CD4 antibody | W3/25























Mouse anti Rat CD4 (Domain 1)
- Product Type
- Monoclonal Antibody
- Clone
- W3/25
- Isotype
- IgG1
- Specificity
- CD4
- Region
- (DOMAIN 1)
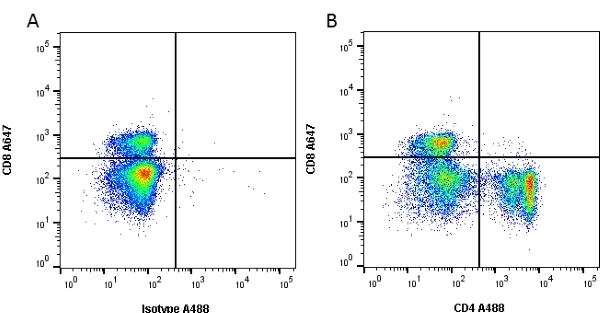
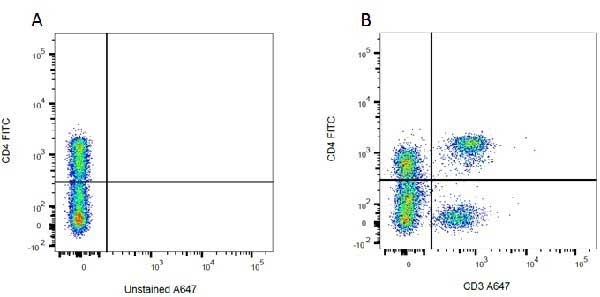
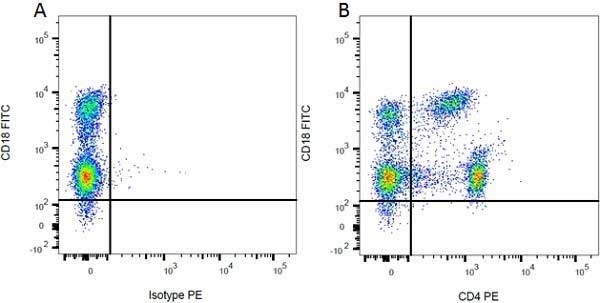
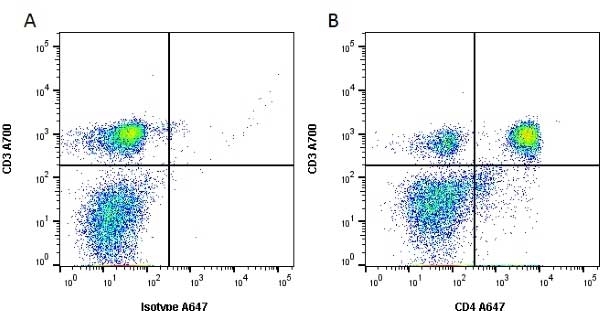
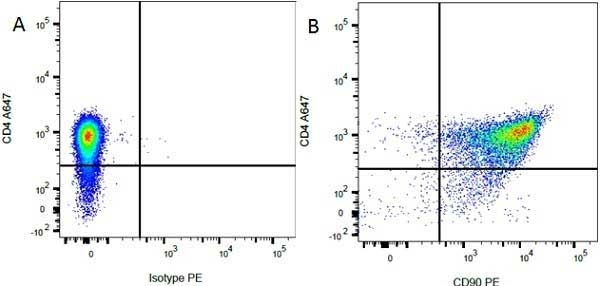
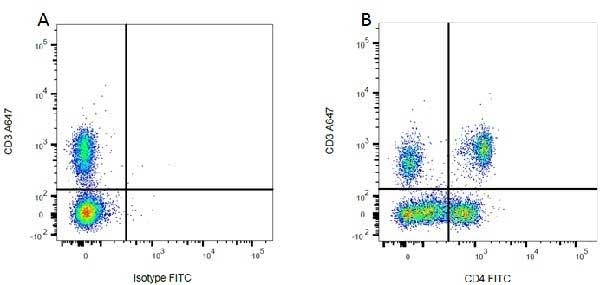
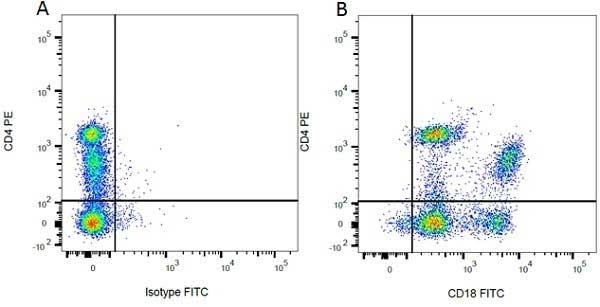
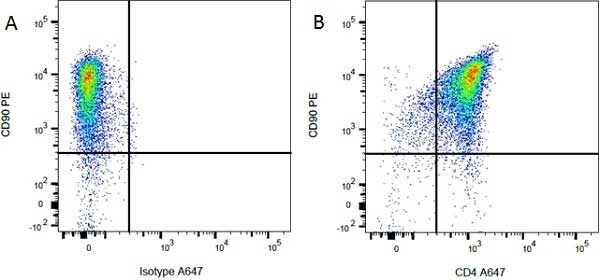
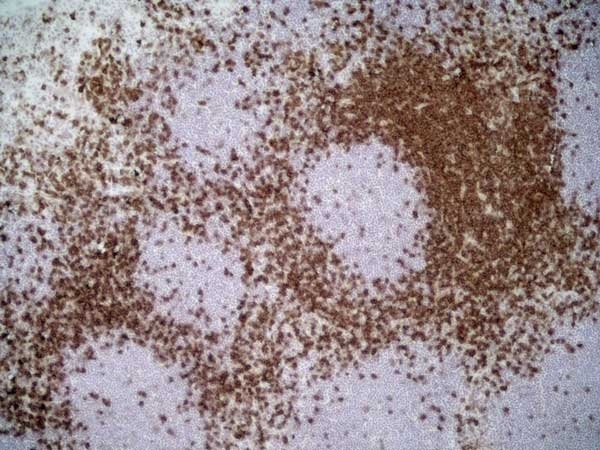
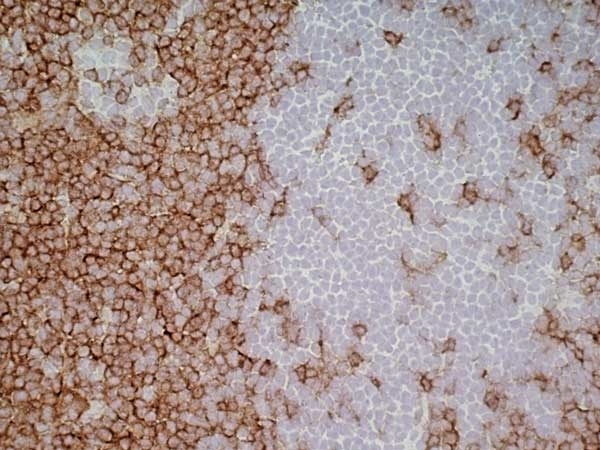
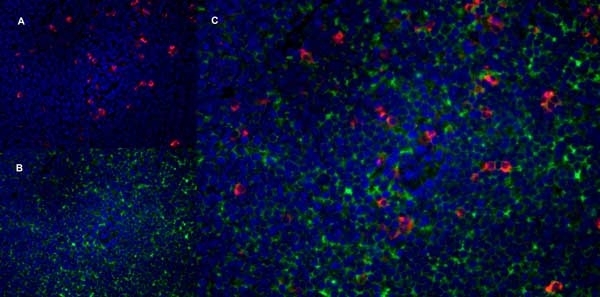
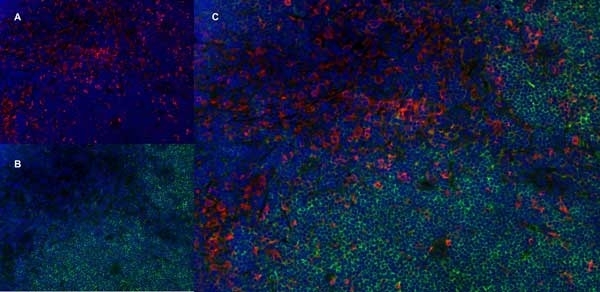
| Mouse anti Rat CD4 antibody, clone W3/25 recognizes the rat CD4 cell surface glycoprotein, a ~55 kDa molecule expressed by helper T cells and weakly by monocytes. This antibody inhibits proliferation and IL-2 production in the MLR reaction. Mouse anti Rat CD4 antibody, clone W3/25 has been described reacting with paraffin-embedded