Cytoplasmic Molecules Antibodies and Kits
Cytoplasmic Molecules Overview
Signaling for apoptosis via intrinsic (mitochondrial) and extrinsic (receptor-mediated) pathways results in the intracellular activation of specific cystolic proteases, called caspases, producing the caspase cascade.
These proteins are present in cells as inactive zymogens with a pro-domain and a catalytic domain. When activated, caspases initiate and regulate apoptosis.
Our range of apoptosis cytoplasmic antibodies are available in different formats and suitable for many applications.
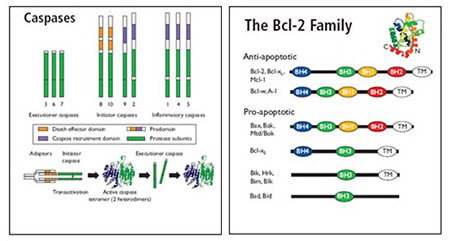
Caspases
Caspases are grouped as:
- Upstream initiators: caspase 8, 9, 10 and 12
- Downstream effectors: caspase 3, 6 and 7
Through the extrinsic apoptosis signalling pathway initiator caspases, e.g. caspase 8, bind to and are activated by a death inducing signaling complex (DISC). They then trigger pro-apoptotic pathways through the proteolytic cleavage and activation of effector caspases, thus creating the caspase cascade. The activated effector caspases amplify the apoptotis signal through enzyme activation and degradation of hundreds of intracellular protein targets. This leads to the loss of cellular function and destruction of cytoskeletal scaffold proteins, e.g. the intermediate filament cytokeratin 18, eventually resulting in cell death.
Our apoptosis range includes cytoplasmic antibodies and apoptosis detection kits to caspases and to caspase substrates that can be used as tools to ascertain if caspase activation has occurred:
Caspase Antibodies - suitable for many applications PARP Antibodies (Poly (ADP-Ribose) Polymerase) – assured for immunohistochemistry, immunoprecipitation and Western blotting
Cytokeratin 18 Antibodies – guaranteed for immunohistochemistry, immunofluorescence and Western blotting
Caspase FLICA™ Apoptosis Detection Kits - our wide range of Caspase FLICA™ apoptosis detection kits allow analysis of active caspases in whole, living cells. We offer two types of Caspase FLICA™ Kit; FAM FLICA™ that fluoresces green and SR FLICA™ which fluoresces red. The latter is useful for studying caspase activity in GFP-transfected cell lines and for dual-color experiments with other green reagents.
- Unique cell-permeable & non-cytotoxic FLICA™ reagent
- Quick & easy method
- Suitable for flow cytometry, fluorescence microscopy or a plate reader
Bcl-2 Family Members
The Bcl-2 family is comprised of over 20 cytosolic proteins characterized by four BH (Bcl-2 homology) domains that are essential for functionality. Bcl-2 proteins are categorized as;
Anti-apoptotic proteins, e.g. Bcl-2, Bcl-xL and Mcl-1, that prevent the release of sequestered mitochondrial cytochrome c and subsequent caspase 9 and 3 activation. The survival activity of the anti-apoptotic proteins may be blocked by the binding of Bcl-2 family pro-apoptotic proteins.
Pro-apoptotic proteins, which are subgrouped into the BH3 only subfamily; Bad, Bid, Bim/Bod, Bik, Blk, Hrk, BNIP3, and BimL, and the Bax subfamily containing several BH domains; Bax, Bak and Bok. The pro-apoptotic cytoplasmic proteins, Bax and Bak, form mitochondrial apoptosis-induced channels (MAC) in the outer mitochondrial membrane.
This activates mitochondrial outer membrane permeabilization (MOMP), which results in cytochrome c release. Bid is cleaved and activated by caspase 8 which starts the release of proapoptotic mitochondrial proteins as part of the intrinsic apoptosis pathway.
View our range of quality anti-apoptotic and pro-apoptotic Bcl-2 cytoplasmic antibodies available in different formats and suitable for many applications here.
