Apoptosis Cell Membrane Receptors - Death Receptors
Sign up to Our Emails

Be the first to know when we launch new products and resources to help you achieve more in the lab.
s
Cancer Research Posters

Epidermal Growth Factor Receptor Pathway, Acute Myeloid Leukemia, Melanoma & Mitochondrial Dynamics Posters
s
Apoptosis Webinar
"A question of Life or Death" - differentiating between healthy and apoptotic cells
Apoptosis Death Receptor
Apoptosis is the process of cell signalling auto-destruction often called ‘programmed cell death’. Apoptosis is a highly regulated cell signalling process characterized by morphological and biochemical cellular changes.
View our range of apoptosis death receptor - cell membrane antibodies available in different formats and suitable for many applications.
Extrinsic or Death Receptor Apoptosis Pathway
The extrinsic or death receptor apoptosis cell signalling pathway is initiated through ligand binding to death receptors on the cell membrane.
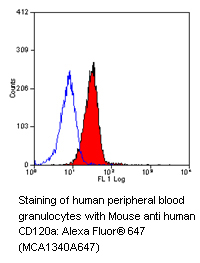
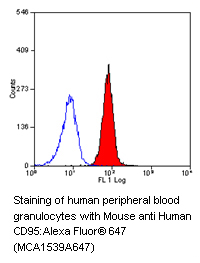
The death receptors TNF-R1 (CD120a), TNF-R2 (CD120b), Fas/APO-1 (CD95) and the TNF-related apoptosis-inducing ligand (TRAIL) receptors called DR3, DR4, DR5 and DR6 are subgrouped under the tumor necrosis factor (TNF) receptor superfamily.
This family also includes TWEAK Receptor (CD266 / Fn14) which interacts with its ligand TWEAK (TNF-like weak inducer of apoptosis) (CD255) inducing apoptosis in some tumour cell lines through pathways yet to be fully elucidated.
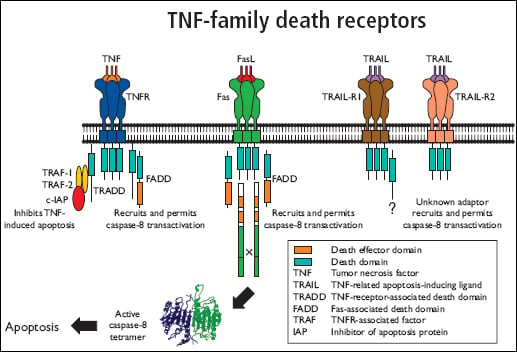
Key Death Receptors and their Ligands:
- TNF-R1 (CD120a) / TNF-R2 (CD120b) and TNF-alpha
- Fas/APO-1 (CD95) and Fas ligand (CD178)
- TRAIL receptors and TRAIL (CD253)
Structurally, the death receptors are categorised as TNFR associated factors (TRAFs) or death domain homologues which have a cysteine rich extracellular domain and a transmembrane or intracellular death domain.
The apoptosis cell signal is initiated through death receptor oligomerization and via the death domain, is transmitted intracellularly through the recruitment of cytoplasmic adaptor proteins such as Fas associated death domain protein (FADD) and tumour necrosis factor receptor 1-associated death domain protein (TRADD).
The adaptor proteins have a death effector domain which recruits cytosolic proteins e.g. pro-caspase 8 to form the death-inducing signaling complex (DISC) with subsequent activation of the caspase cascade.
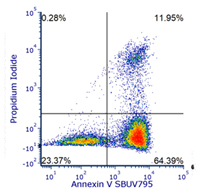
Annexin V — Quick & Easy Method for Detecting Apoptosis Phosphatidylserine Translocation
One of the earliest features of apoptosis is a morphological change in the plasma membrane. This involves the translocation of the membrane phospholipid phosphatidylserine (PS) during apoptosis from the internal layer to the external layer of the cell membrane.
In the presence of calcium ions, annexin V has a high specificity and affinity for PS. Thus, the binding of annexin V to cells with exposed PS provides a very sensitive method for detecting cellular apoptosis.
Further details about our Annexin V Detection Kits are available, formats now include StarBright Dye conjugated Annexin V.
CTL / Granzyme Mediated Apoptosis
Apoptosis of virally-infected cells and primary tumor cells may be induced by cytotoxic T lymphocytes (CTLs) through the release of perforin and granzymes from cytoplasmic cytolytic granules. Further details about this alternative caspase activation pathway are available.
Choose from our quality monoclonal granzyme antibodies:
Dysregulation of apoptosis may lead to pathological conditions. Apoptosis is associated with cancer, autoimmune diseases, transplant rejection, AIDS and neurodegenerative diseases. A greater understanding of the proteins and mechanisms involved in apoptosis will help to develop novel therapeutic approaches to modulate cellular death pathways in the treatment of diseases.
View the other sections in our quality apoptosis antibodies and kits product range:
References:
-
Chaudary, P.M. et al. (1997) Death receptor 5, a new member of the TNFR family, and DR4 induce FADD-dependent apoptosis and activate the NF-kappa B pathway.
Immunity 7: 821-830. -
Dri, P. et al. (1999) Role of the 75-kDa TNF receptor in TNF-induced activation of neutrophil respiratory burst.
J. Immunol. 162: 460-466. -
Kohrgruber, N. et al. (1999) Survival, maturation, and function of CD11c- and CD11c+ peripheral blood dendritic cells are differentially regulated by cytokines.
J Immunol. 163:3250-3259. -
Menegazzi, R. et al. (1994) Evidence that tumor necrosis factor alpha (TNF)-induced activation of neutrophil respiratory burst on biologic surfaces is mediated by the p55 TNF receptor.
Blood 84: 287-293. -
Pan, G. et al. (1997) The receptor for the cytotoxic ligand TRAIL.
Science 276: 111-113. -
Selinsky, C.L. et al. (1998) Multifaceted inhibition of anti-tumour immune mechanisms by soluble tumor necrosis factor receptor type I.
Immunology. 94: 88-93. -
Sheenhan, K.C.F. et al. (1995) Monoclonal Antibodies Specific for Murine p55 and p75 Tumor Necrosis Factor Receptors: Identification of a Novel In Vivo Role for p75.
J. Exp. Med.181: 607-617. -
Spahr, L. et al. (2004) Soluble TNF-R1, but not tumor necrosis factor alpha, predicts the 3-month mortality in patients with alcoholic hepatitis.
J. Hepatol. 41: 229-234. -
Testa, U. et al. (1998) The PML/RARalpha fusion protein inhibits tumor necrosis factor-alpha-induced apoptosis in U937 cells and acute promyelocytic leukemia blasts.
J. Clin. Invest. 101: 2278-2289. -
Thoma, B. et al. (1990) Identification of a 60-kD tumor necrosis factor (TNF) receptor as the major signal transducing component in TNF responses.
J. Exp. Med. 172: 1019-1023.