material following PLP fixation (periodate-lysine-paraformaldehyde) (Whiteland et al. 1995). Mouse anti Rat CD4 antibody, clone W3/25 is routinely tested in flow cytometry on rat splenocytes. |
- Target Species
- Rat
- Product Form
- Purified IgG - liquid
- Preparation
- MCA55R: Purified IgG prepared by affinity chromatography on Protein G from tissue culture supernatant
- MCA55GA, MCA55G: Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
0.09% Sodium Azide - Carrier Free
- Yes
- Immunogen
- Rat Thymocyte Membrane Glycoproteins.
- Approx. Protein Concentrations
- IgG concentration 1.0 mg/ml
- Fusion Partners
- Spleen cells from immunized BALB/c mouse were fused with cells of the mouse NS-1 myeloma cell line.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | 1/100 | 1/200 | |
| Immunofluorescence | |||
| Immunohistology - Frozen | |||
| Immunohistology - Paraffin 1 |
- 1PLP fixation is recommended for optimal results, see Whiteland et al. for details
- Flow Cytometry
- Use 10ul of the suggested working dilution to label 106 cells in 100ul.
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG1 Negative Control | MCA1209 | F | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG1 Negative Control | ||||||
References for CD4 antibody
-
Williams, A.F. et al. (1977) Analysis of cell surfaces by xenogeneic myeloma-hybrid antibodies: differentiation antigens of rat lymphocytes.
Cell. 12 (3): 663-73. -
Barclay, A.N. (1981) The localization of populations of lymphocytes defined by monoclonal antibodies in rat lymphoid tissues.
Immunology. 42 (4): 593-600. -
Pelegrí, C. et al. (1995) Immunohistochemical changes in synovial tissue during the course of adjuvant arthritis.
J Rheumatol. 22 (1): 124-32. -
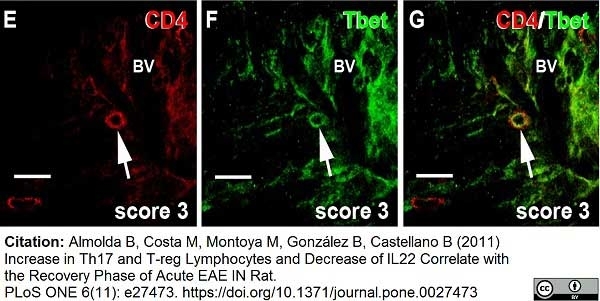
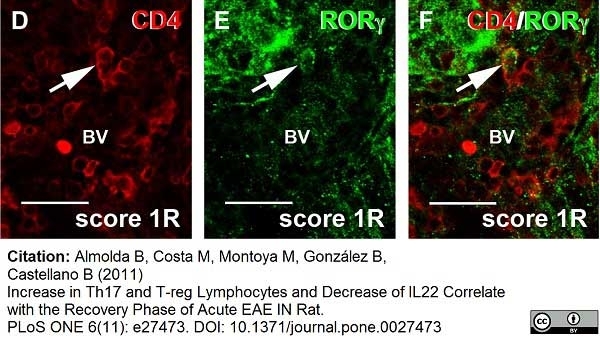
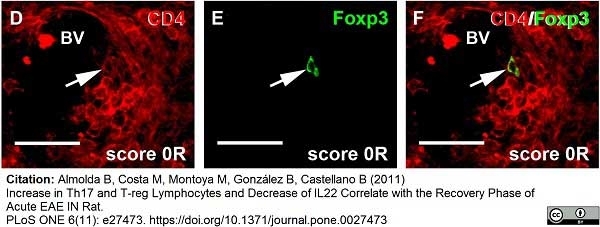
Whiteland, J.L. et al. (1995) Immunohistochemical detection of T-cell subsets and other leukocytes in paraffin-embedded rat and mouse tissues with monoclonal antibodies.
