Protocol: Antigen Retrieval procedures in F4/80 staining of Paraffin-Embedded Tissues
Antigen unmasking procedures are recommended to achieve optimum IHC staining for F4/80, as the F4/80 antigen can be masked by prolonged formalin-fixation and the paraffin-embedding process.

Antigen Retrieval Techniques: For use with F4/80 Immunostaining of Paraffin-Embedded Sections
Notes
- The following two procedures should be used alongside the Indirect Immunostaining of Paraffin-Embedded Tissues protocol
- Recommended fixative for the F4/80 antigen is Neutral Buffered Formalin
-
Appropriate controls should always be carried out
- Recommended positive control tissue: Liver and lymph node
Option 1. Enzyme pre-treatment using proteinase K
A number of buffers may be used for this purpose; the precise choice is usually recommended on the product specific datasheet or determined experimentally. For your convenience Bio-Rad offers a range of pre-prepared Antigen Retrieval Buffer at different pH levels guaranteeing consistency of performance and reliability.
Alternatively, buffers may be prepared using the recipes listed below. For further details contact Technical Support
This method may cause tissue damage. Users are advised to consider incubation time, tissue type and degree of fixation when optimizing their assay.
Reagents
- 1. TE buffer (50 mM Tris base, 1 mM EDTA, pH 8.0)
- Tris Base, 6.10 g
EDTA, 0.37 g
Distilled water, 1000 ml
Mix to dissolve. Adjust pH to 8.0 using concentrated HCl (10 M HCl)
Store at room temperature - 2. 20x Proteinase K stock solution (400 μg/ml in TE buffer, pH 8.0)
- Proteinase K, 4 mg
TE buffer, pH 8.0, (Reagent 1.) 10 ml
Mix well
Store in aliquots at -20°C - 3. 1x Proteinase K working solution (20 μg/ml in TE buffer, pH 8.0)
- 20x Proteinase K stock solution, (Reagent 2.) 1 ml
TE Buffer, pH 8.0, (Reagent 1.) 19 ml
Mix well
Discard working solution after use
Method
- Dewax paraffin sections and rehydrate using preferred procedure.
- Cover sections completely with Proteinase K working solution and incubate for 3 minutes at room temperature. (N.B. As the specific activity of Proteinase K can vary between batches, it can be advantageous to determine the optimal incubation time for the current batch by comparing incubation times of 2, 3 and 4 minutes in a test run).
- Rinse sections with Phosphate Buffered Saline (PBS).
- Optional step; if required proceed with endogenous peroxidase de-activation, Bio-Rad offers peroxide blocking reagent (BUF017B).
- Proceed with preferred staining protocol.
Option 2. Heat-mediated antigen retrieval using citrate buffer, pH 6.0
Users are advised to consider tissue type and degree of fixation when optimizing their assay.
Reagent
- Citrate buffer (10 mM citric acid, pH 6.0)
- Citric acid (anhydrous), 1.92 g
Distilled water, 1000 ml
Mix to dissolve
Adjust pH to 6.0 with 1 M NaOH (be sure to mix well)
Store this solution at room temperature for 3 months, or at 4°C for longer usage
Method
- Dewax paraffin sections and rehydrate using preferred protocol.
- Pre-heat sodium citrate buffer in a staining vessel to 95-100°C.
- Immerse slides in the citrate buffer and incubate for 10 minutes at 95-100°C. Check the citrate buffer level, add more if necessary, and then incubate for a further 10 minutes at 95-100°C.
- Allow sections to cool for 20 minutes.
- Rinse sections with PBS.
- Optional step; if required proceed with endogenous peroxidase de-activation, Bio-Rad offers peroxide blocking reagent (BUF017B).
- Proceed with preferred staining protocol.
Benefits of using this antigen retrieval procedure can be seen below. Image A. shows IHC staining with no pre-treatment and image B. shows IHC staining that can be achieved after pre-treatment, clearly resulting in superior staining.
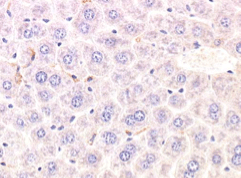
Image A. F4/80 antibody staining of liver tissue fixed in NBF for 28 days, no antigen retrieval
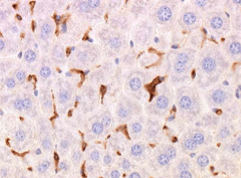
Image B. F4/80 antibody staining of liver tissue fixed in NBF for 28 days with citrate buffer & heat-mediated antigen retrieval
Recommended F4/80 Antibody
Our F4/80 antibody (Clone Cl: A3-1) (MCA497) is a well characterized and highly cited F480 antibody. Research by Citeab recently showed that it was the most cited physiology antibody, with 155 citations (at the time of publishing).
Related pages:
- Crucial Controls and Tips For IHC Experiments
- IHC1. Indirect Immunostaining of Frozen Tissue Sections
- IHC2. Streptavidin-Biotin Immunostaining of Frozen Tissue Sections
- IHC3. PAP/APAAP Immunostaining of Frozen Tissue Sections
- IHC4. Indirect Immunostaining of Paraffin-Embedded Tissue Sections
- IHC5. Streptavidin-Biotin Immunostaining of Paraffin-Embedded Tissue Sections
- IHC6. PAP/APAAP immunostaining of paraffin embedded tissue sections
- IHC7. Antigen Retrieval Techniques for use with Formalin-Fixed Paraffin-Embedded Sections
- IHC8. Guidance for staining with Mouse anti-Bromodeoxyuridine
- Fixative Recipes







