CD90 antibody | OX-7
Mouse anti Rat CD90
- Product Type
- Monoclonal Antibody
- Clone
- OX-7
- Isotype
- IgG1
- Specificity
- CD90
| Product Code | Applications | Pack Size | List Price | Your Price | Qty | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
Filter by Application:
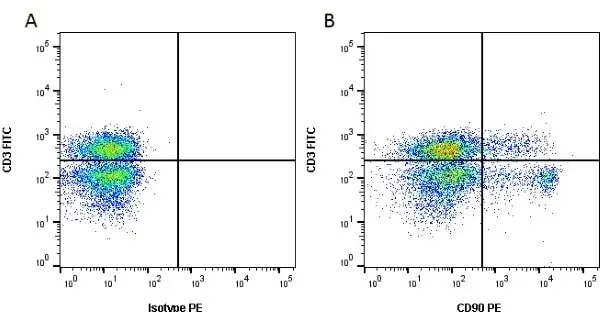
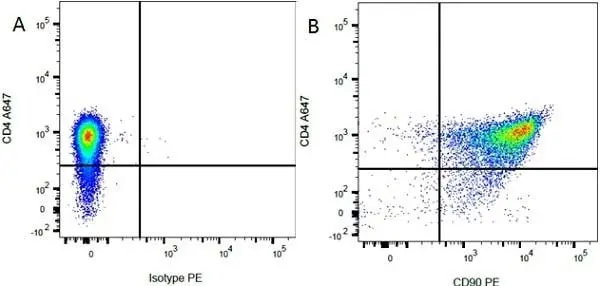
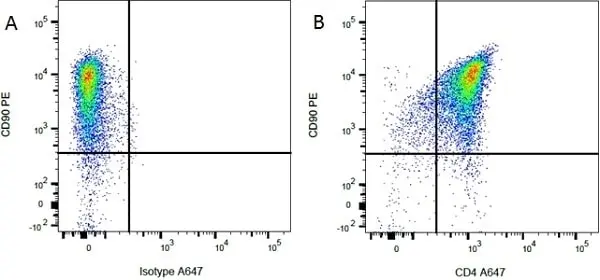
F C Reset| Mouse anti Rat CD90 antibody, clone OX-7 recognizes rat and CD90, also known as Thy1.1, a GPI-anchored membrane protein containing a single V type Ig-like domain CD90 is expressed on a variety of cell types including thymocytes, neuronal cells, stem cells, immature B cells and connective tissues, CD90 is also expressed in T cells in mice. Since Thy1.1 is a monomorphic determinant in rat but polymorphic in mice, clone MRC OX-7 reacts with Thy1.1 mice e.g. AKR and FVB, but not Thy1.2 mice such as CBA and BALB/c. The affinity of the Fab′ of MRC OX-7 for rat Thy1 is 3 x 109m-1 and for mouse Thy1.1 is 3 x 108m-1(1). Mouse anti rat CD90, clone MRC OX-7 has been demonstrated to promote neurite outgrowths on peripherin-stained sympathetic rat neurons, using fluorescence microscopy (Jeng et al. 1998). Clone OX-7 has also been reported to induce glomerular nephritis in Wistar rats (Tamura et al. 1996). This product is routinely tested in flow cytometry on rat thymocytes. |
- Target Species
- Rat
- Species Cross-Reactivity
-
Target Species Cross Reactivity Rabbit Mouse Guinea Pig - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
0.09% Sodium Azide - Carrier Free
- Yes
- Immunogen
- Rat Thy1 antigen.
- Approx. Protein Concentrations
- IgG concentration 1.0 mg/ml
- Fusion Partners
- Spleen cells from immunized BALB/c mice were fused with cells of the mouse NS1 myeloma cell line.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | 1/50 | 1/100 | |
| Immunofluorescence | |||
| Immunohistology - Frozen | |||
| Immunoprecipitation | |||
| Western Blotting |
- Flow Cytometry
- Use 10ul of the suggested working dilution to label 106 cells in 100ul.
