CD8 Alpha antibody | OX-8








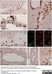
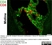
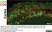




Mouse anti Rat CD8 Alpha
- Product Type
- Monoclonal Antibody
- Clone
- OX-8
- Isotype
- IgG1
- Specificity
- CD8 Alpha
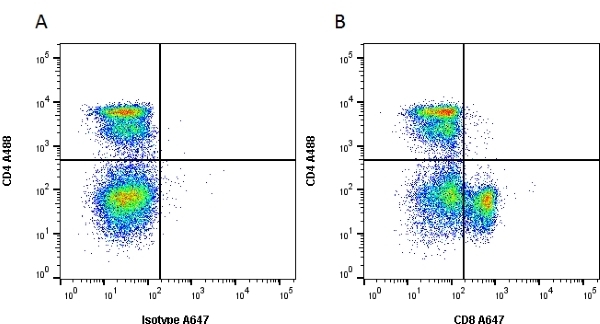
| Mouse anti Rat CD8α, clone MRC OX-8, recognizes the rat CD8 alpha cell surface antigen, expressed by a subset of T lymphocytes, most thymocytes and the majority of NK cells. Mouse anti Rat CD8α, clone MRC OX-8 is suitable for use in in vitro blocking assays (Popov et al.2001). Mouse anti Rat CD8α, clone MRC OX-8 reacts with paraffin-embedded material following PLP Fixation (periodate-lysine paraformaldehyde) (Whiteland et al. 1995). Mouse anti Rat CD8α, clone MRC OX-8 is routinely tested in flow cytometry on rat splenocytes. |
- Target Species
- Rat
- Product Form
- Purified IgG - liquid
- Preparation
- MCA48GA, MCA48R: Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant.
- MCA48G: Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
0.09% Sodium Azide - Carrier Free
- Yes
- Immunogen
- Rat thymocyte membrane glycoproteins.
- Approx. Protein Concentrations
- IgG concentration 1.0 mg/ml
- Fusion Partners
- Spleen cells from immunized BALB/c mice were fused with cells of the mouse NS1 myeloma cell line.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | 1/50 | 1/100 | |
| Immunofluorescence | |||
| Immunohistology - Frozen | |||
| Immunohistology - Paraffin | |||
| Immunoprecipitation | |||
| Western Blotting |
- Flow Cytometry
- Use 10ul of the suggested working dilution to label 106 cells in 100ul.
- Immunohistology
- This product does not require protein digestion pre-treatment of paraffin embedded sections.
This product does not require antigen retrieval using heat treatment prior to staining of paraffin embedded sections.
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG1 Negative Control | MCA1209 | F | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG1 Negative Control | ||||||
Source Reference
-
Brideau, R.J. et al. (1980) Two subsets of rat T lymphocytes defined with monoclonal antibodies.
Eur J Immunol. 10 (8): 609-15.
References for CD8 Alpha antibody
-
Thomas, M.L. & Green, J.R. (1983) Molecular nature of the W3/25 and MRC OX-8 marker antigens for rat T lymphocytes: comparisons with mouse and human antigens.
Eur J Immunol. 13 (10): 855-8. -
Bukovský A et al. (1984) Association of some cell surface antigens of lymphoid cells and cell surface differentiation antigens with early rat pregnancy.
Immunology. 52 (4): 631-40. -
Torres-Nagel, N. et al. (1992) Differential thymus dependence of rat CD8 isoform expression.
Eur J Immunol. 22 (11): 2841-8. -
Whiteland, J.L. et al. (1995) Immunohistochemical detection of T-cell subsets and other leukocytes in paraffin-embedded rat and mouse tissues with monoclonal antibodies.
J Histochem Cytochem. 43 (3): 313-20. -
Mitnacht, R. et al. (1998) Opposite CD4/CD8 lineage decisions of CD4+8+ mouse and rat thymocytes to equivalent triggering signals: correlation with thymic expression of a truncated CD8 alpha chain in mice but not rats.
J Immunol. 160 (2): 700-7. -
Popov, I. et al. (2001) The effect of an anti-HLA-B27 immune response on CTL recognition of Chlamydia.
