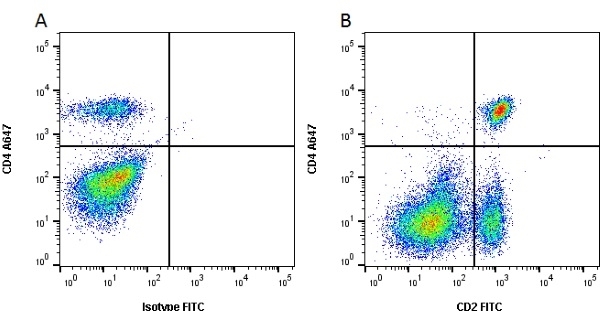
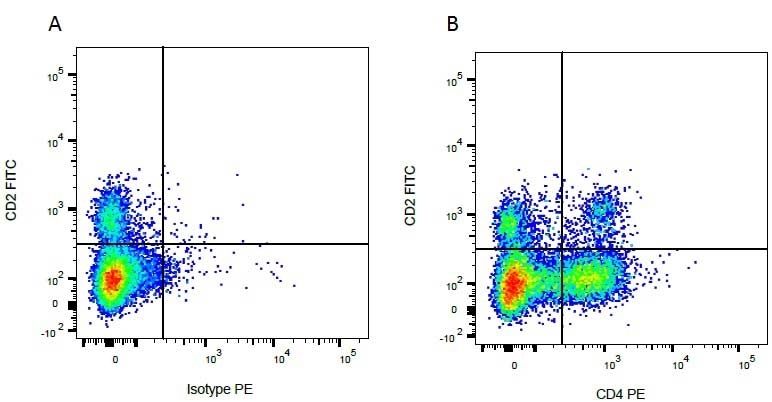
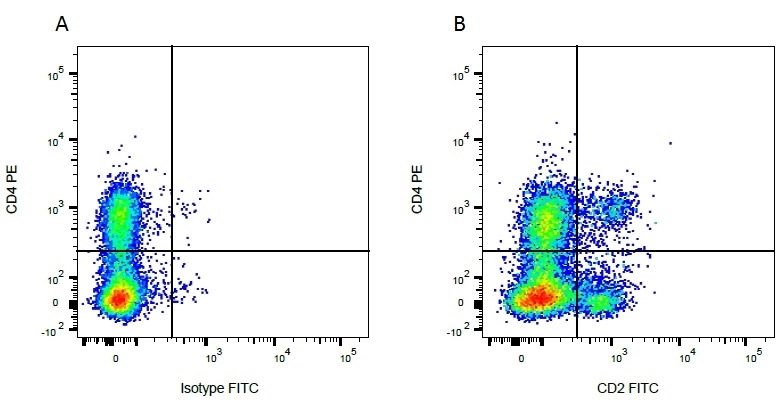
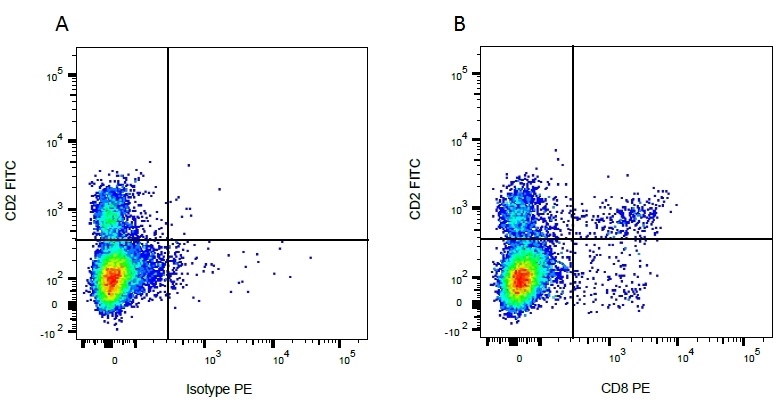
CD2 antibody | CC42









Mouse anti Bovine CD2
- Product Type
- Monoclonal Antibody
- Clone
- CC42
- Isotype
- IgG1
- Specificity
- CD2
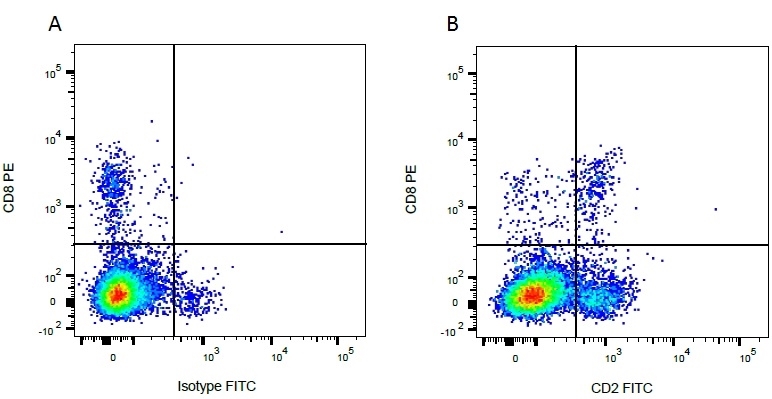
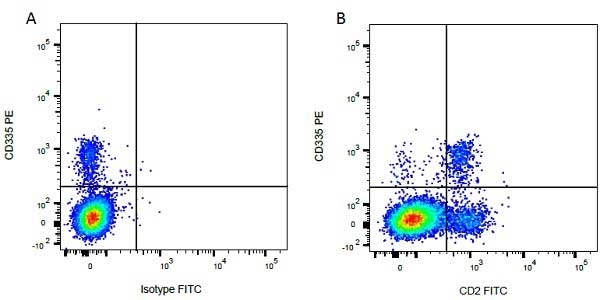
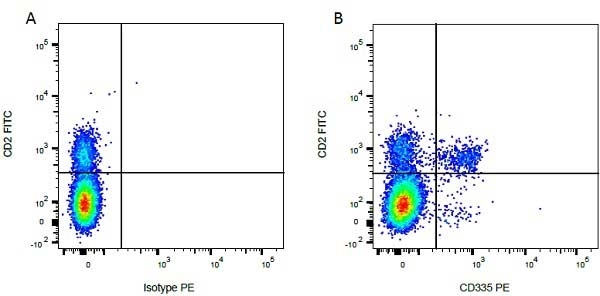
| Mouse anti Bovine CD2, clone CC42, recognizes the bovine homologue of human CD2. Clone CC42 inhibits rosetting with SRBC and stains cells in primary and secondary lymphoid organs in patterns consistent with those seen by human CD2 monoclonal antibodies. |
- Target Species
- Bovine
- Species Cross-Reactivity
-
Target Species Cross Reactivity Goat - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% Sodium Azide (NaN3)
- Carrier Free
- Yes
- Approx. Protein Concentrations
- IgG concentration 1.0 mg/ml
- Fusion Partners
- Spleen cells from an immunised mouse were fused with cells of the mouse NS1 myeloma cell line
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | 1/10 | 1/20 | |
| Immunofluorescence | |||
| Immunohistology - Frozen | |||
| Immunohistology - Paraffin 1 | |||
| Immunoprecipitation |
- 1This product requires antigen retrieval using heat treatment prior to staining of paraffin sections.
EDTA pH8.0 is recommended for this purpose.
- Flow Cytometry
- Use 10ul of the suggested working dilution to label 106 cells in 100ul
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG1 Negative Control | MCA928 | F | 100 Tests |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG1 Negative Control | ||||||
References for CD2 antibody
-
Davis, W.C. & Splitter, G.S. (1991) Individual antigens of cattle. Bovine CD2 (BoCD2).
Vet Immunol Immunopathol. 27: 43-50. -
Sopp, P. et al. (1991) Investigating monoclonal antibodies to bovine "null" cell antigens using two-colour immunofluorescence.
