CD239 antibody | BRIC221


Mouse anti Human CD239
- Product Type
- Monoclonal Antibody
- Clone
- BRIC221
- Isotype
- IgG2b
- Specificity
- CD239
| Mouse anti Human CD239 antibody, clone BRIC221 recognizes human CD239, also known as Lutheran antigen or basal cell aghesion molecule.CD239 is a 597 amino acid, ~85 kDa single pass type I membrane glycoprotein. Clone BRIC221 recognizes a monomorphic determinant expresses on both the 85 and 78 kDa Lutheran (Lu) glycoforms (El Nemer et al. 1998). BRIC 221 recognizes an epitope in the fourth extracellular domain of Lu glycoprotein (Parsons et al. 1997). Lutheran glycoprotein is a member of the immunoglobulin superfamily and was designated CD239 (B-CAM) at the 7th leucocyte typing workshop. CD239 is expressed by erythrocytes in the peripheral blood. |
- Target Species
- Human
- Species Cross-Reactivity
-
Target Species Cross Reactivity Pig - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein G from tissue culture supernatant
- Buffer Solution
- TRIS buffered glycine
- Preservative Stabilisers
- MCA1982: <0.1% sodium azide (NaN3)
- MCA1982T: <0.1% Sodium Azide (NaN3)
- Immunogen
- Human erythrocytes.
- Approx. Protein Concentrations
- IgG concentration 1.0 mg/ml
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| ELISA | |||
| Flow Cytometry | |||
| Immunofluorescence | |||
| Immunohistology - Frozen | |||
| Western Blotting 1 |
- 1Non-reducing conditions required
- Flow Cytometry
- Use 10μl of the suggested working dilution to label 106 cells in 100μl
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG2b Negative Control | MCA691 | F | 100 Tests |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG2b Negative Control | ||||||
Source Reference
-
Parsons, S.F. et al. (1997) Use of domain-deletion mutants to locate Lutheran blood group antigens to each of the five immunoglobulin superfamily domains of the Lutheran glycoprotein: elucidation of the molecular basis of the Lu(a)/Lu(b) and the Au(a)/Au(b) polymorphisms.
Blood. 89 (11): 4219-25.
References for CD239 antibody
-
Sakai, E. et al. (2007) Construction of recombinant hemagglutinin derived from the gingipain-encoding gene of Porphyromonas gingivalis, identification of its target protein on erythrocytes, and inhibition of hemagglutination by an interdomain regional peptide.
J Bacteriol. 189: 3977-86. -
Bruce, L.J. et al. (2003) A band 3-based macrocomplex of integral and peripheral proteins in the RBC membrane.
Blood. 101: 4180-8. -
Kjellgren, D. et al. (2004) Laminin isoforms in human extraocular muscles.
Invest Ophthalmol Vis Sci. 45: 4233-9. -
Vainionpää, N. et al. (2006) Laminin-10 and Lutheran blood group glycoproteins in adhesion of human endothelial cells.
Am J Physiol Cell Physiol. 290: C764-75. -
Chen, J. et al. (2009) Expression of laminin isoforms, receptors, and binding proteins unique to nucleus pulposus cells of immature intervertebral disc.
Connect Tissue Res. 50: 294-306. -
Määttä, M. et al. (2005) Differential expression of laminin isoforms in ovarian epithelial carcinomas suggesting different origin and providing tools for differential diagnosis.
J Histochem Cytochem. 53: 1293-300. -
Virtanen, I. et al. (2003) Laminin isoforms in fetal and adult human adrenal cortex.
