MHC Class II RT1B antibody | OX-6
Mouse anti Rat MHC Class II RT1B
- Product Type
- Monoclonal Antibody
- Clone
- OX-6
- Isotype
- IgG1
- Specificity
- MHC Class II RT1B
Filter by Application:
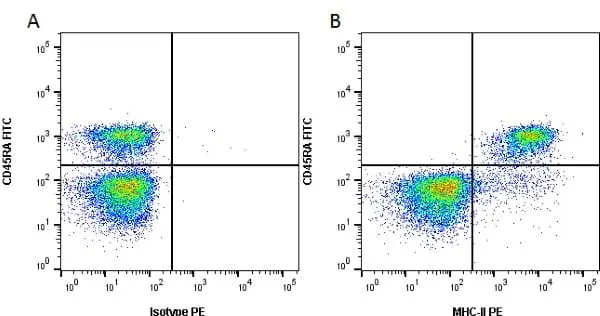
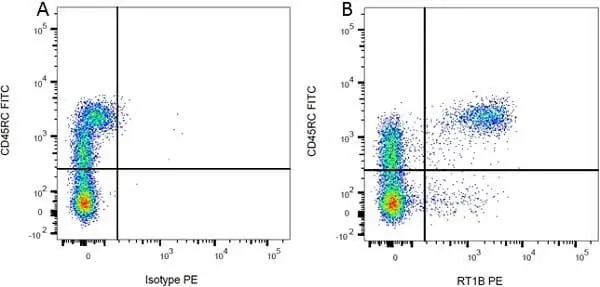
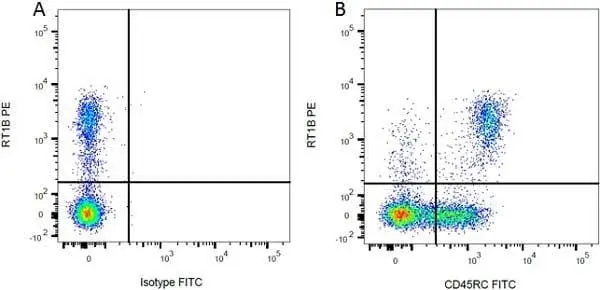
F IF C Reset| Mouse anti Rat MHC Class II RT1B antibody, clone OX-6 recognizes a monomorphic determinant of the rat RT1B MHC class II antigen present on B lymphocytes, dendritic cells, some macrophages and certain epithelial cells. Rat MHC Class II RT1B antibody, clone OX-6 does not react with the rat BDIX strain due to a defect in RT1B expression (Male et al. 1987). The major histocompatibility complex (MHC) is a cluster of genes that are important in the immune response to infections. In rats, this complex is referred to as the RT1 region. In mice, this complex is referred to as the H-2 region. Mouse anti Rat MHC Class II RT1B antibody, clone OX-6 also cross reacts with a polymorphic determinant on mouse strains of the H-2 haplotypes k and s. Analysis of recombinant mouse strains has mapped the OX-6 determinant to the H-2I-A region (McMaster and Williams 1979 and Male et al. 1987). Mouse anti Rat MHC Class II RT1B antibody, clone OX-6 is routinely tested in flow cytometry on rat splenocytes. |
- Target Species
- Rat
- Species Cross-Reactivity
-
Target Species Cross Reactivity Mouse - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
0.09% Sodium Azide - Carrier Free
- Yes
- Immunogen
- Rat thymocyte membrane glycoproteins.
- Approx. Protein Concentrations
- IgG concentration 1.0 mg/ml
- Fusion Partners
- Spleen cells from immunized BALB/c mice were fused with cells from the NS1 mouse myeloma cell line.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | 1/50 | 1/100 | |
| Immunofluorescence | |||
| Immunohistology - Frozen | |||
| Immunohistology - Paraffin 1 | 1/50 | 1/100 |
- 1This product requires antigen retrieval using heat treatment prior to staining of paraffin sections.Sodium citrate buffer pH 6.0 is recommended for this purpose. PLP fixation is recommended for optimal results.
- Flow Cytometry
- Use 10ul of the suggested working dilution to label 106 cells in 100ul.
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG1 Negative Control | MCA1209 | F | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG1 Negative Control | ||||||
Source Reference
-
McMaster, W.R. & Williams, A.F. (1979) Identification of Ia glycoproteins in rat thymus and purification from rat spleen.
