Porcine Innate Immunity
Pig Innate Immune System Cells
The immune systems of swine and human are similar in their anatomy, organization and immune responses. The pig’s immune response is more similar to human (80%) than mouse (10%) and this, along with its size, makes it a more suitable and clinically relevant model for comparative immunology studies for human diseases.
The innate immune system is the first line of defense against infection from invading organisms and unlike the adaptive immune system it responds immediately.
The innate immune response is comprised of the complement system and different cell types: macrophages and neutrophils (phagocytes); basophils, mast cells and eosinophils (release inflammatory mediators); and natural killer (NK) cells. The germline encoded conserved pattern recognition receptors (PRRs) are mainly expressed on macrophages, neutrophils and dendritic cells.
Complement System
The components of the complement system are important mediators of the innate (and adaptive) immune system. In short, activation of the complement system results in the binding of C3b to the surface of the invading cell, the formation of membrane attack complexes (MACs) in the membrane of the pathogen leading to pore formation and finally, cell lysis. Macrophages and neutrophils express complement receptor 3 (CR3) that recognizes iC3b and induces phagocytosis of the lysed cells. The pig exhibits considerable single nucleotide polymorphisms (SNP) within the complement system which makes it an ideal model to study genes providing more resistance (and hence protection) to infection.
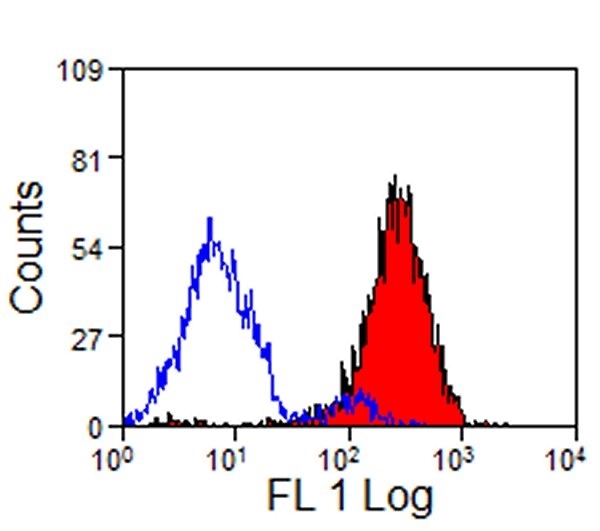
Staining of pig peripheral blood granulocytes with mouse anti-pig CD14 (MCA1218GA) followed by goat anti-mouse IgG:FITC (STAR117F)
Monocytes
In the pig, CD14 is expressed in most tissues, being highest on monocytes and tissue macrophages and lowest on granulocytes. Human, mouse and pig all express the CD16 monocyte marker. However, only 10% of human monocytes express CD16, whereas all mouse monocytes and most pig monocytes are CD16+.
As with human and mouse, pig monocytes can be subdivided into two distinct populations. In swine, these two subpopulations are identified by the expression of CD163 (haptoglobin scavenger receptor): CD14lo/CD163+/SLA-DR+ (called steady state) and CD14hi/CD163-/SLA-DR- (inflammatory monocyte). After infection, the CD14lo/CD163+/SLA-DR+ phenotype changes from a “steady state monocyte” to an “inflammatory monocyte” where SLA-DR is lost and expression of CD14 and CD16 is increased (Ondrackova et al. 2013).
Macrophages
Pig macrophages express PRRs that recognize pathogen-associated molecular patterns (PAMPs) intrinsically expressed by pathogens. Their response is rapid. For example, detecting LPS increases the production of pro-inflammatory cytokines TNF-alpha, IL-1beta, IL-6, IL-8, and IL-12 (Fairbairn et al. 2011) and of inflammatory chemokines CCL2, CCL8, CXCL2, CXCL3, CXCL5, IL-8 to recruit and activate other immune cells.
Tissue macrophages express high levels of CD163 making it an ideal marker for their detection.
Toll like receptors (TLRs) are the most characterized of the PPRs. TLR4 functions as a receptor for LPS and is involved in pro-inflammatory signaling in macrophages. Pig and human adaptor protein, MyD88 are very similar and as a result TLR signaling through the adaptor protein can be studied in the pig.
Dendritic Cells
Pig blood dendritic cells are CD135 (Flt3)+CD14- differentiating them from monocytes. Blood dendritic cells can be subdivided into two types: plasmacytoid cells (pDC) (secrete high amounts of type-I interferon (IFN) and represent 0.1-0.5% of peripheral blood mononuclear cells (PBMC)) and classical dendritic cells (cDC) which specialize in antigen presentation.
Pig pDC cells are CD14-CD4+CD172aloCD123+CD135+ cells, which express low levels of CD8, CD16, CD45RA and low levels of MHC class II. cDC cells have a similar phenotype to pDC but express high levels of MHC class II and lack CD4 and CD123. Two subsets of cDC can be identified using CD1, CD16 and CD172a.
Identifying DCs in tissues is more difficult due to overlapping functions and phenotypes of dendritic cells, macrophages and B cells. At least four possible DCs reside in the skin (Marquet et al. 2011): in the epidermis, Langerhans cells are CD172+CD1+CD16-CD163-CADM1+D207+MHCII+; in the dermis two populations (a CD207- and CD207+) can be found - CD172a-CD16-D163-CD207-CADM1+ and CD172a+CD163-/loCD16+CD207+CADM1-. A further subset of CD172a+ cells expresses higher levels of CD16: CD172a+CD16hiCD163+CD207-CADM1- CD206+CD209+.