Vet Immunol Immunopathol. 27 (1-3): 163-8. -
Eskra, L. et al. (1991) Effect of monoclonal antibodies on in vitro function of T-cell subsets.
Vet Immunol Immunopathol. 27 (1-3): 215-22. -
Gutierrez, M. et al. (1999) The detection of CD2+, CD4+, CD8+, and WC1+ T lymphocytes, B cells and macrophages in fixed and paraffin embedded bovine tissue using a range of antigen recovery and signal amplification techniques.
Vet Immunol Immunopathol. 71 (3-4): 321-34. -
Fikri, Y. et al. (2000) Purification and characterisation of bovine WC1+ gammadelta T lymphocytes from peripheral blood.
Vet Res. 31: 229-39. -
Brackenbury, L.S. et al. (2005) Identification of a cell population that produces alpha/beta interferon in vitro and in vivo in response to noncytopathic bovine viral diarrhea virus.
J Virol. 79: 7738-44. -
Wilson, E. et al. (1999) A circulating bovine gamma delta T cell subset, which is found in large numbers in the spleen, accumulates inefficiently in an artificial site of inflammation: correlation with lack of expression of E-selectin ligands and L-selectin.
J Immunol.162: 4914-9. -
Weiss, D.J. et al. (2006) Mucosal immune response in cattle with subclinical Johne's disease.
Vet Pathol. 43: 127-35.
View The Latest Product References
-
Grell, S.N. et al. (2005) Age-dependent differences in cytokine and antibody responses after experimental RSV infection in a bovine model.
Vaccine 23: 3412-23. -
Bednarek, D. et al. (2003) Effect of steroidal and non-steroidal anti-inflammatory drugs in combination with long-acting oxytetracycline on non-specific immunity of calves suffering from enzootic bronchopneumonia.
Vet Microbiol. 96: 53-67. -
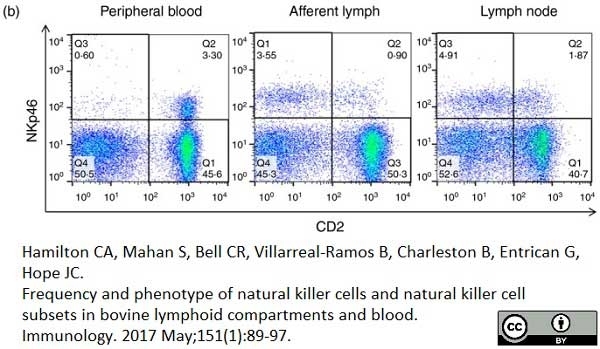
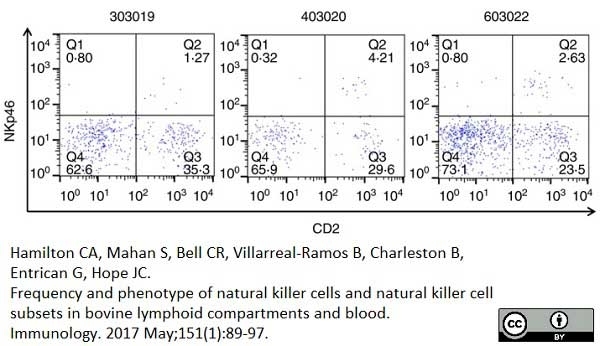
Hamilton, C.A. et al. (2017) Frequency and phenotype of natural killer cells and natural killer cell subsets in bovine lymphoid compartments and blood.
Immunology. 151 (1): 89-97. -
Elsayed, M.S.A.E. et al. (2022) Real-time PCR using atpE, conventional PCR targeting different regions of difference, and flow cytometry for confirmation of Mycobacterium bovis. in buffaloes and cattle from the Delta area of Egypt.
BMC Microbiol. 22 (1): 154.
- Synonyms
- LFA-2
- UniProt
- Q148M9
MCA833GA
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Bovine ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up