Metapneumovirus antibody | HMPV123

Mouse anti Metapneumovirus
- Product Type
- Monoclonal Antibody
- Clone
- HMPV123
- Isotype
- IgG2a
- Specificity
- Metapneumovirus
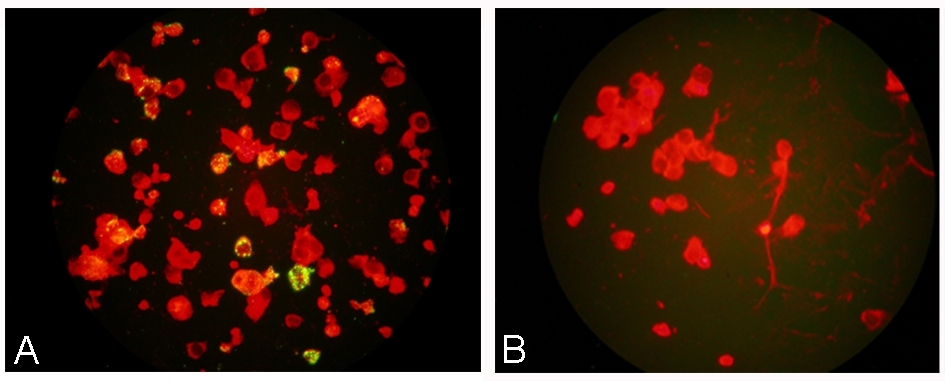
| Mouse anti Metapneumovirus antibody, clone HMPV123 recognizes human metapneumovirus (hMPV), a member of the Paramyxoviridae RNA virus family. Metapneumovirus was discovered in 2001 and is distributed worldwide. Infection in the upper respiratory tract is associated with mild illness, while lower respiratory tract infection can cause fever, cough, pneumonia, bronchiolitis and wheezing. Severe lower respiratory tract infection is most common in the very young, the very old and the immunosuppressed. Metapneumovirus is a common cause of respiratory infection. Phylogenetic analysis has shown two major genotypes, A and B, each containing two sub-divisions, suggesting that four distinct lineages circulate in the population with different lineages predominating in different years. Mouse anti Metapneumovirus antibody, clone HMPV123 recognizes the nucleoprotein of all sub-types (A1, A2, B1 and B2) of hMPV and may be used in conjunction with clones HMPV57, HMPV33 and HMPV24 to detect all strains of hMPV. Mouse anti Metapneumovirus antibody, clone HMPV123 does not cross-react with cell cultures infected with hRSV, influenzaviruses A and B, adenovirus, parainfluenza viruses 1, 2, 3 and 4b, mumps virus, measles virus, varicella-zoster virus, herpes simplex virus types 1 and 2, human cytomegalovirus, human herpesvirus type 6, ECHOvirus 19, Coxsachievirus B4, poliovirus types 1-3, HHV6 or uninfected HeLa, MA104, 3MK, LLC-MK, HEp2, MRC-5 and HSB-2 cells. It also does not react with sputum bacteria. |
- Target Species
- Viral
- Product Form
- Purified IgG - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% Sodium Azide (NaN3)
- Carrier Free
- Yes
- Immunogen
- Sub-type A hMPV virus isolate NCL03-4/145.
- Approx. Protein Concentrations
- IgG concentration 1.0mg/ml
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Immunofluorescence | 1/40 | 1/80 |
References for Metapneumovirus antibody
-
Fenwick, F. et al. (2007) Diagnosis of human metapneumovirus by immunofluorescence staining with monoclonal antibodies in the North-East of England.
J Clin Virol. 40 (3): 193-6.
- Synonyms
- MPV
- RRID
- AB_1628870
MCA4671
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Viral ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up