OX-62 antibody | OX-62
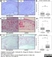




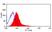
Mouse anti Rat OX-62
- Product Type
- Monoclonal Antibody
- Clone
- OX-62
- Isotype
- IgG1
- Specificity
- OX-62
| Mouse anti Rat OX-62 antibody, clone OX-62 recognizes the OX-62 antigen, also known as rat alpha E integrin or CD103. OX-62 appears as an 1150 amino acid, ~130 kDa single pass type I transmembrane protein expressed by intestinal dendritic cells, dendritic epidermal T cells, intraepithelial lymphocytes in the small intestine and by cells of dendritic morphology in lymphoid organs, at sites where gamma delta T cells are present. |
- Target Species
- Rat
- Product Form
- MCA1029G: Purified IgG - liquid
- MCA1029GA: Purified IgG - liquid.
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% sodium azide (NaN3)
- Carrier Free
- Yes
- Immunogen
- Density gradient enriched PVG rat thoracic duct dendritic cells.
- Approx. Protein Concentrations
- IgG concentration 1.0 mg/ml
- Fusion Partners
- Spleen cells of BALB/c mice were fused with cells of the mouse NS0 myeloma cell line.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
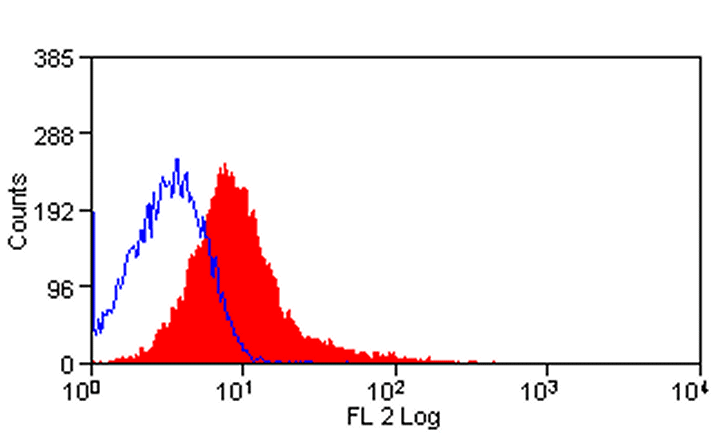
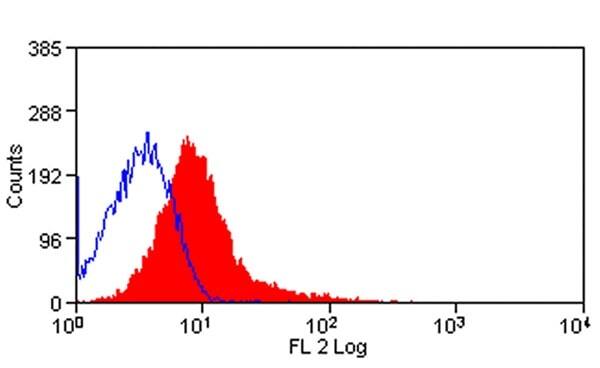
| Flow Cytometry | 1/50 | 1/100 | |
| Immunohistology - Frozen | 1/25 | 1/100 | |
| Immunoprecipitation | |||
| Western Blotting |
- Flow Cytometry
- Use 10μl of the suggested working dilution to label 106 cells in 100μl
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG1 Negative Control | MCA1209 | F | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG1 Negative Control | ||||||
Source Reference
-
Brenan, M. & Puklavec, M. (1992) The MRC OX-62 antigen: a useful marker in the purification of rat veiled cells with the biochemical properties of an integrin.
J Exp Med. 175 (6): 1457-65.
References for OX-62 antibody
-
Chen-Woan, M. et al. (1995) A new protocol for the propagation of dendritic cells from rat bone marrow using recombinant GM-CSF, and their quantification using the mAb OX-62.
J Immunol Methods. 178 (2): 157-71. -
Brenan, M. & Rees, D.J. (1997) Sequence analysis of rat integrin alpha E1 and alpha E2 subunits: tissue expression reveals phenotypic similarities between intraepithelial lymphocytes and dendritic cells in lymph.