Neutrophils, eosinophils and basophils
Neutrophils are the major innate phagocyte. They are attracted to sites of infection and inflammation by IL-1, IL-8 and TNF-alpha released from macrophages and complement proteins.
Pig neutrophil proteases are similar to those found in humans, making them an ideal model for testing drugs targeting neutrophil serine protease active sites. Neutrophils are SWC1+CD172+ CD16lo.
The SW1 antigen, although expressed on myeloid, resting T cells, macrophages and granulocytes is a useful marker to discriminate between neutrophils (SW1+CD172a+) and eosinophils (SWC1-CD172a+) and between T and NK cells (SWC1+CD172a-) and B cells (SWC1-CD172a-) and monocytes (SWC1+CD172a+).
The 2B2 antibody recognizes a granulocyte specific marker detected on neutrophils, eosinophils and basophils in both bone marrow and blood (Ezquerra et al. 2009). During neutrophil development, cells acquire the 6D10 antigen and up regulate SWC8 and CD16 to become CD172aloSWC8medCD16med6D10hi cells. Mature neutrophils express CD14 and 2B2 antigen and the expression level of 6D10 is reduced, whilst the expression of CD172a, SWC8 and CD16 is increased (CD14+2B2+CD172ahiSWC8hiCD16hi6D10med).
Pig neutrophils, basophils and eosinophils differ in their expression of SWC1, SWC8, CD14, 2B2 and 6D10 antigens. Whereas neutrophils are positive for all these antigens, eosinophils are SWC1-SWC8hiCD14-2B2+6D10-, and basophils are SWC1+SWC8-CD14-2B2+6D10-. Blood neutrophils heterogeneously express CD11R1, lung neutrophils are CD11R1-.
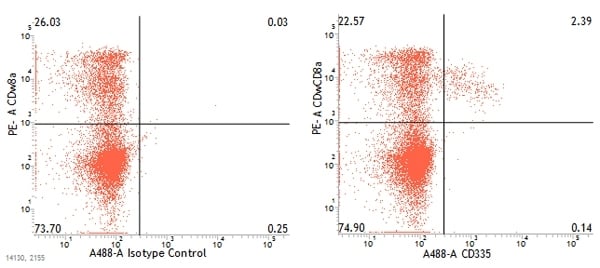
Dual staining of pig peripheral blood lymphocytes with mouse anti-pig CD335:Alexa Fluor®488 (MCA5972A488) and mouse anti-pig wCD8a:RPE (MCA1223PE)
Natural Killer Cells
Natural killer (NK) cells have lytic activity and can produce cytokines (IFN-gamma). They are a first line of defense against tumor and viral infections. NK cells are attracted to these cells by the cytokines (IL-1, IL-12, IL-15, and IL-18) and chemokine C ligand 5 (CCL5) they release. Cytokine stimulation results in an increased expression of CD25 and perforin and the induction of IFN-gamma in the porcine NK cells.
Compared to humans, pigs lack an NK specific marker and their NK cells are small in size and lack granules. They can be identified by the absence of T cell and T cell co-receptors and the expression of CD2, CD8alpha, CD16, CD45RC and perforin (Denyer et al. 2006). SLA-DR is present on a minor population of NK cells. Three subsets of NK cells can be identified in swine based on the level of NKp46 (CD335) expression: NKp46-, NKp46+ and NKp46hi. These three populations also express different levels of CD27 and CD8alpha: NKp46-CD27loCD8alpha; NKp46+CD27loCD8alpha and NKp46hi CD27hiCD8alphadim/-. NKp46+ and NKp46- cells can be found in all organs in pigs. In contrast, few NKp46hi NK cells are found in blood but they are abundant in both lymphoid and non-lymphoid tissues. The expression of the chemokine receptor CXCR3 on NKp46hi suggests that these cells exhibit a different migration and recruitment pattern than NKp46+ and NKp46- NK cells. The levels of NKp46 expression correlate with the levels of IFN-gamma and IFN-alpha produced by NK cells. NKp46- cells produce lower levels of these cytokines than NKp46hi NK cells.
To help you find further antibodies needed to investigate the innate side of the porcine immune system, please visit our pig antibodies page.
References:
- Denyer M et al (2006). Perforin expression can define CD8 positive lymphocyte subsets in pigs allowing phenotypic and functional analysis of natural killer, cytotoxic T, natural killer T and MHC un-restricted cytotoxic T-cells. Vet. Immunol. Immunopathol. 110(3-4):279
- Ezquerra A et al. (2009). Porcine myelomonocytic markers and cell populations. Dev. Comp. Immunol. 33(3): 284.
- Fairbairn L et al. (2011). The mononuclear phagocyte system of the pig as a model for understanding human innate immunity and disease. J. Leukoc. Biol. 89(6):855.
- Marquet F et al. (2011). Characterization of dendritic cells subpopulations in skin and afferent lymph in the swine model. PloS One 66(1).
- Ondrackova P et al. (2013). Distribution of porcine monocytes in different lymphoid tissues and the lungs during experimental Actinobacillus pleuropneumoniae infection and the role of chemokines. Vet. Res. 44:98.




