Using Secondary Reagents in Flow Cytometry with Confidence

- On This Page
- Indirect staining
- Guidelines for indirect staining in fc using secondary detection reagents
- Useful flow cytometry links
Flow cytometry predominantly uses direct staining with conjugated primary antibodies to detect markers of interest, especially when multiplexing for immunophenotyping, however, not all antibodies are available with a fluorescent marker or one that is compatible with other fluorophores or fluorescent proteins. For example, if an antibody is only available in an unconjugated version, or only a FITC conjugate is available and staining is required in a GFP positive sample. Often the solution to this problem is to conjugate the unlabeled primary antibody to a fluorophore of your choice, using a LYNX or ReadiLink Kit. However, there may be occasions when this approach is not practical; there may be a limited amount of antibody available, the antibody may be impure or contain a carrier protein, critical amino acids at the antigen binding site may be disrupted by conjugation, or in the case of screening hybridomas there may simply be too many antibodies. In this situation, indirect staining using a secondary antibody is invaluable for detecting your marker.
Indirect Staining Using Secondary Detection Reagents
When direct staining is impractical, the use of secondary detection reagents will give you the flexibility to choose from a range of fluorophores to fit into most flow cytometry panels. As multiple secondary antibody molecules will bind to each primary antibody, the signal will be amplified, a great benefit when looking at low density antigens. With careful optimization, excellent staining can still be obtained using secondary antibodies, as can be seen in Figure 1. Note, with signal amplication, all nonspecific binding and incorrect binding will also be amplified. There are however some simple guidelines that you can follow to ensure you have specific and optimized staining to maximize your results.
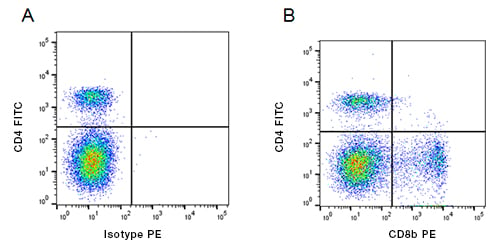
Fig. 1. Staining of bovine peripheral blood T lymphocytes using direct and indirect staining. A, FITC conjugated Mouse Anti-Bovine CD4 (MCA1653F) and Mouse IgG1 Isotype Control (MCA928) detected with PE conjugated Anti-Mouse IgG1 (STAR132PE). B, FITC conjugated Mouse Anti-Bovine CD4 (MCA1653F) and Mouse Anti-Bovine CD8 Beta (MCA1654GA) detected with PE conjugated Anti-Mouse IgG1 (STAR132PE). All experiments performed on bovine blood gated on live single lymphocytes, in the presence of 10% bovine serum. Data acquired on the ZE5 Cell Analyzer.
Guidelines for Indirect Staining in Flow Cytometry Using Secondary Detection Reagents
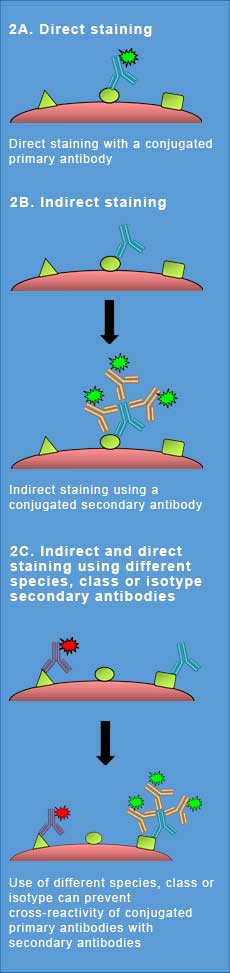
Fig. 2. Staining Strategies. Illustration of the different types of staining protocols that may need to be employed to have successful indirect and direct staining in flow cytometry.
1. Simple Single Primary Antibody Staining
When staining samples with a directly conjugated primary antibody, after the initial incubation and a few simple washes, the sample is ready to be acquired (Figure 2A). Indirect staining using an unconjugated primary antibody, detected with a secondary antibody, involves extra steps: incubate with the primary antibody, wash, and then incubate with the fluorescently-labeled secondary antibody that recognizes your primary antibody. After more washes, the sample is ready to be acquired (Figure 2B).
However if you have a combination of conjugated and unconjugated primary antibodies, which require secondary antibodies in your panel, staining can be more complicated. To avoid nonspecific binding, there are staining protocols, blocking and washing steps, and antibody choices to consider in addition to the usual flow cytometry controls.