Eur J Immunol. 9 (6): 426-33.
References for MHC Class II RT1B antibody
-
Fernandez, J.L. & Weeks, M. (1986) Genetic monitoring of inbred strains of mice using monoclonal antibodies to major histocompatibility haplotypes and lymphocyte alloantigens.
Lab Anim. 20 (4): 293-7. -
Male, D.K. et al. (1987) Serological evidence for a defect in RT1.B (I-A) expression by the BDIX rat strain.
J Immunogenet. 14 (6): 301-12. -
Charteris, D.G. & Lightman, S.L. (1993) In vivo lymphokine production in experimental autoimmune uveoretinitis.
Immunology. 78 (3): 387-92. -
Whiteland, J.L. et al. (1995) Immunohistochemical detection of T-cell subsets and other leukocytes in paraffin-embedded rat and mouse tissues with monoclonal antibodies.
J Histochem Cytochem. 43 (3): 313-20. -
McKechnie, N.M. et al. (1997) Immunization with the cross-reactive antigens Ov39 from Onchocerca volvulus and hr44 from human retinal tissue induces ocular pathology and activates retinal microglia.
J Infect Dis. 176 (5): 1334-43. -
Burrows, G.G. et al. (1998) Two-domain MHC class II molecules form stable complexes with myelin basic protein 69-89 peptide that detect and inhibit rat encephalitogenic T cells and treat experimental autoimmune encephalomyelitis.
J Immunol. 161 (11): 5987-96. -
Hofmann, N. et al. (2002) Increased expression of ICAM-1, VCAM-1, MCP-1, and MIP-1 alpha by spinal perivascular macrophages during experimental allergic encephalomyelitis in rats.
BMC Immunol. 3:11. -
Banerjee, S. et al. (2003) Development of organised conjunctival leucocyte aggregates after corneal transplantation in rats.
Br J Ophthalmol. 87: 1515-22.
View The Latest Product References
-
Bode, U. et al. (2008) Dendritic cell subsets in lymph nodes are characterized by the specific draining area and influence the phenotype and fate of primed T cells.
Immunology. 123 (4): 480-90. -
King, G.D. et al. (2008) Flt3L in combination with HSV1-TK-mediated gene therapy reverses brain tumor-induced behavioral deficits.
Mol Ther. 16: 682-90. -
Wang, Q. et al. (2009) Pyruvate protects against experimental stroke via an anti-inflammatory mechanism.
Neurobiol Dis. 2009 Oct;36(1):223-31. -
Zilka, N. et al. (2009) Human misfolded truncated tau protein promotes activation of microglia and leukocyte infiltration in the transgenic rat model of tauopathy.
J Neuroimmunol. 209 (1-2): 16-25. -
Baca Jones, C.C. et al. (2009) Rat cytomegalovirus infection depletes MHC II in bone marrow derived dendritic cells.
Virology. 388: 78-90. -
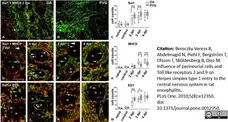
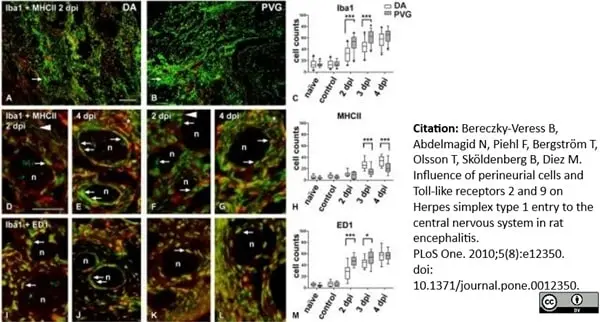
Bereczky-Veress, B. et al. (2010) Influence of perineurial cells and Toll-like receptors 2 and 9 on Herpes simplex type 1 entry to the central nervous system in rat encephalitis.
PLoS One. 5(8): e12350. -
Kawamura, J. et al. (2010) Neuron-immune Interactions in the Sensitized Thalamus Induced by Mustard Oil Application to Rat Molar Pulp.
J Dent Res. 89: 1309-14. -
Calvo, M. et al. (2010) Neuregulin-ErbB signaling promotes microglial proliferation and chemotaxis contributing to microgliosis and pain after peripheral nerve injury.
