Influenza A Matrix Protein antibody | GA2B










Mouse anti Influenza A Matrix Protein
- Product Type
- Monoclonal Antibody
- Clone
- GA2B
- Isotype
- IgG1
- Specificity
- Influenza A Matrix Protein
| Mouse anti Influenza A matrix protein 1 antibody, clone GA2B recognizes an epitope within the influenza A matrix protein 1. In both strains of virus used as immunogen to isolate clone GA2B, the matrix protein 1 is a 252 amino acid, highly conserved viral protein playing a crucial role in replication. Mouse anti Influenza A matrix protein 1 antibody, clone GA2B can be used in influenza A IFA typing in conjunction with Mouse anti Influenza A matrix protein, clone AA5H. |
- Target Species
- Viral
- Product Form
- Purified IgG - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant.
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- <0.1% Sodium Azide (NaN3)
- Immunogen
- Influenza A / Puerto Rico / 8 / 34 (H1N1) and A/Bangkok / 1 / 79 (H3N2) viruses.
- Purity
- >90% IgG content as established by SDS PAGE
- Approx. Protein Concentrations
- IgG concentration 1.0 mg/ml
- Fusion Partners
- Spleen cells from immunised BALB/c mice were fused with cells of the P3 Ag8.653 mouse myeloma cell line.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Immunofluorescence | 1/100 | ||
| Immunohistology - Paraffin | |||
| Western Blotting |
References for Influenza A Matrix Protein antibody
-
Latham, T. & Galarza, J.M. (2001) Formation of wild-type and chimeric influenza virus-like particles following simultaneous expression of only four structural proteins.
J Virol. 75 (13): 6154-65. -
Viemann, D. et al. (2011) H5N1 virus activates signaling pathways in human endothelial cells resulting in a specific imbalanced inflammatory response.
J Immunol. 186 (1): 164-73. -
Yamamoto, Y. et al. (2008) Avian influenza virus (H5N1) replication in feathers of domestic waterfowl.
Emerg Infect Dis. 14: 149-51. -
Doucet, J.D. et al. (2011) Endogenously expressed matrix protein M1 and nucleoprotein of influenza A are efficiently presented by class I and class II major histocompatibility complexes.
J Gen Virol. 92 (Pt 5): 1162-71. -
Tanimura N et al. (2006) Pathology of fatal highly pathogenic H5N1 avian influenza virus infection in large-billed crows (Corvus macrorhynchos) during the 2004 outbreak in Japan.
Vet Pathol. 43 (4): 500-9. -
Kirkeby, S. et al. (2009) Infection with human H1N1 influenza virus affects the expression of sialic acids of metaplastic mucous cells in the ferret airways.
Virus Res. 144: 225-32. -
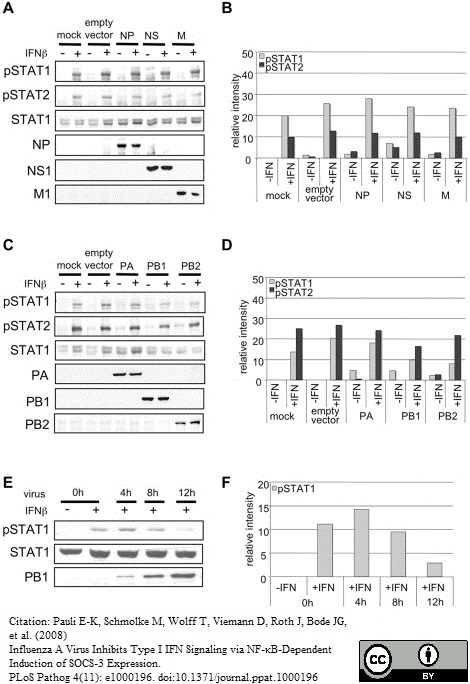
Pauli, E.K. et al. (2008) Influenza A virus inhibits type I IFN signaling via NF-kappaB-dependent induction of SOCS-3 expression.