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG1 Negative Control | MCA1209 | F | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG1 Negative Control | ||||||
Source Reference
-
Mason, D.W. & Williams, A.F. (1980) The kinetics of antibody binding to membrane antigens in solution and at the cell surface.
Biochem J. 187 (1): 1-20.
References for CD90 antibody
-
Campbell, D.G. et al. (1981) Rat brain Thy-1 glycoprotein. The amino acid sequence, disulphide bonds and an unusual hydrophobic region.
Biochem J. 195 (1): 15-30. -
Bukovský, A. et al. (1983) The localization of Thy-1.1, MRC OX 2 and Ia antigens in the rat ovary and fallopian tube.
Immunology. 48 (3): 587-96. -
Kawachi, H. et al. (1992) Epitope-specific induction of mesangial lesions with proteinuria by a MoAb against mesangial cell surface antigen.
Clin Exp Immunol. 88 (3): 399-404. -
Tamura, M. et al. (1996) Enhanced glomerular profilin gene and protein expression in experimental mesangial proliferative glomerulonephritis.
Biochem Biophys Res Commun. 222 (3): 683-7. -
Banerjee, S.A. et al. (1997) An antibody to the tetraspan membrane protein CD9 promotes neurite formation in a partially alpha3beta1 integrin-dependent manner.
J Neurosci. 17 (8): 2756-65. -
Lee, W.S. et al. (1998) Thy-1, a novel marker for angiogenesis upregulated by inflammatory cytokines.
Circ Res. 82 (8): 845-51. -
Keller, R.K. et al. (2004) Formation of 7-dehydrocholesterol-containing membrane rafts in vitro and in vivo, with relevance to the Smith-Lemli-Opitz syndrome.
J Lipid Res. 45: 347-55. -
Rutigliano, J.A. et al. (2008) Screening monoclonal antibodies for cross-reactivity in the ferret model of influenza infection.
J Immunol Methods. 336: 71-7.
View The Latest Product References
-
Stevenson, K.S. et al. (2009) Isolation, characterization, and differentiation of thy1.1-sorted pancreatic adult progenitor cell populations.
Stem Cells Dev. 18 (10): 1389-98. -
Ohashi, N. et al. (2010) Glomerular angiotensinogen is induced in mesangial cells in diabetic rats via reactive oxygen species--ERK/JNK pathways.
Hypertens Res. 33:1174-81. -
Biermann, J. et al. (2011) Histone deacetylase inhibitors sodium butyrate and valproic acid delay spontaneous cell death in purified rat retinal ganglion cells.
Mol Vis. 17: 395-403. -
Freisinger, W. et al. (2013) Sensory renal innervation: a kidney-specific firing activity due to a unique expression pattern of voltage-gated sodium channels?
Am J Physiol Renal Physiol. 304: F491-7. -
Shimizu T et al. (2016) Bioactivity of sol-gel-derived TiO2 coating on polyetheretherketone: In vitro and in vivo studies.
Acta Biomater. 35: 305-17. -
Maia, L. et al. (2017) Conditioned medium: a new alternative for cryopreservation of equine umbilical cord mesenchymal stem cells.
Cell Biol Int. 41 (3): 239-48. -
Maia, L. et al. (2017) A proteomic study of mesenchymal stem cells from equine umbilical cord.
Theriogenology. 100: 8-15. -
Zhao, Y. et al. (2017) A new electrospun graphene-silk fibroin composite scaffolds for guiding Schwann cells.
J Biomater Sci Polym Ed. 28 (18): 2171-85. -
Chang, J.C. et al. (2019) Early Immune Response to Acute Gastric Fluid Aspiration in a Rat Model of Lung Transplantation.
Exp Clin Transplant. 17 (1): 84-92. -
Huang, X. et al. (2019) MRI Tracking of SPIO- and Fth1-Labeled Bone Marrow Mesenchymal Stromal Cell Transplantation for Treatment of Stroke.
Contrast Media Mol Imaging. 2019: 5184105. -
Kuriyama, T. et al. (2020) A novel rat model of inflammatory bowel disease developed using a device created with a 3D printer.