J Neurosci. 30 (15): 5437-50. -
McClain, J.A. et al. (2011) Adolescent binge alcohol exposure induces long-lasting partial activation of microglia.
Brain Behav Immun. 25 Suppl 1: S120-8. -
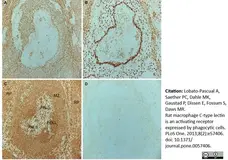
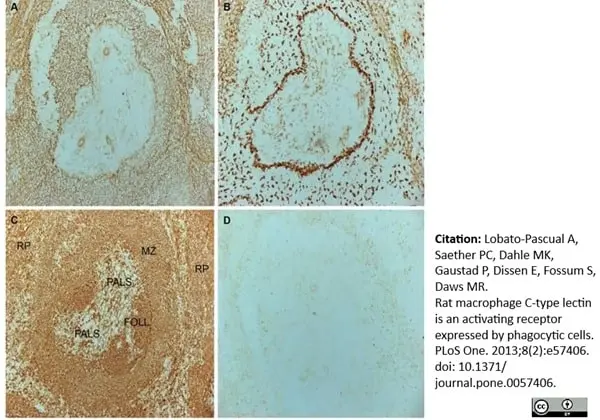
Lobato-Pascual, A. et al. (2013) Rat macrophage C-type lectin is an activating receptor expressed by phagocytic cells.
PLoS One. 8: e57406. -
Maneu, V. et al. (2016) Immunosuppression, peripheral inflammation and invasive infection from endogenous gut microbiota activate retinal microglia in mouse models.
Microbiol Immunol. 60 (9): 617-25. -
Takizawa, T. et al. (2017) High-mobility group box 1 is an important mediator of microglial activation induced by cortical spreading depression.
J Cereb Blood Flow Metab. 37 (3): 890-901. -
Liu, M. et al. (2017) Pioglitazone Attenuates Neuroinflammation and Promotes Dopaminergic Neuronal Survival in the Nigrostriatal System of Rats after Diffuse Brain Injury.
J Neurotrauma. 34 (2): 414-22. -
Collins, J.J.P. et al. (2018) Impaired Angiogenic Supportive Capacity and Altered Gene Expression Profile of Resident CD146+ Mesenchymal Stromal Cells Isolated from Hyperoxia-Injured Neonatal Rat Lungs.
Stem Cells Dev. 27 (16): 1109-24. -
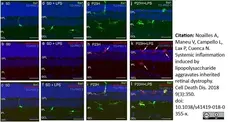
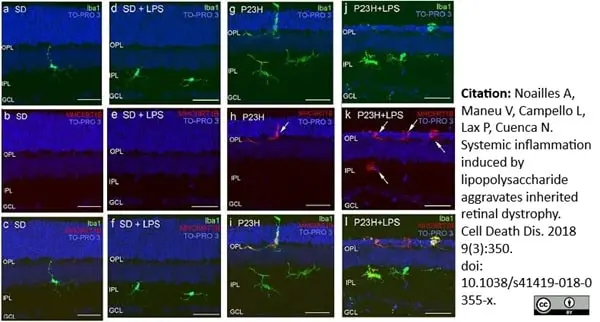
Noailles, A. et al. (2018) Systemic inflammation induced by lipopolysaccharide aggravates inherited retinal dystrophy.
Cell Death Dis. 9 (3): 350. -
Lodygin, D. et al. (2019) β-Synuclein-reactive T cells induce autoimmune CNS grey matter degeneration.
Nature. 566 (7745): 503-8. -
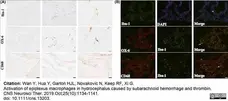
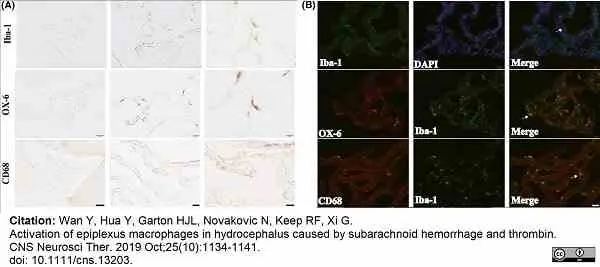
Wan, Y. et al. (2019) Activation of epiplexus macrophages in hydrocephalus caused by subarachnoid hemorrhage and thrombin.
