SLA Class II DR antibody | 2E9/13



Mouse anti Pig SLA Class II DR
- Product Type
- Monoclonal Antibody
- Clone
- 2E9/13
- Isotype
- IgG2b
- Specificity
- SLA Class II DR
| Mouse anti Pig SLA Class II DR antibody, clone 2E9/13 recognizes SLA DR molecules which are expressed on all B cells, antigen presenting cells and on certain subsets of resting and activated T cells. Mouse anti Pig SLA Class II DR antibody, clone 289/13 reacts with lymphocytes from all outbred and miniature pigs so far tested, suggesting that it recognizes a monomorphic determinant of porcine SLA DR. The major histocompatibility complex (MHC) is a cluster of genes that are important in the immune response to infections. In pigs, this is referred to as the swine leukocyte antigen (SLA) region. There are 3 major MHC class II proteins encoded by the SLA which are SLA DP, SLA DQ and SLA DR. Mouse anti pig SLA class II DR, clone 2E9/13 immunoprecipitates a heterodimer composed of two polypeptides of ~28 and ~35 kDa from NP-40 extracts of biotin surface-labeled porcine peripheral blood mononuclear cells. Mouse anti Pig SLA Class II DR antibody, clone 289/13 is reported to inhibit the mixed lymphocyte reaction and T cell stimulation induced by African swine fever virus and staphylococcal enterotoxin B (Bullido et al. 1997). |
- Target Species
- Pig
- Species Cross-Reactivity
-
Target Species Cross Reactivity Bovine - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% sodium azide (NaN3)
- Carrier Free
- Yes
- Immunogen
- Porcine monocytes.
- Approx. Protein Concentrations
- IgG concentration 1.0 mg/ml
- Fusion Partners
- Spleen cells from immunized BALB/c mice were fused with cells of the mouse X63-Ag.8.653 myeloma cell line.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
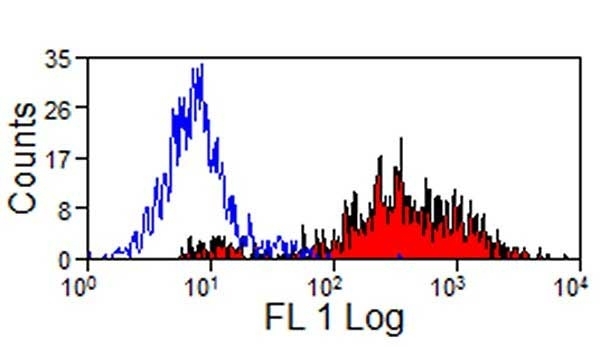
| Flow Cytometry | 1/25 | 1/200 | |
| Immunohistology - Frozen | |||
| Immunoprecipitation |
- Flow Cytometry
- Use 10μl of the suggested working dilution to 1x106 cells in 100μl
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG2b Negative Control | MCA691 | F | 100 Tests |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG2b Negative Control | ||||||
References for SLA Class II DR antibody
-
Bullido, R. et al. (1997) Characterization of five monoclonal antibodies specific for swine class II major histocompatibility antigens and crossreactivity studies with leukocytes of domestic animals.
Dev Comp Immunol. 21 (3): 311-22. -
Jeong, H.J. et al. (2010) Comparative measurement of cell-mediated immune responses of swine to the M and N proteins of porcine reproductive and respiratory syndrome virus.
Clin Vaccine Immunol. 17: 503-12. -
Ding, Q. et al. (2011) Human PD-L1-overexpressing porcine vascular endothelial cells induce functionally suppressive human CD4+CD25hiFoxp3+ Treg cells.
J Leukoc Biol. 90 (1): 77-86. -
Thierry, A. et al. (2012) Identification of invariant natural killer T cells in porcine peripheral blood.
Vet Immunol Immunopathol. 149 (3-4): 272-9. -
Iwase H et al. (2015) Initial in vivo experience of pig artery patch transplantation in baboons using mutant MHC (CIITA-DN) pigs.
Transpl Immunol. 32 (2): 99-108. -
Zanotti, C. et al. (2015) Differential Biological Activities of Swine Interferon-α Subtypes.
J Interferon Cytokine Res. 35 (12): 990-1002. -
Wang, Y. et al. (2016) Genipin crosslinking reduced the immunogenicity of xenogeneic decellularized porcine whole-liver matrices through regulation of immune cell proliferation and polarization.
