CD107a antibody | 4E9/11
Mouse anti Pig CD107a
- Product Type
- Monoclonal Antibody
- Clone
- 4E9/11
- Isotype
- IgG1
- Specificity
- CD107a
Filter by Application:
F Reset| Mouse anti Pig CD107a, clone 4E9/11 recognizes porcine CD107a, a cell surface antigen, also known as lysosomal-associated membrane protein-1 or LAMP-1. CD107a is a type 1 single pass transmembrane glycoprotein expressed on macrophages and more weakly on monocytes and granulocytes. |
- Target Species
- Pig
- Product Form
- Purified IgG - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% sodium azide (NaN3)
- Carrier Free
- Yes
- Immunogen
- Porcine alveolar macrophages.
- Approx. Protein Concentrations
- IgG concentration 1.0 mg/ml
- Fusion Partners
- Spleen cells from immunized BALB/c mice were fused with cells of the mouse X63-Ag.8.653 myeloma cell line.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry 1 | 1/25 | 1/50 | |
| Immunohistology - Frozen | |||
| Immunohistology - Paraffin 2 | |||
| Immunoprecipitation | |||
| Western Blotting |
- 1 Membrane permeabilization is required for this application. The use of Leucoperm (Product Code BUF09) is recommended for this purpose.
- 2 This product requires antigen retrieval using heat treatment prior to staining of paraffin sections.
Sodium citrate buffer pH 6.0 is recommended for this purpose.
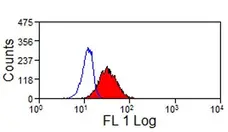
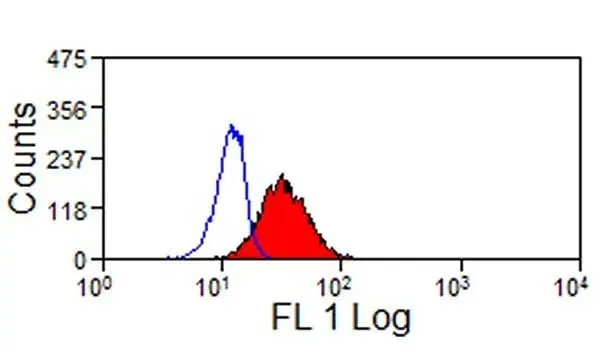
- Flow Cytometry
- Use 10μl of the suggested working dilution to label 1x106 cells in 100μl
- Histology Positive Control Tissue
- Porcine spleen
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG1 Negative Control | MCA928 | F | 100 Tests |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG1 Negative Control | ||||||
References for CD107a antibody
-
Bullido, R. et al. (1997) Monoclonal antibodies specific for porcine monocytes/macrophages: macrophage heterogeneity in the pig evidenced by the expression of surface antigens.
Tissue Antigens. 49 (4): 403-13. -
Carrillo, A. et al. (2002) Isolation and characterization of immortalized porcine aortic endothelial cell lines.
Vet Immunol Immunopathol. 89 (1-2): 91-8. -
Domenech, N. et al. (2003) Identification of porcine macrophages with monoclonal antibodies in formalin-fixed, paraffin-embedded tissues.
Vet Immunol Immunopathol. 94 (1-2): 77-81. -
Sánchez-Torres, C. et al. (2003) Expression of porcine CD163 on monocytes/macrophages correlates with permissiveness to African swine fever infection.
Arch Virol. 148 (12): 2307-23. -
Toka, F.N. et al. (2009) Natural killer cell dysfunction during acute infection with foot-and-mouth disease virus.
Clin Vaccine Immunol. 16: 1738-49. -
Toka, F.N. et al. (2009) Activation of porcine natural killer cells and lysis of foot-and-mouth disease virus infected cells.