CNS Neurosci Ther. 25 (10): 1134-41. -
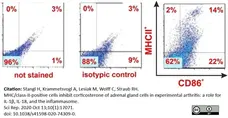
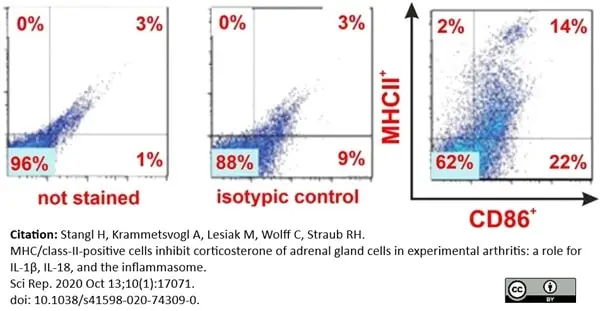
Stangl, H. et al. (2020) MHC/class-II-positive cells inhibit corticosterone of adrenal gland cells in experimental arthritis: a role for IL-1β, IL-18, and the inflammasome.
Sci Rep. 10 (1): 17071. -
Sinha, S. et al. (2020) Maternal Spirulina supplementation during pregnancy and lactation partially prevents oxidative stress, glial activation and neuronal damage in protein malnourished F1 progeny.
Neurochem Int. 141: 104877. -
Koppe, C. et al. (2021) Local Inflammatory Response after Intramuscularly Implantation of Anti-Adhesive Plasma-Fluorocarbon-Polymer Coated Ti6AI4V Discs in Rats.
Polymers (Basel). 13 (16): 2684. -
Campello, L. et al. (2020) New Nrf2-Inducer Compound ITH12674 Slows the Progression of Retinitis Pigmentosa in the Mouse Model rd10.
Cell Physiol Biochem. 54 (1): 142-59. -
Matsuyama, S. et al. (2021) Properties of macrophages and lymphocytes appearing in rat renal fibrosis followed by repeated injection of cisplatin.
J Vet Med Sci. 83 (9): 1435-42. -
Čepcová, D. et al. (2021) The protective effect of 1-methyltryptophan isomers in renal ischemia-reperfusion injury is not exclusively dependent on indolamine 2,3-dioxygenase inhibition.
Biomed Pharmacother. 135: 111180. -
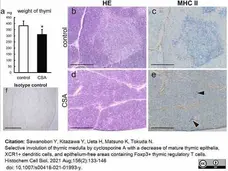
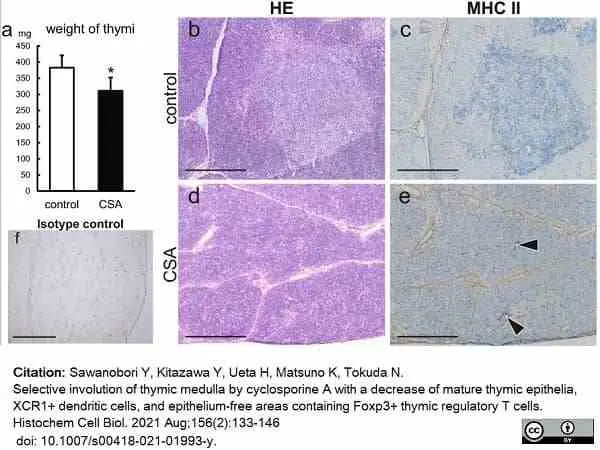
Sawanobori, Y. et al. (2021) Selective involution of thymic medulla by cyclosporine A with a decrease of mature thymic epithelia, XCR1+ dendritic cells, and epithelium-free areas containing Foxp3+ thymic regulatory T cells.
Histochem Cell Biol. 156 (2): 133-146. -
Silva, B.A. et al. (2022) Understanding the role of the blood brain barrier and peripheral inflammation on behavior and pathology on ongoing confined cortical lesions.
Mult Scler Relat Disord. 57: 103346. -
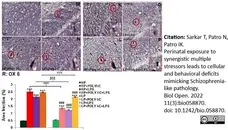
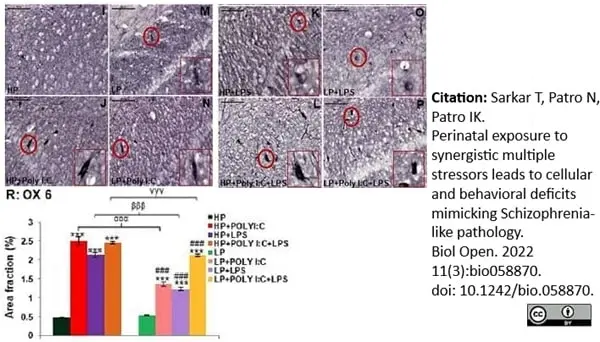
Sarkar, T. et al. (2022) Perinatal exposure to synergistic multiple stressors leads to cellular and behavioral deficits mimicking Schizophrenia-like pathology.