Eur J Immunol. 27 (11): 3070-9. -
Varas, A. et al. (1998) Interleukin-7 influences the development of thymic dendritic cells.
Blood. 92: 93-100. -
Henry, F. et al. (1999) Antigen-presenting cells that phagocytose apoptotic tumor-derived cells are potent tumor vaccines.
Cancer Res. 59: 3329-32. -
Aiello, S. et al. (2000) Thymic Dendritic Cells Express Inducible Nitric Oxide Synthase and Generate Nitric Oxide in Response to Self- and Alloantigens
J Immunol. 164: 4649-58. -
O'Sullivan, N.L. et al. (2001) Lymphocyte lineages at mucosal effector sites: rat salivary glands.
J Immunol. 166: 5522-9. -
Kostulas, N. et al. (2002) Dendritic cells are present in ischemic brain after permanent middle cerebral artery occlusion in the rat.
Stroke. 33 (4): 1129-34. -
Tsuchiya, T. et al. (2002) Dendritic cell involvement in pulmonary granuloma formation elicited by bacillus calmette-guérin in rats.
Am J Respir Crit Care Med. 165 (12): 1640-6.
View The Latest Product References
-
Camelo, S. et al. (2003) Local retention of soluble antigen by potential antigen-presenting cells in the anterior segment of the eye.
Invest Ophthalmol Vis Sci. 44: 5212-9. -
Penttila, I.A. et al. (2003) Maternal milk regulation of cell infiltration and interleukin 18 in the intestine of suckling rat pups.
Gut. 52: 1579-86. -
Camelo, S. et al. (2004) The distribution of antigen in lymphoid tissues following its injection into the anterior chamber of the rat eye.
J Immunol. 172: 5388-95. -
Sugawara, I. et al. (2004) Pathological and immunological profiles of rat tuberculosis.
Int J Exp Pathol. 85: 125-34. -
Stephens, L.A. et al. (2004) Phenotypic characterization of regulatory CD4+CD25+ T cells in rats.
Int Immunol. 16: 365-75. -
Rival, C. et al. (2006) Identification of a dendritic cell population in normal testis and in chronically inflamed testis of rats with autoimmune orchitis.
Cell Tissue Res. 324 (2): 311-8. -
Baca Jones, C.C. et al. (2009) Rat cytomegalovirus infection depletes MHC II in bone marrow derived dendritic cells.
Virology. 388: 78-90. -
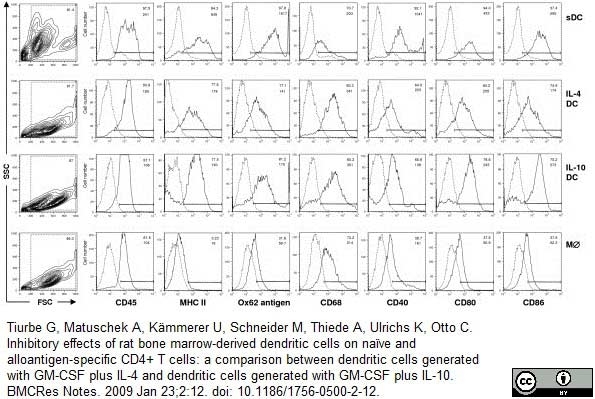
Tiurbe, G. et al. (2009) Inhibitory effects of rat bone marrow-derived dendritic cells on naïve and alloantigen-specific CD4+ T cells: a comparison between dendritic cells generated with GM-CSF plus IL-4 and dendritic cells generated with GM-CSF plus IL-10.
BMC Res Notes. 2: 12. -
Zilka, N. et al. (2009) Human misfolded truncated tau protein promotes activation of microglia and leukocyte infiltration in the transgenic rat model of tauopathy.
J Neuroimmunol. 209 (1-2): 16-25. -
Schwartzkopff, J. et al. (2010) NK cell depletion delays corneal allograft rejection in baby rats.
Mol Vis. 16: 1928-35. -
Zhou, Y.J. et al. (2010) The role of the lactadherin in promoting intestinal DCs development in vivo and vitro.