J Immunol. 167 (6): 3375-82. -
Hashimoto, Y. et al. (2003) Two major histocompatibility complex class I-restricted epitopes of the Borna disease virus p10 protein identified by cytotoxic T lymphocytes induced by DNA-based immunization.
J Virol. 77: 6076-81. -
Bradl, M. et al. (2005) Complementary contribution of CD4 and CD8 T lymphocytes to T-cell infiltration of the intact and the degenerative spinal cord.
Am J Pathol. 166: 1441-50.
View The Latest Product References
-
Latta, M. et al. (2007) CXCR6 is expressed on T cells in both T helper type 1 (Th1) inflammation and allergen-induced Th2 lung inflammation but is only a weak mediator of chemotaxis.
Immunology. 121: 555-64 -
King, G.D. et al. (2008) Flt3L in combination with HSV1-TK-mediated gene therapy reverses brain tumor-induced behavioral deficits.
Mol Ther. 16: 682-90 -
Bode, U. et al. (2008) Dendritic cell subsets in lymph nodes are characterized by the specific draining area and influence the phenotype and fate of primed T cells.
Immunology. 123: 480-90. -
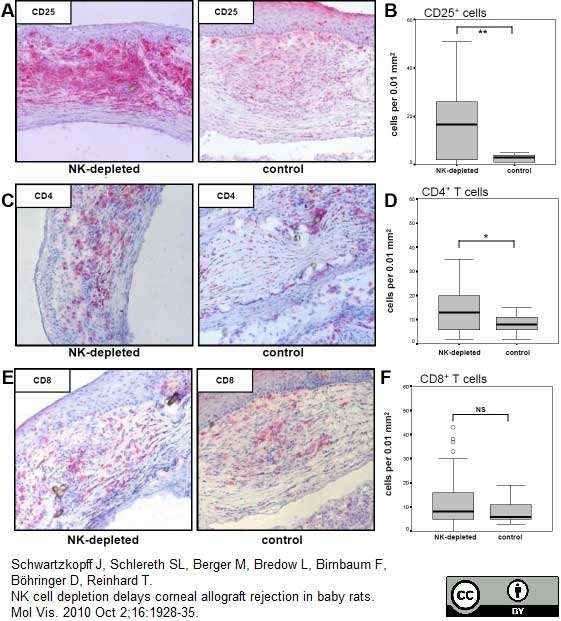
Schwartzkopff, J. et al. (2010) NK cell depletion delays corneal allograft rejection in baby rats.
Mol Vis. 16: 1928-35. -
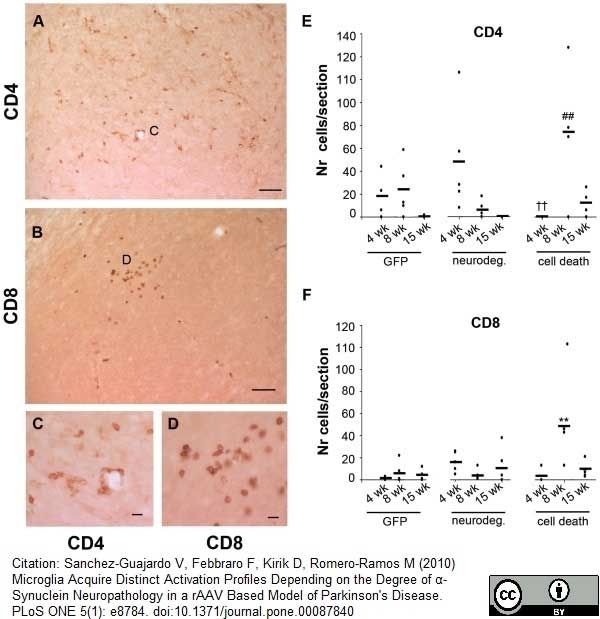
Sanchez-Guajardo, V. (2010) Microglia acquire distinct activation profiles depending on the degree of alpha-synuclein neuropathology in a rAAV based model of Parkinson's disease.