Biol Open. 11(3):bio058870. -
Pervin, M. et al. (2022) Possible Cytoprotection of Low Dose Lipopolysaccharide in Rat Thioacetamide-Induced Liver Lesions, Focusing on the Analyses of Hepatic Macrophages and Autophagy.
Toxicol Pathol. : 1926233221076758. -
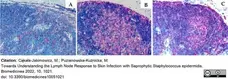
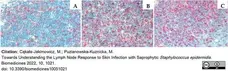
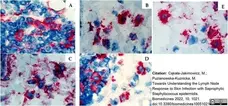
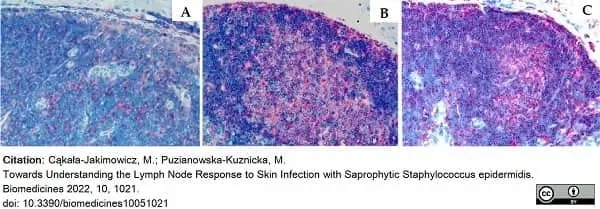
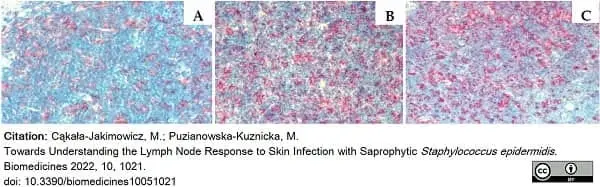
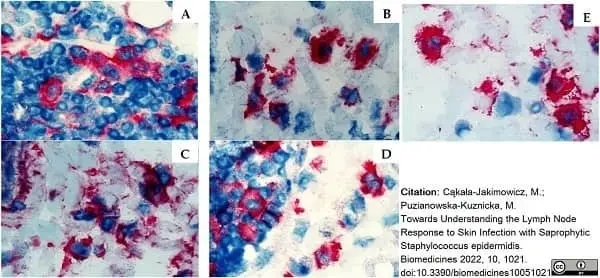
Cąkała-Jakimowicz, M. & Puzianowska-Kuznicka, M. (2022) Towards Understanding the Lymph Node Response to Skin Infection with Saprophytic Staphylococcus epidermidis.
Biomedicines. 10(5):1021. -
Zakani, M. et al. (2023) Paths to hippocampal damage in neuromyelitis optica spectrum disorders
Neuropathol App Neurobiol. 49 (2) e12893 -
Takami, Y. et al. (2023) The effect of lipopolysaccharide on liver homeostasis and diseases based on the mutual interaction of macrophages, autophagy, and damage-associated molecular patterns in male F344/DuCrlCrlj rats.
Vet Pathol. 60 (4): 461-72. -
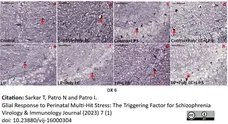
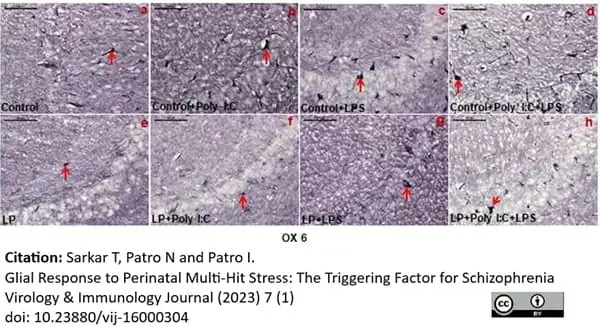
Sukar, T. et al. (2023) Glial Response to Perinatal Multi-Hit Stress: The Triggering Factor for Schizophrenia
Virology & Immunology Journal. 7 (1): 1-21. -
Pervin, M. et al. (2021) Immunophenotypic analysis of the distribution of hepatic macrophages, lymphocytes and hepatic stellate cells in the adult rat liver.
Anat Histol Embryol. 50 (4): 736-745.
- RRID
- AB_322113
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Rat ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up