Regen Ther. 14: 1-10. -
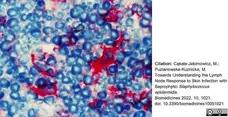
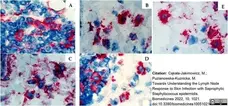
Cąkała-Jakimowicz, M. & Puzianowska-Kuznicka, M. (2022) Towards Understanding the Lymph Node Response to Skin Infection with Saprophytic Staphylococcus epidermidis..
Biomedicines. 10 (5): 1021. -
Huang, S. et al. (2022) Hydrogen sulfide supplement preserves mitochondrial function of retinal ganglion cell in a rat glaucoma model.
Cell Tissue Res. 389 (2): 171-85. -
Cacciamali, A. et al. (2022) Engineered nanoparticles toxicity on adipose tissue derived mesenchymal stem cells: A preliminary investigation
Res Vet Sci. 152: 134-49. -
Eweida, A. et al. (2022) Systemically injected bone marrow mononuclear cells specifically home to axially vascularized tissue engineering constructs.
PLoS One. 17 (8): e0272697. -
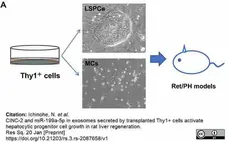
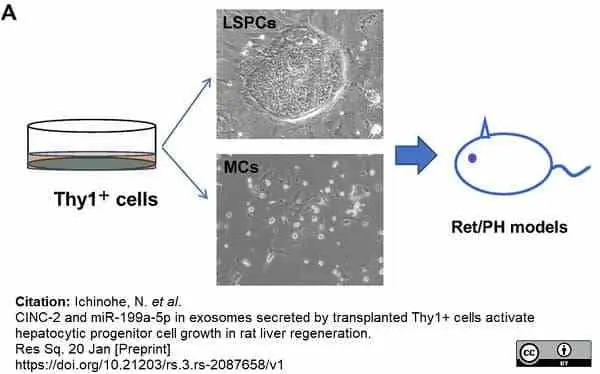
Ichinohe, N. et al. (2023) CINC-2 and miR-199a-5p in EVs secreted by transplanted Thy1(+) cells activate hepatocytic progenitor cell growth in rat liver regeneration.
Stem Cell Res Ther. 14 (1): 134. -
Numata-Uematasu, Y. et al. (2023) In vitro myelination using explant culture of dorsal root ganglia: An efficient tool for analyzing peripheral nerve differentiation and disease modeling.
PLoS One. 18 (5): e0285897. -
Gao, H. et al. (2025) Monocyte Membrane-Fused Liposomes for Enhanced Targeted Treatment of Mesangioproliferative Glomerulonephritis.
ACS Appl Mater Interfaces. 17 (50): 67641-67654.
- Synonyms
- THY1
- RRID
- AB_321889
- UniProt
- P01830
- Entrez Gene
- Thy1
- GO Terms
- GO:0001525 angiogenesis
- GO:0007010 cytoskeleton organization
- GO:0005829 cytosol
- GO:0005100 Rho GTPase activator activity
- GO:0006469 negative regulation of protein kinase activity
- GO:0016337 cell-cell adhesion
- GO:0019901 protein kinase binding
- GO:0030336 negative regulation of cell migration
- GO:0030426 growth cone
- View More GO Terms
- GO:0034235 GPI anchor binding
- GO:0043066 negative regulation of apoptosis
- GO:0043547 positive regulation of GTPase activity
- GO:0045576 mast cell activation
- GO:0046549 retinal cone cell development
- GO:0048041 focal adhesion assembly
- GO:0048147 negative regulation of fibroblast proliferation
- GO:0050731 positive regulation of peptidyl-tyrosine phosphorylation
- GO:0050771 negative regulation of axonogenesis
- GO:0050852 T cell receptor signaling pathway
- GO:0050860 negative regulation of T cell receptor signaling pathway
- GO:0050870 positive regulation of T cell activation
- GO:0051281 positive regulation of release of sequestered calcium ion into cytosol
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Rat ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up