Clin Dev Immunol. 2010: 357541. -
Bufan, B. et al. (2015) Aging Impairs Endocytic Capacity Of Splenic Dendritic Cells From Dark Agouti Rats And Alters Their Response To TLR4 Stimulation
Acta Veterinaria. Volume 65, Issue 1, Pages 30–55. -
Syrjälä, S. et al. (2015) Donor Heart Treatment With COMP-Ang1 Limits Ischemia-Reperfusion Injury and Rejection of Cardiac Allografts.
Am J Transplant. 15 (8): 2075-84. -
Stojić-Vukanić, Z. et al. (2016) Estradiol enhances capacity of TLR-matured splenic dendritic cells to polarize CD4+ lymphocytes into IL-17/GM-CSF-producing cells in vitro.
Int Immunopharmacol. 40: 244-253. -
Palin, N.K. et al. (2017) Activin inhibition limits early innate immune response in rat kidney allografts-a pilot study.
Transpl Int. 30 (1): 96-107. -
Raissadati, A. et al. (2017) Vascular Endothelial Growth Factor-B Overexpressing Hearts Are Not Protected From Transplant-Associated Ischemia-Reperfusion Injury.
Exp Clin Transplant. 15 (2): 203-12. -
Stojić-Vukanić, Z. et al. (2020) Age and sex determine CD4+ T cell stimulatory and polarizing capacity of rat splenic dendritic cells.
Biogerontology. 21 (1): 83-107. -
Pilipović, I. et al. (2019) Noradrenaline modulates CD4+ T cell priming in rat experimental autoimmune encephalomyelitis: a role for the α1-adrenoceptor.
Immunol Res. 67 (2-3): 223-40. -
Lauzon-Joset, J.F. et al. (2020) Oestrogen amplifies pre-existing atopy-associated Th2 bias in an experimental asthma model.
Clin Exp Allergy. 50 (3): 391-400. -
Yu, J. et al. (2022) Topical Administration of 0.3% Tofacitinib Suppresses M1 Macrophage Polarization and Allograft Corneal Rejection by Blocking STAT1 Activation in the Rat Cornea
Transl Vis Sci Technol. 11 (3): 34 -
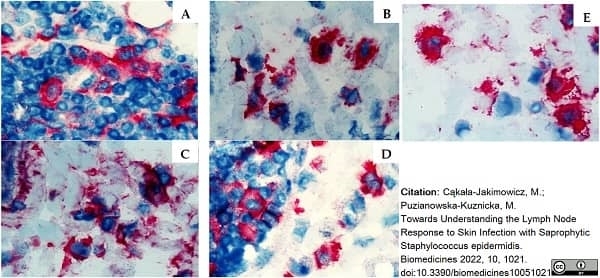
Cąkała-Jakimowicz, M. & Puzianowska-Kuznicka, M. (2022) Towards Understanding the Lymph Node Response to Skin Infection with Saprophytic Staphylococcus epidermidis..
Biomedicines. 10 (5): 1021. -
de Jesus, E.J. et al. (2016) Adoptive Transfer of Dendritic Cells Expressing Fas Ligand Modulates Intestinal Inflammation in a Model of Inflammatory Bowel Disease.
J Clin Cell Immunol. 7 (2): 411. -
Leffler, J. et al. (2018) Functional differences in airway dendritic cells determine susceptibility to IgE-sensitization.
Immunol Cell Biol. 96 (3): 316-29. -
Yu, J. et al. (2021) Subconjunctival injections of dimethyl fumarate inhibit lymphangiogenesis and allograft rejection in the rat cornea.
Int Immunopharmacol. 96: 107580. -
Djuretić, J. et al. (2019) Natural killer cells as participants in pathogenesis of rat experimental autoimmune encephalomyelitis (EAE): lessons from research on rats with distinct age and strain.
Cent Eur J Immunol. 44 (4): 337-356.
- Synonyms
- CD103
- RRID
- AB_2128733
- UniProt
- O88341
MCA1029G
MCA1029GA
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Rat ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up