PLoS One. 5: e8784. -
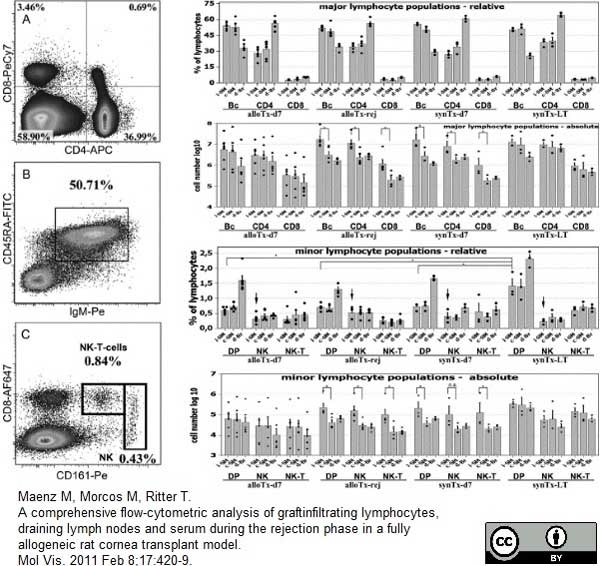
Maenz, M. et al. (2011) A comprehensive flow-cytometric analysis of graft infiltrating lymphocytes, draining lymph nodes and serum during the rejection phase in a fully allogeneic rat cornea transplant model.
Mol Vis. 2011 Feb 8;17:420-9. -
Arndt, T. et al. (2013) A variable CD3⁺ T-cell frequency in peripheral blood lymphocytes associated with type 1 diabetes mellitus development in the LEW.1AR1-iddm rat.
PLoS One. 8 (5): e64305. -
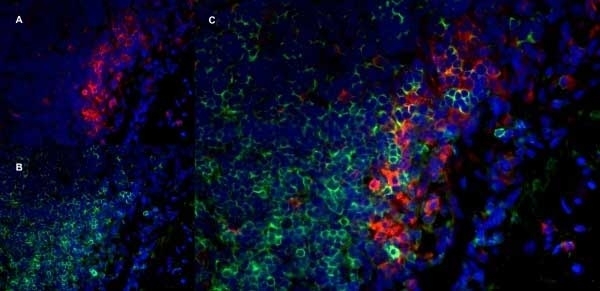
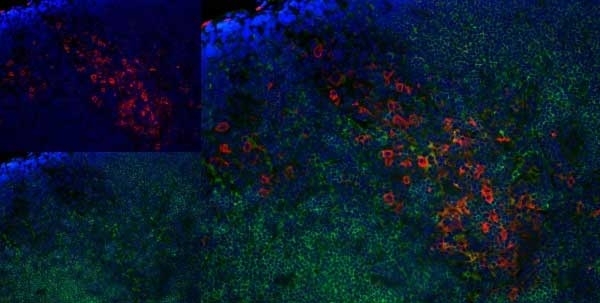
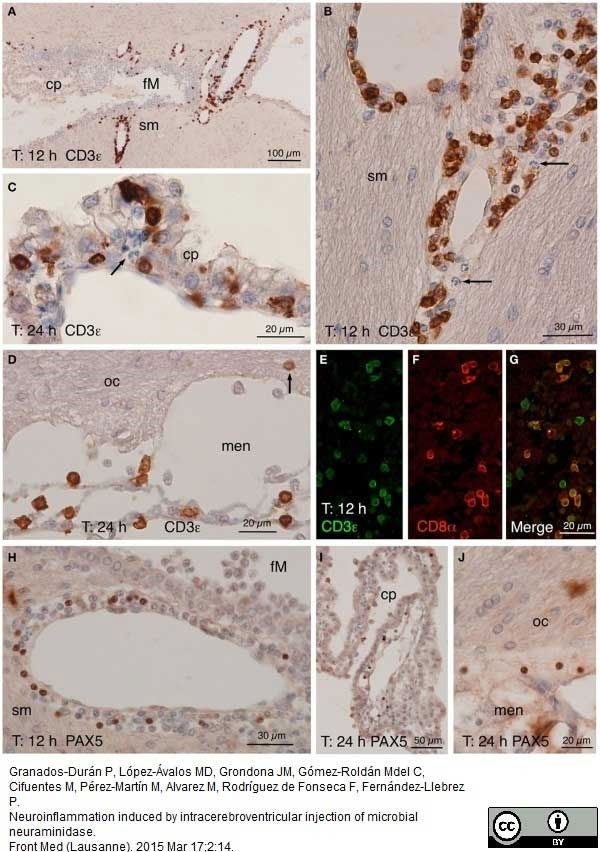
Granados-Durán P et al. (2015) Neuroinflammation induced by intracerebroventricular injection of microbial neuraminidase.