PLoS Pathog. 4(11): e1000196. -
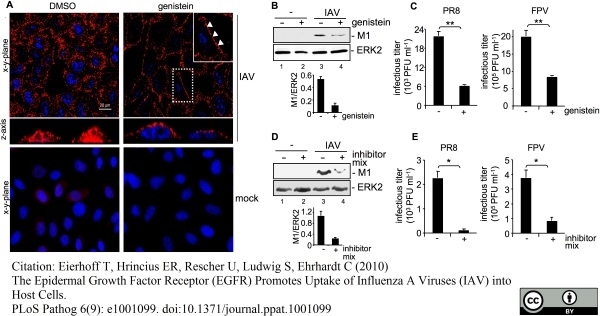
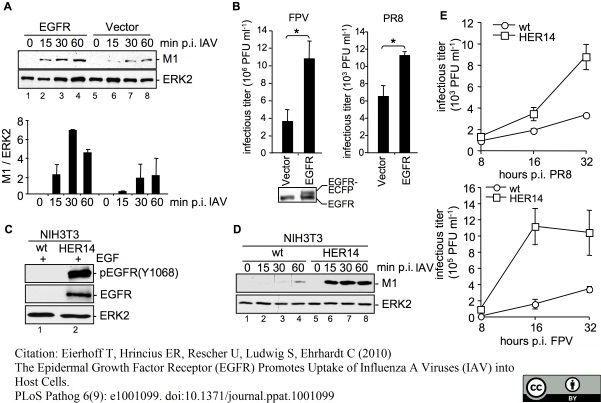
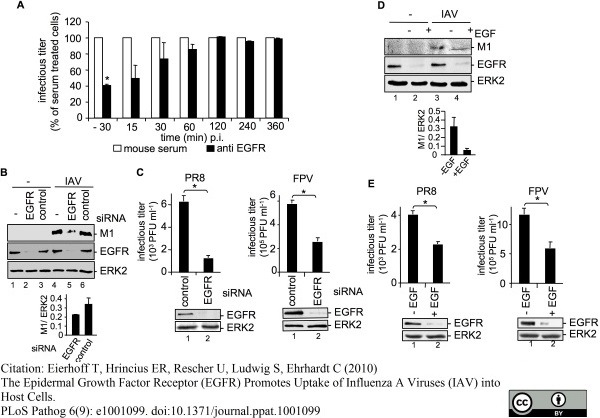
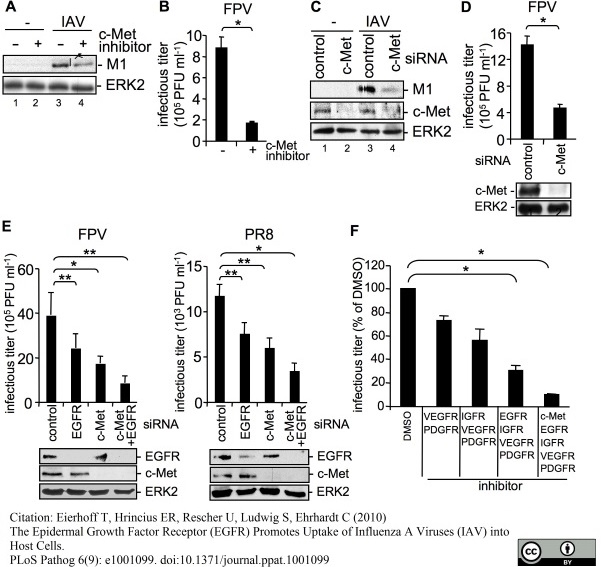
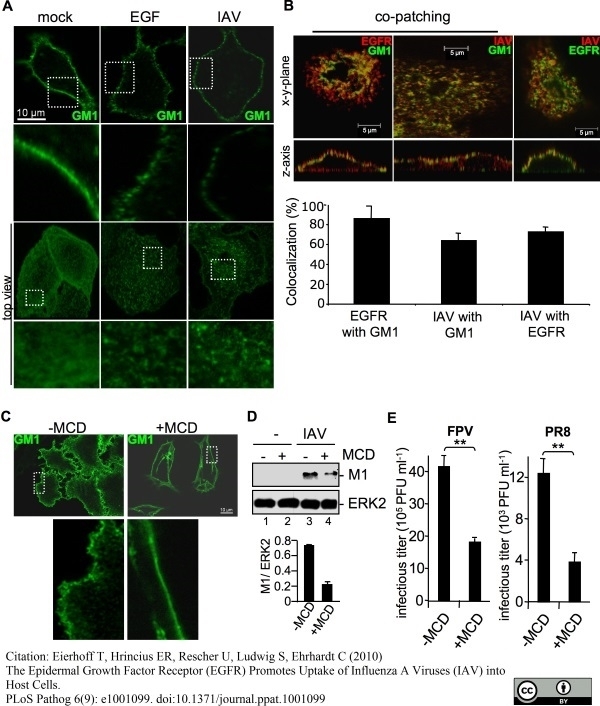
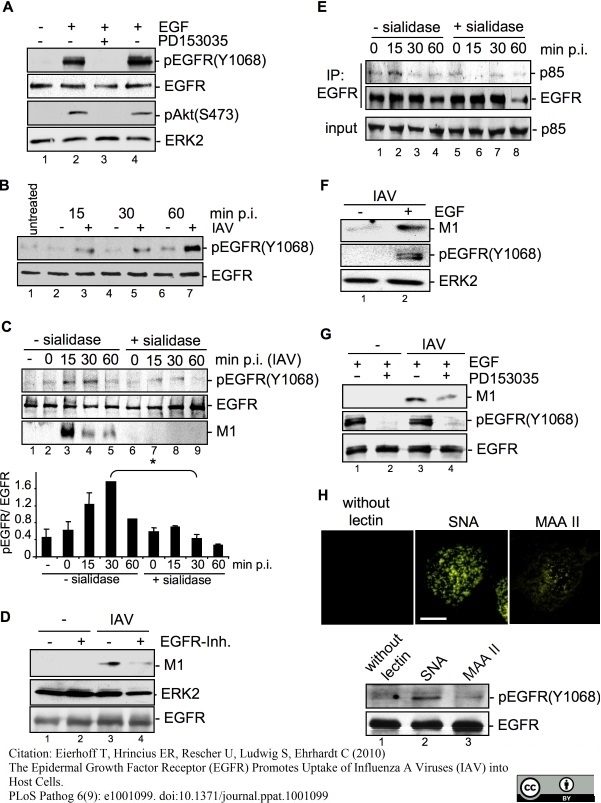
Eierhoff, T. et al. (2010) The epidermal growth factor receptor (EGFR) promotes uptake of influenza A viruses (IAV) into host cells.
PLoS Pathog. 6. pii: e1001099.
View The Latest Product References
-
Wang, D. et al. (2010) The lack of an inherent membrane targeting signal is responsible for the failure of the matrix (M1) protein of influenza A virus to bud into virus-like particles.
J Virol. 84: 4673-81. -
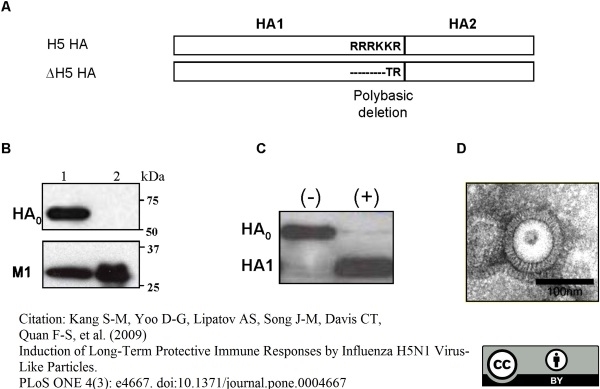
Kang, S.M. et al. (2009) Induction of long-term protective immune responses by influenza H5N1 virus-like particles.
PLoS One. 4: e4667. -
Luig, C. et al. (2010) MAP kinase-activated protein kinases 2 and 3 are required for influenza A virus propagation and act via inhibition of PKR.
FASEB J. 24: 4068-77. -
Schmolke, M. et al. (2009) Essential impact of NF-kappaB signaling on the H5N1 influenza A virus-induced transcriptome.
J Immunol. 183: 5180-9. -
Reinhardt, J. and Wolff, T. (2000) The influenza A virus M1 protein interacts with the cellular receptor of activated C kinase (RACK) 1 and can be phosphorylated by protein kinase C.
Vet Microbiol. 74: 87-100. -
Das, S.C. et al. (2012) The Highly Conserved Arginine Residues at Positions 76 through 78 of Influenza A Virus Matrix Protein M1 Play an Important Role in Viral Replication by Affecting the Intracellular Localization of M1.
J Virol. 86: 1522-30. -
Liu, Y.V. et al. (2011) Chimeric severe acute respiratory syndrome coronavirus (SARS-CoV) S glycoprotein and influenza matrix 1 efficiently form virus-like particles (VLPs) that protect mice against challenge with SARS-CoV.