J Clin Endocrinol Metab. 88: 4960-6. -
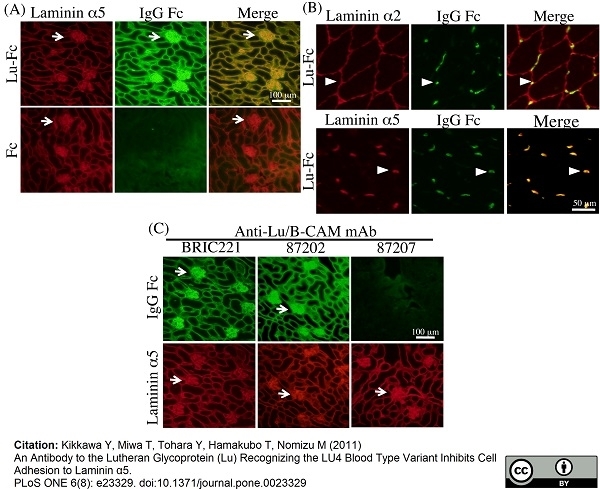
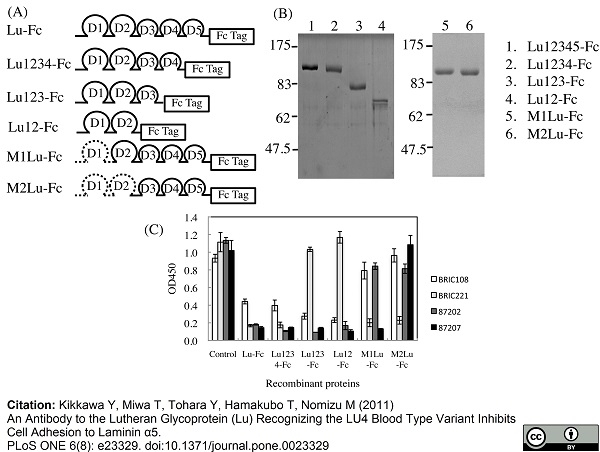
Kikkawa, Y. et al. (2011) An antibody to the lutheran glycoprotein (Lu) recognizing the LU4 blood type variant inhibits cell adhesion to laminin α5.
PLoS One. 6: e23329.
View The Latest Product References
-
Takkunen, M. et al. (2008) Epithelial-mesenchymal transition downregulates laminin alpha5 chain and upregulates laminin alpha4 chain in oral squamous carcinoma cells.
Histochem Cell Biol. 130: 509-25. -
Hasenson, S. et al. (2005) The immortalized human corneal epithelial cells adhere to laminin-10 by using Lutheran glycoproteins and integrin alpha3beta1.
Exp Eye Res. 81: 415-21. -
Liu, J.X. et al. (2011) Different impact of ALS on laminin isoforms in human extraocular muscles versus limb muscles.
Invest Ophthalmol Vis Sci. 52: 4842-52. -
Vuoristo, S. et al. (2009) Laminin isoforms in human embryonic stem cells: synthesis, receptor usage and growth support.
J Cell Mol Med. 13: 2622-33. -
Kikkawa, Y. et al. (2016) Down-regulation of cell adhesion via rho-associated protein kinase (ROCK) pathway promotes tumor cell migration on laminin-511.
Exp Cell Res. 344 (1): 76-85. -
Vainionpää N et al. (2007) Basement membrane protein distribution in LYVE-1-immunoreactive lymphatic vessels of normal tissues and ovarian carcinomas.
Cell Tissue Res. 328 (2): 317-28. -
Kikkawa Y et al. (2014) Soluble Lutheran/basal cell adhesion molecule is detectable in plasma of hepatocellular carcinoma patients and modulates cellular interaction with laminin-511in vitro.
Exp Cell Res. 328 (1): 197-206. -
Kikkawa, Y. et al. (2008) Laminin alpha 5 mediates ectopic adhesion of hepatocellular carcinoma through integrins and/or Lutheran/basal cell adhesion molecule.
Exp Cell Res. 314 (14): 2579-90. -
Kikkawa, Y. et al. (2007) The LG1-3 tandem of laminin alpha5 harbors the binding sites of Lutheran/basal cell adhesion molecule and alpha3beta1/alpha6beta1 integrins.
J Biol Chem. 282 (20): 14853-60. -
Kikkawa, Y. et al. (2013) The lutheran/basal cell adhesion molecule promotes tumor cell migration by modulating integrin-mediated cell attachment to laminin-511 protein.
J Biol Chem. 288 (43): 30990-1001. -
Enomoto-Okawa, Y. et al. (2017) An Anti-Human Lutheran Glycoprotein Phage Antibody Inhibits Cell Migration on Laminin-511: Epitope Mapping of the Antibody.
PLoS One. 12 (1): e0167860. -
Noda, G.S. et al. (2020) Specificities and isotypes of erythrocytes autoantibodies in patients with warm autoimmune hemolytic anemia
Rev Cubana Hematol Inmunol Hemoter 36 (4): e1283
- Synonyms
- B-CAM
- RRID
- AB_2065308
- UniProt
- P50895
- Entrez Gene
- BCAM
- GO Terms
- GO:0005055 laminin receptor activity
- GO:0005887 integral to plasma membrane
- GO:0007160 cell-matrix adhesion
- GO:0009897 external side of plasma membrane
- GO:0043236 laminin binding
MCA1982
MCA1982T
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Human ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up