J Interferon Cytokine Res. 29 (3): 179-92. -
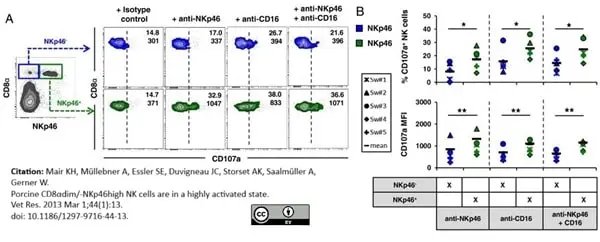
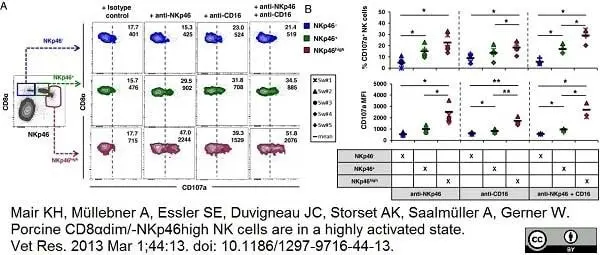
Mair, K.H. et al. (2013) Porcine CD8αdim/-NKp46high NK cells are in a highly activated state.
Vet Res. 44: 13. -
Cruz, J.L. et al. (2013) Alphacoronavirus Protein 7 Modulates Host Innate Immune Response
J Virol. 87: 9754-67.
View The Latest Product References
-
Bullers, S.J. et al. (2014) The human tissue-biomaterial interface: a role for PPARγ-dependent glucocorticoid receptor activation in regulating the CD163+ M2 macrophage phenotype.
Tissue Eng Part A. 20: 2390-401. -
van Hout, G.P. et al. (2015) Invasive surgery reduces infarct size and preserves cardiac function in a porcine model of myocardial infarction.
J Cell Mol Med. 19 (11): 2655-63. -
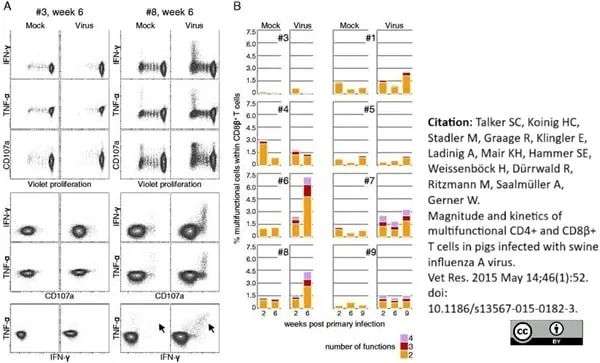
Talker, S.C. et al. (2015) Magnitude and kinetics of multifunctional CD4+ and CD8β+ T cells in pigs infected with swine influenza A virus.
Vet Res. 46: 52. -
Bongoni, A.K. et al. (2015) Porcine extrahepatic vascular endothelial asialoglycoprotein receptor 1 mediates xenogeneic platelet phagocytosis in vitro and in human-to-pig ex vivo xenoperfusion.
Transplantation. 99 (4): 693-701. -
Dash, R. et al. (2018) Dose-Dependent Cardioprotection of Moderate (32°C) Versus Mild (35°C) Therapeutic Hypothermia in Porcine Acute Myocardial Infarction.
JACC Cardiovasc Interv. 11 (2): 195-205. -
Chand, D.J. et al. (2026) Intradermal administration of a novel adjuvant cyclopolyphosphazene (CPZ-75B) in pigs induces innate immune responses comparable to poly[di(sodiumcarboxylatoethylphenoxy)phosphazene] (PCEP).
Vet Immunol Immunopathol. 292: 111061. -
Nishimura, Y. et al. (2023) Application of polyglycolic acid sheets and basic fibroblast growth factor to prevent esophageal stricture after endoscopic submucosal dissection in pigs.
J Gastroenterol. 58 (11): 1094-104. -
Álvarez, B. et al. (2023) Porcine Macrophage Markers and Populations: An Update.
Cells. 12 (16): 2103.
Further Reading
-
Piriou-Guzylack, L. (2008) Membrane markers of the immune cells in swine: an update.
Vet Res. 39: 54.
- Synonyms
- LAMP-1
MCA2315GA
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Pig ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up