J Histochem Cytochem. 43 (3): 313-20. -
Bjersing, J.L. et al. (2002) Loss of ileal IgA+ plasma cells and of CD4+ lymphocytes in ileal Peyer's patches of vitamin A deficient rats.
Clin Exp Immunol. 130 (3): 404-8. -
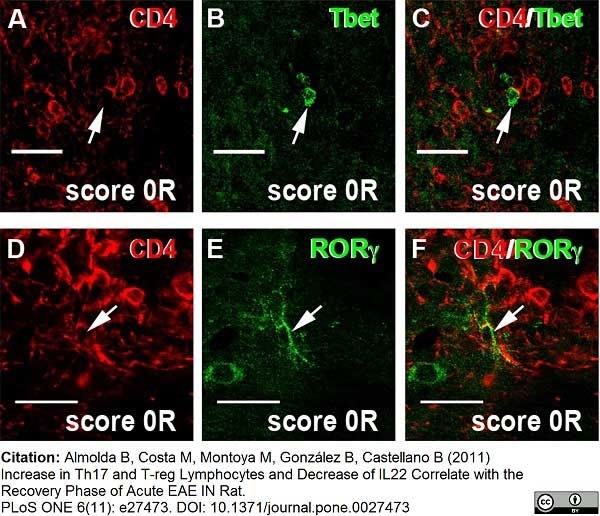
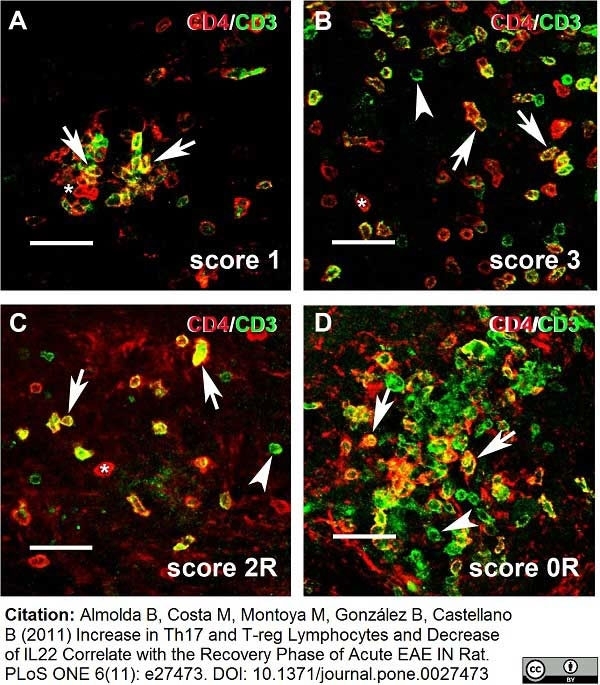
Hofmann, N. et al. (2002) Increased expression of ICAM-1, VCAM-1, MCP-1, and MIP-1 alpha by spinal perivascular macrophages during experimental allergic encephalomyelitis in rats.