Negative Regulation of Apoptosis
The complex apoptotic pathways are negatively regulated by cytoplasmic factors such as inhibitor of apoptosis (IAP) proteins e.g. livin and survivin, protein kinase Akt, FLICE inhibitory protein (FLIP), and anti-apoptotic Bcl-2 proteins.
Dysregulation of apoptosis may lead to pathological conditions. Apoptosis is associated with cancer, autoimmune diseases, transplant rejection, AIDS and neurodegenerative diseases.
A greater understanding of the proteins and mechanisms involved in apoptosis will help to develop novel therapeutic approaches to modulate cellular death pathways in the treatment of diseases.
View the other sections in our quality apoptosis antibodies and kits product range:
References
Caspases:
-
Cohen, G. M. (1997) Caspases: the executioners of apoptosis.
Biochem. J. 326: 1 - 16. -
Krajewski, M. et al. (1997) Immunohistochemical analysis of in vivo patterns of expression of CPP32 (Caspase-3), a cell death protease.
Cancer Res. 57: 1605-1613. -
Li, P. et al. (1997) Cytochrome c and dATP-dependent formation of Apaf-1/caspase-9 complex initiates an apoptosis protease cascade.
Cell 91: 479-489. -
Muzio, M. et al. (1996) FLICE, a novel FADD-homologous ICE/CED-3-like protease, is recruited to the CD95 (Fas/APO-1) death—inducing signaling complex.
Cell 85: 817 - 827. -
Pan, G. et al. (1998) Caspase-9, Bcl-XL and Apaf-1 form a ternary complex.
J. Biol. Chem. 273: 5841-5845. -
Wolf, B. B. and Green, D. R. (1999) Suicidal tendencies: apoptotic cell death by caspase family proteinases.
J. Biol. Chem. 274: 20049 - 20052.
Caspase Substrates:
-
Eto, H. et al. (2000) Mapping and regulation of the Tumor-associated epitope recognized by monoclonal antibody RS-11.
J. Biol. Chem. 275: 27075-27083. -
Freire, R. et al. (2001) Cleavage of the Bloom’s syndrome gene product during apoptosis by caspase-3 results in an impaired interaction with topoisomerase IIIalpha.
Nucleic Acids Res. 29: 3172-3180. -
Harris, J.L. et al. (2009) Aprataxin, poly-ADP ribose polymerase 1 (PARP-1) and apurinic endonuclease 1 (APE1) function together to protect the genome against oxidative damage.
Hum Mol Genet. 18: 4102-17. -
Holliday, D. et al. (2009) Novel multicellular organotypic models of normal and malignant breast: tools for dissecting the role of the microenvironment in breast cancer progression.
Breast Cancer Res 11: R3 -
Krohn, A. et al. (1998) Staurosporine-induced apoptosis of cultured rat hippocampal neurons involves caspase-1-like proteases as upstream initiators and increased production of superoxide as a main downstream effector.
J. Neurosci. 18: 8186-8197. - Macfarlane, M. et al. (2000) Active caspases and cleaved cytokeratins are sequestered into cytoplasmic inclusions in TRAIL-induced Apoptosis. J. Cell. Biol. 148: 1239-1254.
Bcl-2 Family:
-
Aster, J.C. and Logtine, J.A. (2002) Detection of BCL2 rearrangements in follicular lymphoma.
Am. J. Pathol. 160: 759-763. -
Bewarder, N. et al. (1996) In vivo and in vitro specificity of protein tyrosine kinases for immunoglobulin G receptor (Fc gamma R II) phosphorylation.
Mol. Cell Biol. 16: 4735-4743. -
Cleary, M.L. et al. (1986) Cloning and structural analysis of cDNAs for bcl-2 and a hybrid bcl-2/immunoglobulin transcript resulting from the t(14;18) translocation.
Cell. 47: 19-28. -
Hockenbery, D. et al. (1990) Bcl-2 is an inner mitochondrial membrane protein that blocks programmed cell death.
Nature. 348: 334-346. -
Hsu, Y.T. et al. (1997) Cytosol-to membrane redistribution of Bax and Bcl-XL during apoptosis.
