IgG2 antibody | K68 Ig2


Mouse anti Pig IgG2
- Product Type
- Monoclonal Antibody
- Clone
- K68 Ig2
- Isotype
- IgG1
- Specificity
- IgG2
| Mouse anti Pig IgG2, clone K68 Ig2 recognizes porcine IgG2 and shows no cross-reactivity with IgG1, IgA or IgM. IgG2 (IgG2a and IgG2b), along with IgG1, IgG3 and IgG4 comprise the major known subclasses of IgG in swine. Combined, the various subclases of IgG comprise approximately 85% of immunoglobulin in porcine serum. Porcine IgG2 and Ifn-γ levels are significantly elevated in parasite infections by worm species such as Schistosoma japonicum (Tian, F. et. al. 2010). Antibodies recognizing porcine immunoglobulins such as clone K68 Ig2 have potential use in porcine vaccine development assays (Rodríguez-Calvo et. al. 2010). Clone K68 Ig2 forms part of a range of monoclonal antibodies specific for porcine immunoglobulins and immunoglobulin subclasses available from Bio-Rad. |
- Target Species
- Pig
- Product Form
- Purified IgG - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% Sodium Azide (NaN3)
- Carrier Free
- Yes
- Immunogen
- Porcine IgG2
- Approx. Protein Concentrations
- IgG concentration 1.0 mg/ml
- Fusion Partners
- Spleen cells from immunised mice were fused with cells of the mouse P3-X63-Ag8.653 myeloma cell line.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
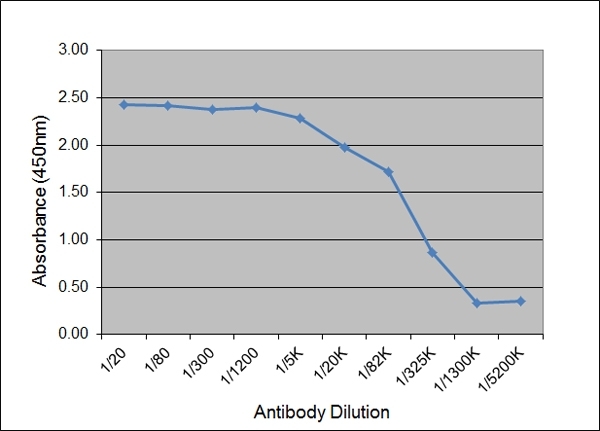
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| ELISA | 1/1000 | 1/50,000 |
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse anti Pig IgG1 | MCA635GA | E | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse anti Pig IgG1 | ||||||
| Mouse anti Pig IgA | MCA638GA | C E F | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse anti Pig IgA | ||||||
References for IgG2 antibody
-
Rivera, E. et al. (2003) Ginseng extract in aluminium hydroxide adjuvanted vaccines improves the antibody response of pigs to porcine parvovirus and Erysipelothrix rhusiopathiae.
Vet Immunol Immunopathol. 91 (1): 19-27. -
Nejsum, P. et al. (2009) Population dynamics of Trichuris suis in trickle-infected pigs.
Parasitology. 136:691-7. -
Tian, F. et al. (2010) Immune Events Associated with High Level Protection against Schistosoma japonicum Infection in Pigs Immunized with UV-Attenuated Cercariae.
PLoS One. 2010 Oct 15;5(10):e13408. -
Bailey, M. et al. (2004) Effects of infection with transmissible gastroenteritis virus on concomitant immune responses to dietary and injected antigens
Clin Diagn Lab Immunol. 11:337-43. -
Lin, D. et al. (2011) Multiple vaccinations with UV- attenuated cercariae in pig enhance protective immunity against Schistosoma japonicum infection as compared to single vaccination.
Parasit Vectors. 4:103. -
Lefevre, E.A. et al. (2012) Immune responses in pigs vaccinated with adjuvanted and non-adjuvanted A(H1N1)pdm/09 influenza vaccines used in human immunization programmes.
PLoS One. 7: e32400. -
Baums CG et al. (2010) Immunogenicity of an autogenous Streptococcus suis bacterin in preparturient sows and their piglets in relation to protection after weaning.
Clin Vaccine Immunol. 17 (10): 1589-97. -
Jayashi, C.M. et al. (2012) Characterisation of antibody responses in pigs induced by recombinant oncosphere antigens from Taenia solium.
Vaccine. 30 (52): 7475-80.
View The Latest Product References
-
Rodríguez-Calvo, T. et al. (2010) New vaccine design based on defective genomes that combines features of attenuated and inactivated vaccines.
PLoS One. 5: e10414. -
Schmied, J. et al. (2012) Effect of Heat-Killed Escherichia coli, Lipopolysaccharide, and Muramyl Dipeptide Treatments on the Immune Response Phenotype and Allergy in Neonatal Pigs Sensitized to the Egg White Protein Ovomucoid.
Clin Vaccine Immunol. 19:1955-64. -
Weber, T.E. and Spurlock, M.E. (2004) Leptin alters antibody isotype in the pig in vivo, but does not regulate cytokine expression or stimulate STAT3 signaling in peripheral blood monocytes in vitro.
J Anim Sci. 82:1630-40. -
Pasternak, J.A. et al. (2015) Oral antigen exposure in newborn piglets circumvents induction of oral tolerance in response to intraperitoneal vaccination in later life.
BMC Vet Res. 11 (1): 350. -
Blanco E et al. (2016) Full protection of swine against foot-and-mouth disease by a bivalent B-cell epitope dendrimer peptide.
Antiviral Res. Mar 5. pii: S0166-3542(16)30132-2. [Epub ahead of print] -
Goyette-Desjardins G et al. (2016) Protection against Streptococcus suis Serotype 2 Infection Using a Capsular Polysaccharide Glycoconjugate Vaccine.
Infect Immun. 84 (7): 2059-75. -
Williams, A.R. et al. (2017) Dietary cinnamaldehyde enhances acquisition of specific antibodies following helminth infection in pigs.
Vet Immunol Immunopathol. 189: 43-52. -
Grodeland, G. et al. (2020) Targeting of HA to chemokine receptors induces strong and cross-reactive T cell responses after DNA vaccination in pigs.
Vaccine. 38 (6): 1280-5. -
Cañas-Arranz, R. et al. (2020) A bivalent B-cell epitope dendrimer peptide can confer long-lasting immunity in swine against foot-and-mouth disease.
Transbound Emerg Dis. 67 (4): 1614-22. -
Urbano, A.C. et al. (2023) Targeted mutagenesis of the β-strand DNA binding region of African swine fever virus histone-like protein (pA104R) impairs DNA-binding activity and antibody recognition.
Antiviral Res. : 105784.
MCA636GA
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Pig ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up