IgM antibody | K52 1C3







Mouse anti Pig IgM
- Product Type
- Monoclonal Antibody
- Clone
- K52 1C3
- Isotype
- IgG1
- Specificity
- IgM
| Mouse anti Pig IgM antibody, clone K52 1C3 recognizes porcine IgM heavy chain. No cross-reactivity with porcine IgA and IgG is seen in ELISA. |
- Target Species
- Pig
- Product Form
- Purified IgG - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% Sodium Azide (NaN3)
- Carrier Free
- Yes
- Immunogen
- Porcine IgM
- Approx. Protein Concentrations
- IgG concentration 1.0 mg/ml
- Fusion Partners
- Spleen cells of immunised mice were fused with cells of the P3 - X63 - Ag 8.653 mouse myeloma line.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
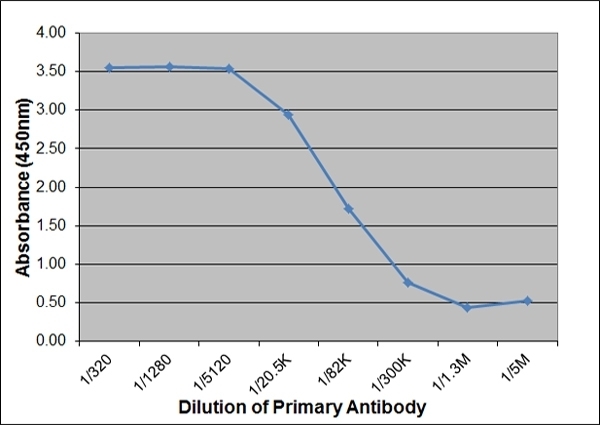
| ELISA | 1/5000 | 1/100,000 | |
| Flow Cytometry | |||
| Immunohistology - Frozen |
- Flow Cytometry
- Use 10ul of the suggested working dilution to label 1x106 cells in 100ul
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG1 Negative Control | MCA928 | F | 100 Tests |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG1 Negative Control | ||||||
References for IgM antibody
-
Andersen, J.K. et al. (1999) Systematic characterization of porcine ileal Peyer's patch, I. apoptosis-sensitive immature B cells are the predominant cell type.
Immunology. 98 (4): 612-21. -
Baltes, N. et al. (2001) Actinobacillus pleuropneumoniae iron transport and urease activity: effects on bacterial virulence and host immune response.
Infect Immun. 69 (1): 472-8. -
Leitão, A. et al. (2001) The non-haemadsorbing African swine fever virus isolate ASFV/NH/P68 provides a model for defining the protective anti-virus immune response.
J Gen Virol. 82 (Pt 3): 513-23. -
Bailey, M. (2004) Effects of infection with transmissible gastroenteritis virus on concomitant immune responses to dietary and injected antigens.
Clin Diagn Lab Immunol. 11: 337-43. -
Hamano, M. et al. (2007) Detection of antibodies to Japanese encephalitis virus in the wild boars in Hiroshima prefecture, Japan.
Epidemiol Infect. 135: 974-7. -
Stepanova, H. et al. (2011) Association of attenuated mutants of Salmonella enterica serovar Enteritidis with porcine peripheral blood leukocytes.
FEMS Microbiol Lett. 321: 37-42. -
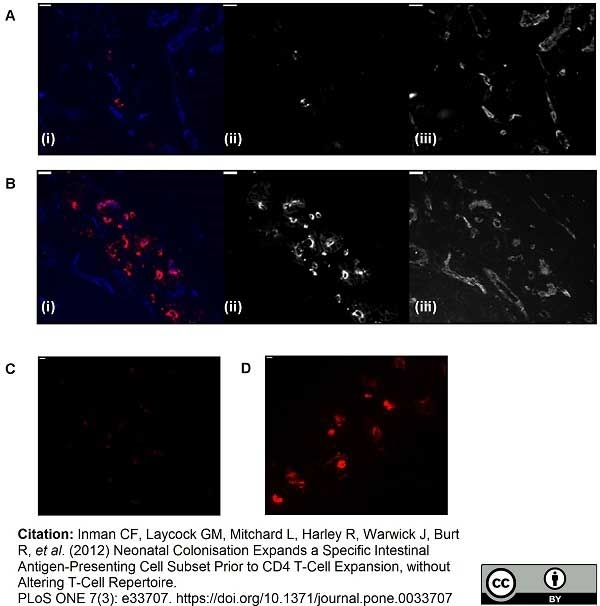
Laycock, G. et al. (2012) A defined intestinal colonization microbiota for gnotobiotic pigs.
Vet Immunol Immunopathol. 149: 216-24. -
Lewis MC et al. (2013) Dietary supplementation with Bifidobacterium lactis NCC2818 from weaning reduces local immunoglobulin production in lymphoid-associated tissues but increases systemic antibodies in healthy neonates.
Br J Nutr. 110: 1243-52.
View The Latest Product References
-
Chen, F. et al. (2015) Generation of B Cell-Deficient Pigs by Highly Efficient CRISPR/Cas9-Mediated Gene Targeting.
J Genet Genomics. 42 (8): 437-44. -
Seele, J. et al. (2015) The immunoglobulin M-degrading enzyme of Streptococcus suis, IdeSsuis, is a highly protective antigen against serotype 2.