Front Med (Lausanne). 2: 14. -
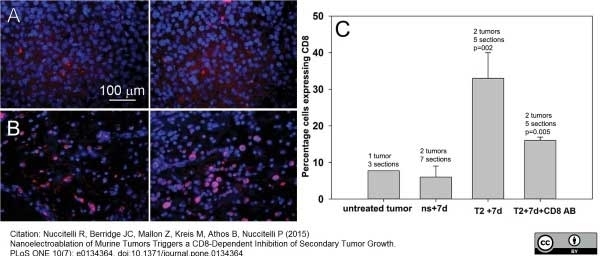
Nuccitelli R et al. (2015) Nanoelectroablation of Murine Tumors Triggers a CD8-Dependent Inhibition of Secondary Tumor Growth.
PLoS One. 10 (7): e0134364. -
Zhang, Z.M. et al. (2016) Lesional accumulation of CD8(+) cells in sciatic nerves of experimental autoimmune neuritis rats.
Neurol Sci. 37 (2): 199-203. -
Pamukcu, O. et al. (2016) Anti-inflammatory role of obestatin in autoimmune myocarditis.
Clin Exp Pharmacol Physiol. 43 (1): 47-55. -
Dabrowska, S. et al. (2019) Human bone marrow mesenchymal stem cell-derived extracellular vesicles attenuate neuroinflammation evoked by focal brain injury in rats.
J Neuroinflammation. 16 (1): 216. -
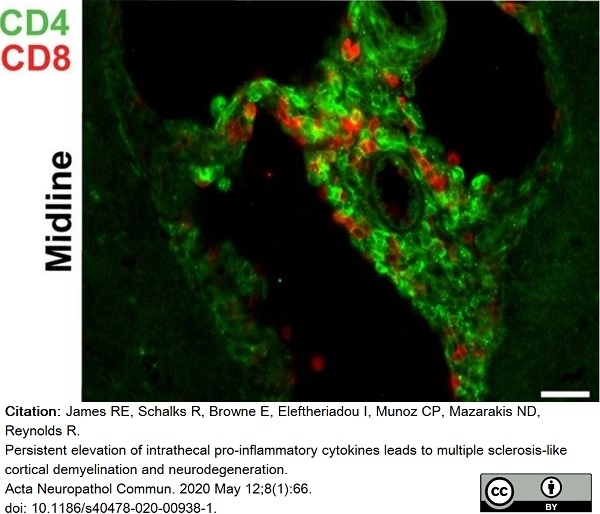
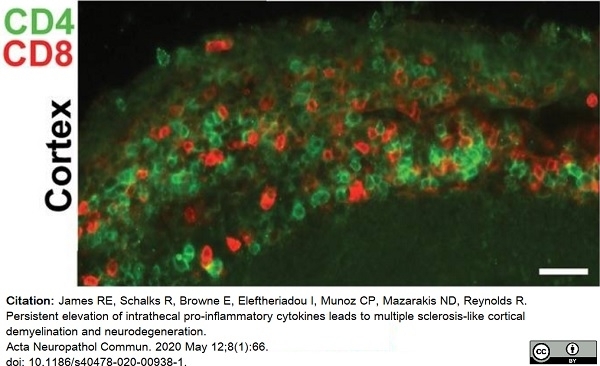
James, R.E. et al. (2020) Persistent elevation of intrathecal pro-inflammatory cytokines leads to multiple sclerosis-like cortical demyelination and neurodegeneration.
Acta Neuropathol Commun. 8 (1): 66. -
Matsuyama, S. et al. (2021) Properties of macrophages and lymphocytes appearing in rat renal fibrosis followed by repeated injection of cisplatin.
J Vet Med Sci. 83 (9): 1435-42. -
Dabrowska, S. et al. (2021) Neuroinflammation evoked by brain injury in a rat model of lacunar infarct.
Exp Neurol. 336: 113531. -
Schmiedl, A. et al. (2021) Lung development and immune status under chronic LPS exposure in rat pups with and without CD26/DPP4 deficiency.