BMC Immunol. 3: 11. -
Banerjee, S. et al. (2003) Development of organised conjunctival leucocyte aggregates after corneal transplantation in rats.
Br J Ophthalmol. 87 (12): 1515-22. -
Ishizuka, S. et al. (2004) Fermentable dietary fiber potentiates the localization of immune cells in the rat large intestinal crypts.
Exp Biol Med (Maywood). 229 (9): 876-84.
View The Latest Product References
-
Latta, M. et al. (2007) CXCR6 is expressed on T cells in both T helper type 1 (Th1) inflammation and allergen-induced Th2 lung inflammation but is only a weak mediator of chemotaxis.
Immunology. 121 (4): 555-64. -
Bode, U. et al. (2008) Dendritic cell subsets in lymph nodes are characterized by the specific draining area and influence the phenotype and fate of primed T cells.
Immunology. 123 (4): 480-90. -
Zilka, N. et al. (2009) Human misfolded truncated tau protein promotes activation of microglia and leukocyte infiltration in the transgenic rat model of tauopathy.
J Neuroimmunol. 209 (1-2): 16-25. -
Schwartzkopff, J. et al. (2010) NK cell depletion delays corneal allograft rejection in baby rats.
Mol Vis. 16: 1928-35. -
Schmiedl, A. et al. (2021) Lung development and immune status under chronic LPS exposure in rat pups with and without CD26/DPP4 deficiency.
Cell Tissue Res. Oct 04 [Epub ahead of print]. -
Matsuyama, S. et al. (2021) Properties of macrophages and lymphocytes appearing in rat renal fibrosis followed by repeated injection of cisplatin.
J Vet Med Sci. 83 (9): 1435-42. -
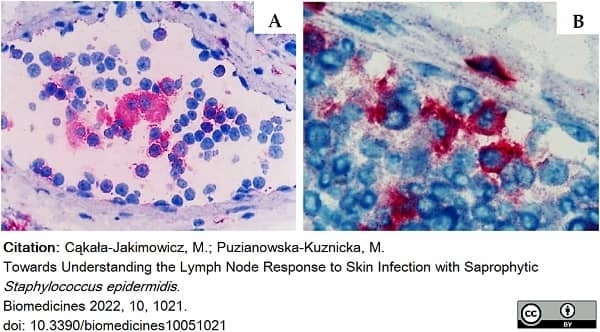
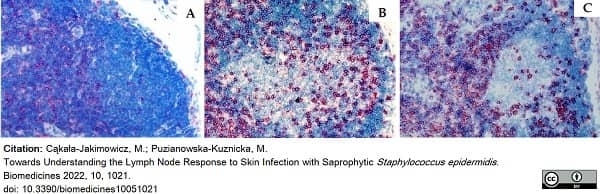
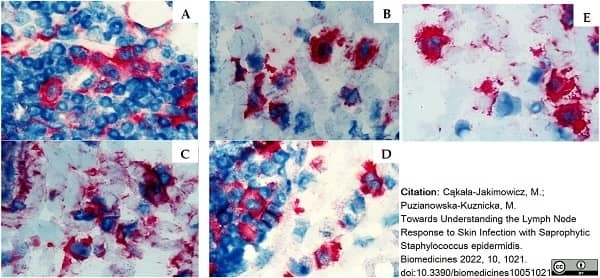
Cąkała-Jakimowicz, M. & Puzianowska-Kuznicka, M. (2022) Towards Understanding the Lymph Node Response to Skin Infection with Saprophytic Staphylococcus epidermidis..
Biomedicines. 10 (5): 1021.
- RRID
- AB_321377
- UniProt
- P05540
- Entrez Gene
- Cd4
- GO Terms
- GO:0007155 cell adhesion
- GO:0016021 integral to membrane
- GO:0006955 immune response
- GO:0019901 protein kinase binding
- GO:0045058 T cell selection
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Rat ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up