2. Combining Conjugated and Unconjugated Antibodies
A common problem when combining unconjugated and conjugated antibodies is inappropriate detection of the conjugated primary antibody/antibodies by the secondary antibody, leading to false positives. This problem is illustrated in a isotype specific secondary antibodies video which demonstrates practically how using Bio-Rad’s mouse IgG isotype specific secondary antibodies can deliver results you can trust in imaging multiplexing, without the risk of false positives, and in only two simple steps. To avoid this problem, it is necessary to be able to distinguish between the unconjugated and conjugated primary antibodies by means of species, class, or isotype. The primary antibodies can be incubated together, washed and then labeled with secondary antibodies, which only detect the species, class, or isotype of the unconjugated primary antibodies (Figure 2C). To optimize your staining, ideally both the primary and secondary antibodies should be titrated to ensure minimum background with a maximal specific signal. It is also recommended to include a secondary antibody-only control and a control containing all antibodies except the unlabeled primary antibody to check for nonspecific staining. To get help with your staining, view our detailed staining protocols.
3. Blocking and Washing
Incubation with an appropriate serum, before adding primary antibodies, can reduce background by blocking Fc receptors. This should be of the same species as the cell type being analyzed, never the same species as the host of the primary antibody, as the secondary antibody will recognize the blocking serum AND the primary antibody. If you are using bovine serum albumin, it may be contaminated with bovine IgG which can cross-react with closely related species such as goat and sheep. Include adequate washing steps to remove excess primary and secondary antibodies. Intracellular staining protocols may require more and longer washing steps to remove all the excess antibody. Furthermore, the choice of permeabilization and wash buffer may affect staining and should be optimized for your experiment.
4. Antibody Choice
The antibody choice for your primary and secondary antibodies will influence your experimental design. Unwanted species cross-reactivity can be removed by using cross-adsorbed or isotype specific secondary antibodies. This will ensure the signal from the secondary antibody is specific to the primary target. If you are staining immune tissue, which contains a lot of Fc receptors, you may want to consider a F(ab) fragment as a secondary antibody in addition to blocking with serum to reduce nonspecific binding. When multiplexing, if there are no primary antibodies raised in different species, different classes of immunoglobulin may be available, for example IgG and IgM, allowing class specific secondary antibodies to be used, from the same species. Many monoclonal primary antibodies are mouse or rat IgG1, IgG2a, or IgG2b. Isotype specific secondary antibodies allow multiplexing of purified antibodies from the same species. You may be able to increase your staining options by mixing and matching these methods, however always ensure your secondary antibodies do not recognize multiple targets and the correct controls are performed.
5. Compensation
When using labeled secondary antibodies in a multicolor panel, you should still perform the correct compensation control for the secondary fluorophore. The easiest way is to carry out the single staining compensation control using the unconjugated primary antibody and the associated secondary antibody. If this proves problematical, due to low expression for example, then compensation beads can be used. Depending upon the antibody-binding capability of the beads, it may be possible to use these directly with the secondary antibody, or with the primary plus secondary antibodies. A third option is to perform compensation using a primary antibody directly conjugated to the same fluorophore that will be used on the secondary antibody. However, the use of tandem fluorophores with this method should be done with extreme caution due to inherent differences between tandem fluorophore batches.
6. Streptavidin as a Secondary Reagent
Sometimes only a biotinylated primary antibody is available, in this case, the use of fluorescently labeled streptavidin with such an antibody will make it suitable in flow cytometry without needing secondary antibodies. This option removes the need to check for cross-reactivity of secondary antibodies with other primary antibodies and can expand co-staining options. However, it is still necessary to perform the correct negative and compensation controls.
7. General Flow Controls/Tips
Specific controls are required for flow cytometry such as unstained, isotype, viability, compensation, and FMO controls. Building multicolor panels can be tricky and following guidelines related to instrument and fluorophore choice are equally important when performing indirect staining. In addition to these standard flow cytometry controls and guidelines, use of secondary antibodies may require additional controls and optimization of both antibodies and staining protocol. The nonspecific binding of the secondary antibody can be assessed by staining with the secondary antibody without the specific primary antibody, either alone, or in combination with primary antibodies that are not the intended target. Alternatively a primary antibody, which the secondary antibody binds to, and known to be negative in your sample, can be used. This will give you an idea of the binding to both the sample and other antibodies in the panel, allowing you to assess the nonspecific background staining.
For more information on secondary antibodies and their use in various applications, visit our dedicated secondary antibodies webpage.
Flow Cytometry Secondary Antibody Range
| Description | Target | Host | Specificity | Format | Citations | Isotype | Code |
|---|