P.N.A.S. 94: 3668-3672. -
Krajewski, S. et al. (1994) Immunohistochemical analysis of in vivo patterns of Bcl-X expression.
Cancer Res. 54: 5501-5507. -
Krajewski, S. et al. (1995) Reduced expression of proapoptotic gene BAX is associated with poor response rates to combination chemotherapy and shorter survival in women with metastatic breast adenocarcinoma.
Cancer Res. 55: 4471-4478. -
Krajewski, S. et al. (1996) Immunohistochemical analysis of in vivo patterns of Bak expression, a proapoptotic member of the Bcl-2 protein family.
Cancer Res. 56: 2849-2855. -
Mathai, J.P. et al. (2005) BH3-only BIK regulates BAX,BAK-dependent release of Ca2+ from endoplasmic reticulum stores and mitochondrial apoptosis during stress-induced cell death.
J. Biol. Chem. 280: 23829-23836. -
Michels, J. et al. (2005) Mcl-1. Int. J. Biochem.
Cell Biol. 37: 267-271. -
Nakamura M. et al. (2005) Frequent HRK inactivation associated with low apoptotic index in secondary glioblastomas.
Acta Neuropathol (Berl). 110: 402-410. -
Pezzella, F. et al. (1990) Expression of the bcl-2 oncogene protein is not specific for the 14; 18 chromosomal translocation.
Am. J. Pathol. 137: 225-232. -
Real, P.J. et al. (2006) Transcriptional activation of the proapoptotic bik gene by E2F proteins in cancer cells.
FEBS Lett. 580: 5905-5909. -
Sanz, C. et al. (2001) Interleukin 3-dependent activation of DREAM is involved in transcriptional silencing of the apoptotic Hrk gene in hematopoietic progenitor cells.
EMBO J. 20: 2286-2292. -
Sturm, I. et al. (2006) Loss of the tissue-specific proapoptotic BH3-only protein Nbk/Bik is a unifying feature of renal cell carcinoma.
Cell Death Differ. 13: 619-627. -
Sunayama, J. et al. (2004) Physical and functional interaction between BH3-only protein Hrk and mitochondrial pore-forming protein p32.
Cell Death Differ. 11: 771-781 - Tsujimoto, Y. et al. (1986) Analysis of the structure, transcripts and protein products of bcl-2, the gene involved in human follicular lymphoma. Proc. Natl. Acad. Sci. (USA) 83: 5214-5218.
-
Yang-Yen, H.F. (2006) Mcl-1: a highly regulated cell death and survival controller.
J. Biomed. Sci. 13: 201-204. -
Yin, X.M. (2006) Bid, a BH3-only multi-functional molecule, is at the cross road of life and death.
Gene. 369: 7-19. -
Zinkel, S. et al. (2006) BCL2 family in DNA damage and cell cycle control.
Cell Death Differ. 13: 1351-1359.
Negative Regulation of Apoptosis:
-
Hu S., et al. (1997) I-FLICE, a novel inhibitor of tumor necrosis factor receptor-1- and CD-95-induced apoptosis.
J. Biol. Chem. 272:17255-17257. -
Irmler M., et al. (1997) Inhibition of death receptor signals by cellular FLIP.
Nature 388:190-195. -
Li, F. et al. (1998) Control of apoptosis and mitotic spindle checkpoint by survivin.
Nature. 396: 580-4. -
Shu H.B., et al. (1997) Casper is a FADD- and caspase-related inducer of apoptosis.
Immunity 6:751-763. -
Stern, D.F. (2004) More than a marker… Phosphorylated Akt in prostate carcinoma.
Clin Cancer Res. 10: 6407-10. -
Thome M., et al. (1997) Viral FLICE-inhibitory proteins (FLIPs) prevent apoptosis induced by death receptors.
Nature 386:517-521. -
Vucic, D. et al. (2002) SMAC negatively regulates the anti-apoptotic activity of melanoma inhibitor of apoptosis (ML-IAP).
J. Biol. Chem. 277: 12275-12279.