Vaccine. 29: 6606-13. -
Moncorgé, O. et al. (2013) Investigation of influenza virus polymerase activity in pig cells.
J Virol. 87 (1): 384-94. -
Khaperskyy, D.A. et al. (2012) Influenza A virus inhibits cytoplasmic stress granule formation.
FASEB J. 26: 1629-39. -
Friesenhagen, J. et al. (2012) Highly pathogenic avian influenza viruses inhibit effective immune responses of human blood-derived macrophages.
J Leukoc Biol. 92: 11-20. -
Londrigan, S.L. et al. (2015) Infection of Mouse Macrophages by Seasonal Influenza Viruses Can Be Restricted at the Level of Virus Entry and at a Late Stage in the Virus Life Cycle.
J Virol. 89 (24): 12319-29. -
Sadewasser, A. et al. (2017) Quantitative Proteomic Approach Identifies Vpr Binding Protein as Novel Host Factor Supporting Influenza A Virus Infections in Human Cells.
Mol Cell Proteomics. 16 (5): 728-42. -
Liu, Y.V. et al. (2015) Recombinant virus-like particles elicit protective immunity against avian influenza A(H7N9) virus infection in ferrets.
Vaccine. 33 (18): 2152-8. -
Huang, M.T. et al. (2015) DcR3 suppresses influenza virus-induced macrophage activation and attenuates pulmonary inflammation and lethality.
J Mol Med (Berl). 93 (10): 1131-43. -
Al-Mubarak, F. et al. (2015) Identification of morphological differences between avian influenza A viruses grown in chicken and duck cells.
Virus Res. 199: 9-19. -
Yang, C.H. et al. (2017) Influenza A virus upregulates PRPF8 gene expression to increase virus production.
Arch Virol. 162 (5): 1223-35. -
Smith, G.E. et al. (2017) Neuraminidase-based recombinant virus-like particles protect against lethal avian influenza A(H5N1) virus infection in ferrets.
Virology. 509: 90-97. -
Usui, T. et al. (2020) Outbreaks of highly pathogenic avian influenza in zoo birds caused by HA clade 2.3.4.4 H5N6 subtype viruses in Japan in winter 2016.
Transbound Emerg Dis. 67 (2): 686-697. -
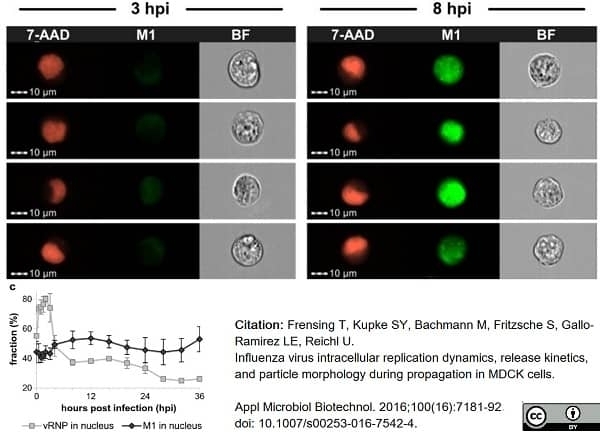
Frensing, T. et al. (2016) Influenza virus intracellular replication dynamics, release kinetics, and particle morphology during propagation in MDCK cells.
Appl Microbiol Biotechnol. 100 (16): 7181-92. -
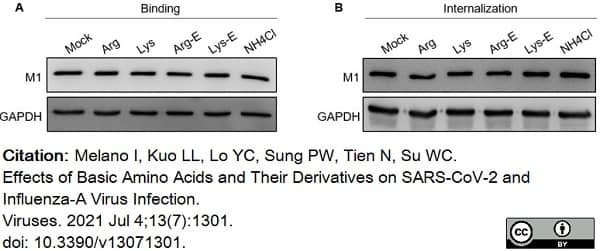
Melano, I. et al. (2021) Effects of Basic Amino Acids and Their Derivatives on SARS-CoV-2 and Influenza-A Virus Infection.
Viruses. 13 (7):1301. -
Soda, K. et al. (2022) Susceptibility of herons (family: Ardeidae) to clade 2.3.2.1 H5N1 subtype high pathogenicity avian influenza virus.