Vaccine. 33 (19): 2207-12. -
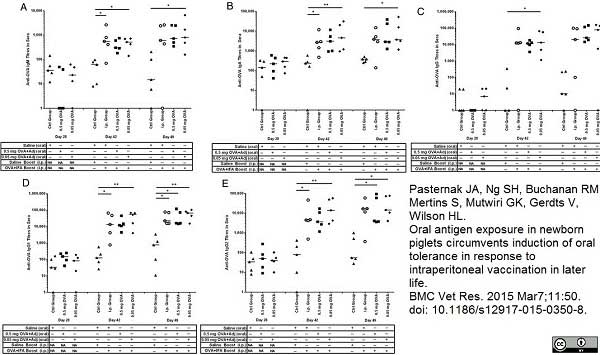
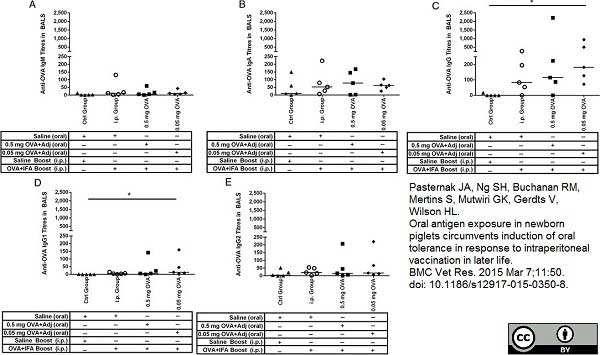
Pasternak, J.A. et al. (2015) Oral antigen exposure in newborn piglets circumvents induction of oral tolerance in response to intraperitoneal vaccination in later life.
BMC Vet Res. 11: 350. -
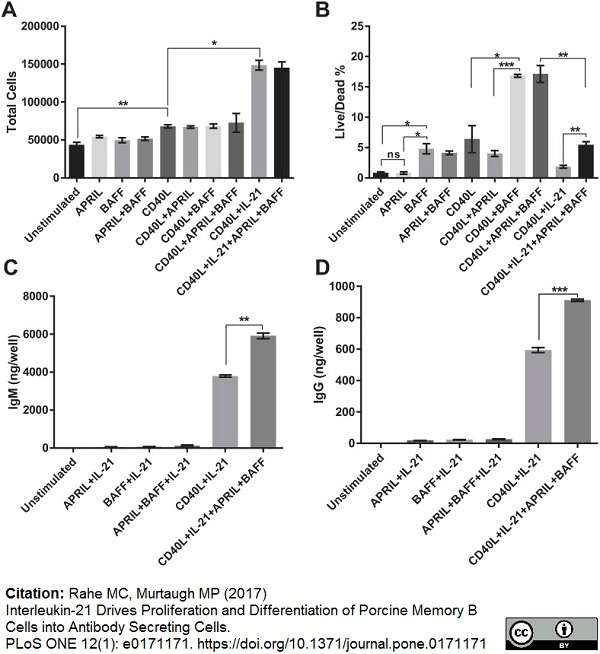
Rahe, M.C. & Murtaugh, M.P. (2017) Interleukin-21 Drives Proliferation and Differentiation of Porcine Memory B Cells into Antibody Secreting Cells.
PLoS One. 12 (1): e0171171. -
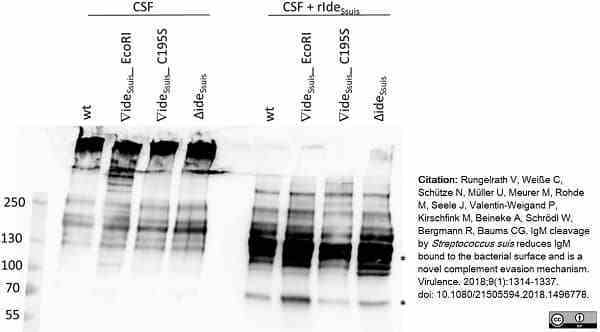
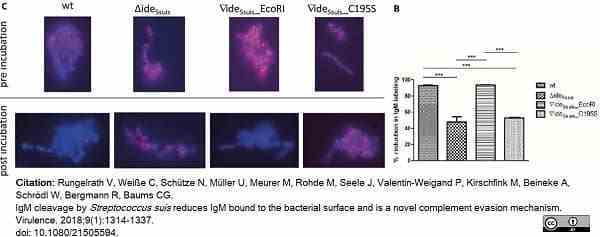
Rungelrath, V. et al. (2018) IgM cleavage by Streptococcus suis. reduces IgM bound to the bacterial surface and is a novel complement evasion mechanism.
Virulence. 9 (1): 1314-1337. -
Buermann, A. et al. (2018) Pigs expressing the human inhibitory ligand PD-L1 (CD 274) provide a new source of xenogeneic cells and tissues with low immunogenic properties.
Xenotransplantation. 25 (5): e12387. -
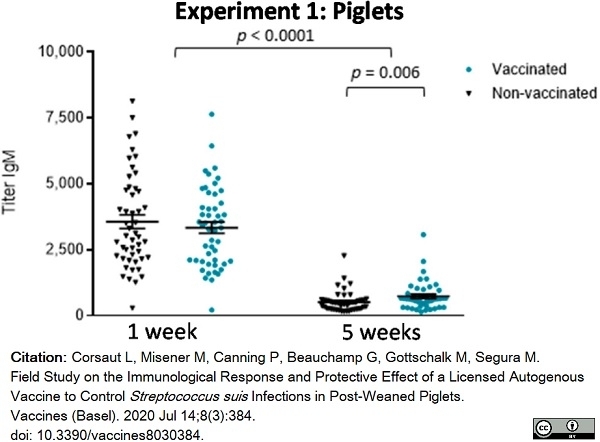
Corsaut, L. et al. (2020) Field Study on the Immunological Response and Protective Effect of a Licensed Autogenous Vaccine to Control Streptococcus suis Infections in Post-Weaned Piglets.
Vaccines (Basel). 8 (3): 384.
MCA637GA
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Pig ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up