Cell Tissue Res. 386 (3): 617-36. -
Zakerkish, F. et al. (2021) Differential effects of the immunosuppressive calcineurin inhibitors cyclosporine-A and tacrolimus on ovulation in a murine model.
Hum Reprod Open. 2021 (2): hoab012. -
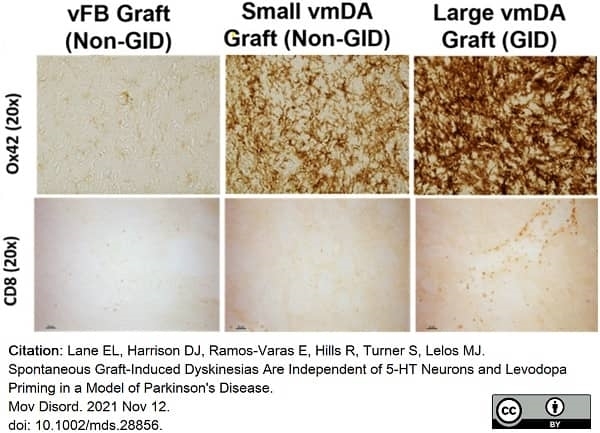
Lane, E.L. et al. (2022) Spontaneous Graft-Induced Dyskinesias Are Independent of 5-HT Neurons and Levodopa Priming in a Model of Parkinson's Disease.
Mov Disord. 37 (3): 613-9. -
Silva, B.A. et al. (2022) Understanding the role of the blood brain barrier and peripheral inflammation on behavior and pathology on ongoing confined cortical lesions.
Mult Scler Relat Disord. 57: 103346. -
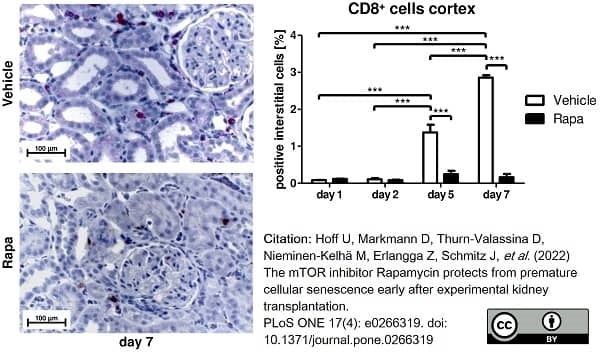
Hoff, U. et al. (2022) The mTOR inhibitor Rapamycin protects from premature cellular senescence early after experimental kidney transplantation.
PLoS One. 17 (4): e0266319. -
Gad, R.A. et al. (2022) Mitigating effects of Passiflora incarnata. on oxidative stress and neuroinflammation in case of pilocarpine-Induced status epilepticus model
J King Saud Uni - Science. 34 (3): 101886. -
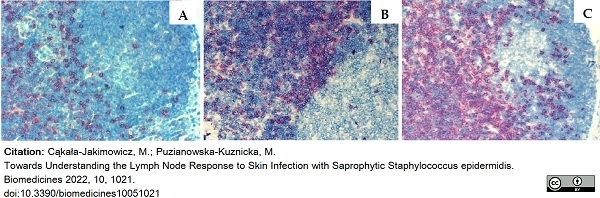
Cąkała-Jakimowicz, M. & Puzianowska-Kuznicka, M. (2022) Towards Understanding the Lymph Node Response to Skin Infection with Saprophytic Staphylococcus epidermidis.
Biomedicines. 10 (5): 1021. -
Du, K. et al. (2023) Pathogenesis of selective damage of granule cell layer in cerebellum of rats exposed to methylmercury
J Toxicol Sci 48 (7): 429-39. -
Santos Filho, L.E.D. et al. (2023) Dietary Soy Isoflavones Prevent Metabolic Disturbs Associated with a Deleterious Combination of Obesity and Menopause.
J Med Food. 26 (2): 104-13.
- RRID
- AB_321476
- UniProt
- P07725
- Entrez Gene
- Cd8a
- GO Terms
- GO:0016021 integral to membrane
- GO:0006954 inflammatory response
- GO:0019901 protein kinase binding
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Rat ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up