Avian Pathol. 51 (2): 146-53. -
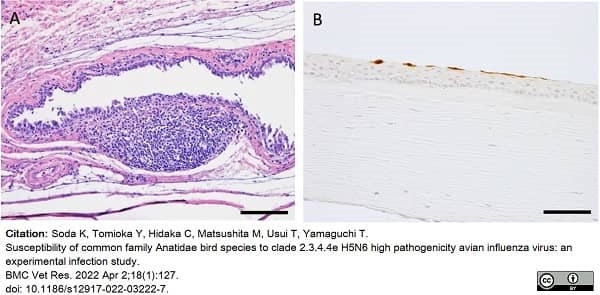
Soda, K. et al. (2022) Susceptibility of common family Anatidae bird species to clade 2.3.4.4e H5N6 high pathogenicity avian influenza virus: an experimental infection study.
BMC Vet Res. 18 (1): 127. -
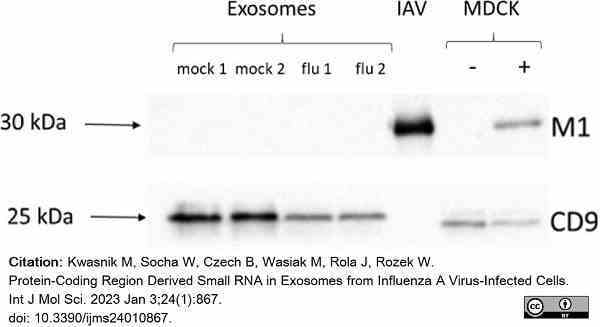
Kwasnik, M. et al. (2023) Protein-Coding Region Derived Small RNA in Exosomes from Influenza A Virus-Infected Cells.
Int J Mol Sci. 24(1):867. -
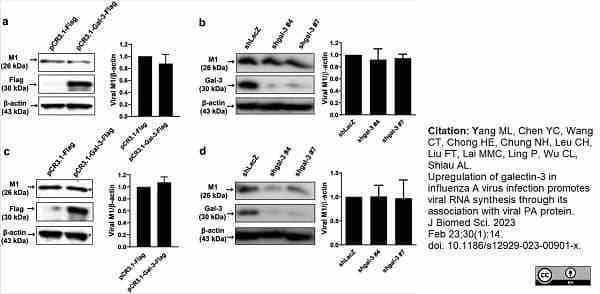
Yang, M.L. et al. (2023) Upregulation of galectin-3 in influenza A virus infection promotes viral RNA synthesis through its association with viral PA protein.
J Biomed Sci. 30 (1): 14. -
Soda, K. et al. (2023) Susceptibility of common dabbling and diving duck species to clade 2.3.2.1 H5N1 high pathogenicity avian influenza virus: an experimental infection study.
J Vet Med Sci. 85 (9): 942-9. -
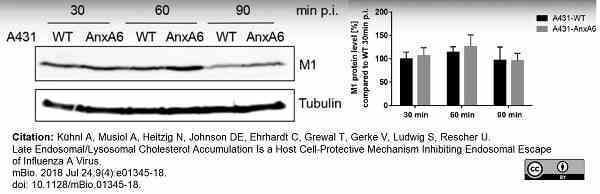
Kühnl, A. et al. (2018) Late Endosomal/Lysosomal Cholesterol Accumulation Is a Host Cell-Protective Mechanism Inhibiting Endosomal Escape of Influenza A Virus.
mBio. 9 (4): e01345-18.
- RRID
- AB_322157
- UniProt
- P03485
- P03487
- Entrez Gene
- M1
- GO Terms
- GO:0005886 plasma membrane
- GO:0003723 RNA binding
- GO:0005829 cytosol
- GO:0005198 structural molecule activity
- GO:0005654 nucleoplasm
- GO:0019064 viral envelope fusion with host membrane
- GO:0019061 uncoating of virus
- GO:0019065 receptor-mediated endocytosis of virus by host
- GO:0019070 viral genome maturation
- View More GO Terms
- GO:0019072 viral genome packaging
- GO:0019076 release of virus from host
- GO:0019083 viral transcription
- GO:0030666 endocytic vesicle membrane
- GO:0030683 evasion by virus of host immune response
- GO:0031904 endosome lumen
- GO:0042025 host cell nucleus
- GO:0046796 viral genome transport in host cell
- GO:0055036 virion membrane
- GO:0016020 membrane
MCA401
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Viral ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up