
Popular topics

-
References
Dejnirattisai W et al. (2015). A new class of highly potent, broadly neutralizing antibodies isolated from viremic patients infected with dengue virus. Nature Immunology. 16, 170-177
Four virus serotypes…One Dengue vaccine?

After years of research, a team of international researchers believe they have found the key to a universal vaccine for the currently incurable Dengue virus. This mosquito-borne systemic disease affects around 400 million people annually with approximately 25% of the cases being symptomatic. Figure 1 identifies the areas commonly affected which include South-East Asia, the Western Pacific, Latin America and the Caribbean.
There are four serotypes of the dengue virus (DENV-1, DENV-2, DENV-3 and DENV-4) differing by 30-35% in amino acid sequence. The first encounter with a Dengue virus strain is not often fatal and results in immunity of that strain but only limited or temporary cross-immunity against other strains. A second attack however, from another strain, increases severity of the infection which can result in hemorrhagic fever leading to severe bleeding and organ failure.
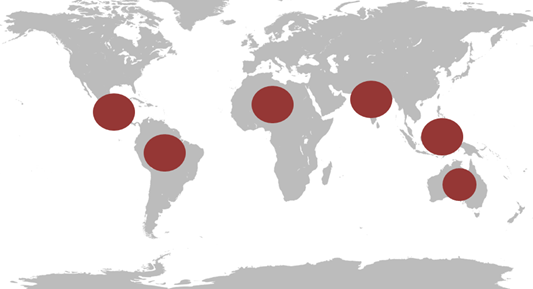
Fig. 1. This image shows areas which are affected by Dengue virus.
This immune response is explained by a process known as antibody-dependent enhancement (ADE) (shown in Figure 2), where the virus binds to the antigen binding site of an antibody, which then binds to the Fc receptor of certain host cells. Viruses use this mechanism to infect human macrophages and monocytes leading to enhanced infection and greater virus production.
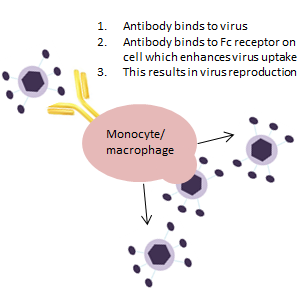
Fig.2. This image gives a snapshot on the process of antibody-dependent enhancement (ADE)
Current methods for vaccine development have focused on raising neutralising antibodies against the DENV envelope (E) protein which coats the virus. Published in Nature Immunology, this group explain how they analysed 145 monoclonal antibodies that target the DENV E protein from patients infected with Dengue virus. They discovered an envelope dimer epitope (EDE), which joins together two envelope (E) protein subunits that make up the viral envelope of the mature viron. The mature viron contains 180 E proteins that form 90 dimers arranged through icosahedral symmetry forming a smooth glycoprotein shell.
The need for a vaccine that can neutralize the effects of all four dengue virus stains has been a tough challenge. For one, there are several conformations of the virus glycoprotein shell that occur at different phases of the virus life cycle. Secondly, during a natural infection with DENV the host is presented with two virus forms, those produced in insect and human cells. Previous studies on neutralizing the Dengue virus have focused on targeting anti-Dengue antibodies against the fusion-loop epitope (FLE) present on the E protein, which is used to fuse viral and host cell membranes. Both FLE and EDE monoclonal antibodies are able to fully neutralize insect-DENV, whereas only antibodies against EDE are able to effectively neutralise human-DENV.
The hope from this study is that the development of a vaccine which was based upon using four type-specific anti-Dengue antibodies against all four virus serotypes, can now focus on a single antigen.
References
Dejnirattisai W et al. (2015). A new class of highly potent, broadly neutralizing antibodies isolated from viremic patients infected with dengue virus. Nature Immunology. 16, 170-177
You may also be interested in...

View more immunology or science-news blogs














