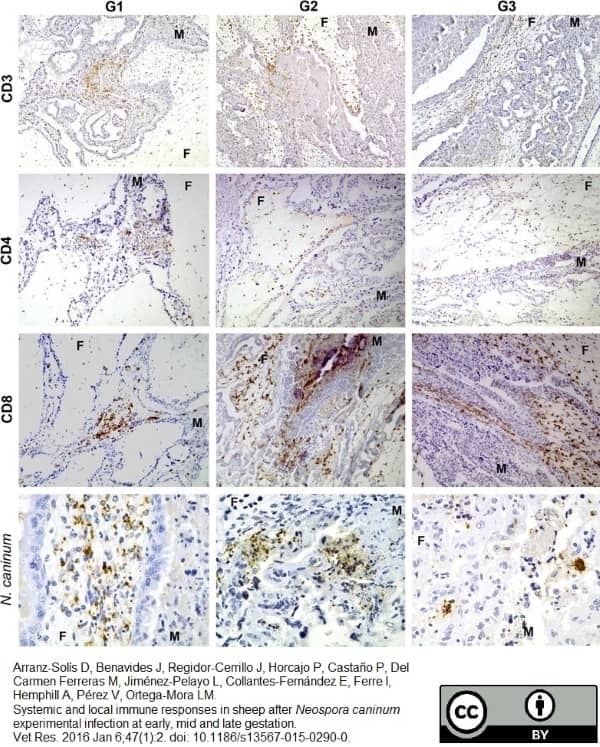
Arranz-Solís D et al. (2016) Systemic and local immune responses in sheep after Neospora caninum experimental infection at early, mid and late gestation.
Vet Res. 47 (1): 2. -
Clapp, B. et al. (2011) DNA vaccination of bison to brucellar antigens elicits elevated antibody and IFN-γ responses.
J Wildl Dis. 47 (3): 501-10. -
Aranday-Cortes, E. et al. (2013) Upregulation of IL-17A, CXCL9 and CXCL10 in early-stage granulomas induced by Mycobacterium bovis in cattle.
Transbound Emerg Dis. 60 (6): 525-37. -
Pirson, C. et al. (2015) Highly purified mycobacterial phosphatidylinositol mannosides drive cell-mediated responses and activate NKT cells in cattle.
Clin Vaccine Immunol. 22 (2): 178-84. -
Fuertes, M. et al. (2015) Immunohistochemical study and mRNA cytokine profile of the local immune response in cattle naturally infected with Calicophoron daubneyi.
Vet Parasitol. 214 (1-2): 178-83. -
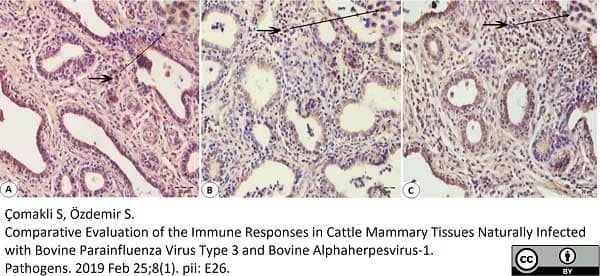
Çomakli, S. & Özdemir, S. (2019) Comparative Evaluation of the Immune Responses in Cattle Mammary Tissues Naturally Infected with Bovine Parainfluenza Virus Type 3 and Bovine Alphaherpesvirus-1.
Pathogens.8 (1): 26. -
Bozkurt, Y.A. et al. (2019) The architecture of the lymph nodes in the abdominal and thoracic cavities of wild boar
Ind J Anim Res. 53 609-15. -
Hecker, Y.P. et al. (2015) Cell mediated immune responses in the placenta following challenge of vaccinated pregnant heifers with Neospora caninum.
Vet Parasitol. 214 (3-4): 247-54. -
Cunha, P. et al. (2019) Expansion, isolation and first characterization of bovine Th17 lymphocytes.
Sci Rep. 9 (1): 16115. -
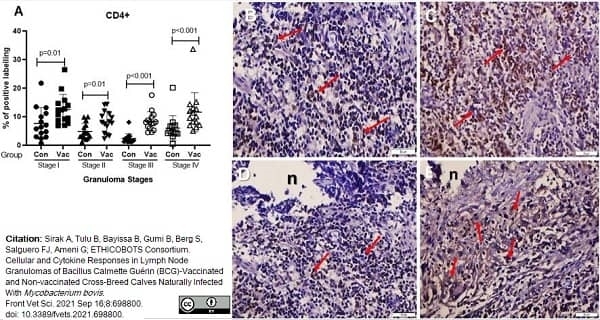
Sirak, A. et al. (2021) Cellular and Cytokine Responses in Lymph Node Granulomas of Bacillus Calmette Guérin (BCG)-Vaccinated and Non-vaccinated Cross-Breed Calves Naturally Infected With Mycobacterium bovis.
Front Vet Sci. 8: 698800. -
Korbonits, L. et al. (2022) Mycobacterium avium subsp. paratuberculosis Infected Cows Reveal Divergent Immune Response in Bovine Peripheral Blood Derived Lymphocyte Proteome.
Metabolites. 12 (10): 924. -
Andrés, S. et al. (2024) Essential oil supplementation in milk replacers: short- and long-term impacts on feed efficiency, the faecal microbiota and the plasma metabolome in dairy calves.
J Dev Orig Health Dis. : 1-11.
- UniProt
- A7YY52
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Bovine ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up