Sci Rep. 6: 24779. -
Gardner, D.S. et al. (2016) Remote effects of acute kidney injury in a porcine model.
Am J Physiol Renal Physiol. 310 (4): F259-71.
View The Latest Product References
-
Singleton, H. et al. (2016) Establishing Porcine Monocyte-Derived Macrophage and Dendritic Cell Systems for Studying the Interaction with PRRSV-1.
Front Microbiol. 7: 832. -
Rahe, M.C. & Murtaugh, M.P. (2017) Interleukin-21 Drives Proliferation and Differentiation of Porcine Memory B Cells into Antibody Secreting Cells.
PLoS One. 12 (1): e0171171. -
Mašek, J. et al. (2017) Multi-layered nanofibrous mucoadhesive films for buccal and sublingual administration of drug-delivery and vaccination nanoparticles - important step towards effective mucosal vaccines.
J Control Release. 249: 183-95. -
Ladowski, J.M. et al. (2018) Swine Leukocyte Antigen Class II Is a Xenoantigen.
Transplantation. 102 (2): 249-54. -
Yang, N. et al. (2018) Reduced antigen presentation capability and modified inflammatory/immunosuppressive cytokine expression of induced monocyte-derived dendritic cells from peripheral blood of piglets infected with porcine circovirus type 2.
Arch Virol. 163 (5): 1231-9. -
López, E. et al. (2019) Identification of very early inflammatory markers in a porcine myocardial infarction model.
BMC Vet Res. 15 (1): 91. -
Liu, S. et al. (2019) Endothelial IL-8 induced by porcine circovirus type 2 affects dendritic cell maturation and antigen-presenting function.
Virol J. 16 (1): 154. -
Radlowski, E.C. et al. (2021) Combination-Feeding Causes Differences in Aspects of Systemic and Mucosal Immune Cell Phenotypes and Functions Compared to Exclusive Sow-Rearing or Formula-Feeding in Piglets.
Nutrients. 13(4):1097. -
Arenal, Á. et al. (2022) Effects of Cardiac Stem Cell on Postinfarction Arrhythmogenic Substrate.
Int J Mol Sci. 23 (24): 16211. -
Franzoni, G. et al. (2022) Analyses of the Impact of Immunosuppressive Cytokines on Porcine Macrophage Responses and Susceptibility to Infection to African Swine Fever Viruses.
Pathogens. 11 (2): 166. -
Haach, V. et al. (2023) A polyvalent virosomal influenza vaccine induces broad cellular and humoral immunity in pigs.
Virol J. 20 (1): 181. -
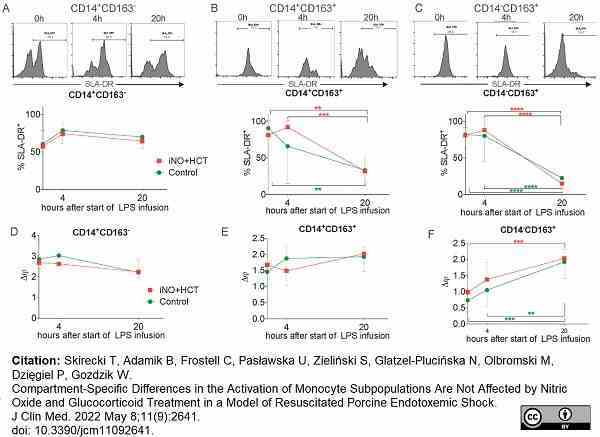
Skirecki, T. et al. (2022) Compartment-Specific Differences in the Activation of Monocyte Subpopulations Are Not Affected by Nitric Oxide and Glucocorticoid Treatment in a Model of Resuscitated Porcine Endotoxemic Shock.
J Clin Med. 11 (9): 2641.
Further Reading
-
Piriou-Guzylack, L. (2008) Membrane markers of the immune cells in swine: an update.
Vet Res. 39: 54. -
Rayat GR et al. (2016) First update of the International Xenotransplantation Association consensus statement on conditions for undertaking clinical trials of porcine islet products in type 1 diabetes - Chapter 3: Porcine islet product manufacturing and release testing criteria.
Xenotransplantation. 23 (1): 38-45.
- UniProt
- Q85ZW4
MCA2314GA
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Pig ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up