Neurofilament H antibody | RT97

Mouse anti Rat Neurofilament 200kDa
- Product Type
- Monoclonal Antibody
- Clone
- RT97
- Isotype
- IgG1
- Specificity
- Neurofilament H
- Region
- 200kDa
| Mouse anti Rat Neurofilament 200kDa antibody, clone RT97 recognizes the 200kDa neurofilament protein in a range of species. Mouse anti Rat Neurofilament 200kDa antibody, clone RT97 stains various tumors including phaeochromocytoma, paraganglioma and ganglioneuroblastoma. Mouse anti Rat Neurofilament 200kDa antibody, clone RT97 also recognizes a phosphorylation dependent epitope on fetal tau, Alzheimer's paired helical filament-tau and on microtubule associated protein 1B (MAP1B) by western blotting, however similar reactivity was not apparent in immunohistochemistry (Johnstone et al. 1997). |
- Target Species
- Rat
- Species Cross-Reactivity
-
Target Species Cross Reactivity Reptile Expected from Sequence Human Chicken Pig Mouse - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG - liquid
- Preparation
- Purified IgG prepared by hydrophobic interaction chromatography from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.1% sodium azide (NaN3)
1% bovine serum albumin - Immunogen
- Triton X-100-insoluble rat brain protein.
- Approx. Protein Concentrations
- IgG concentration 0.1 mg/ml
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Immunohistology - Frozen | |||
| Immunohistology - Paraffin | 1/50 | 1/100 |
- Histology Positive Control Tissue
- Brain or spinal cord
References for Neurofilament H antibody
-
Anderton, B.H. et al. (1982) Monoclonal antibodies show that neurofibrillary tangles and neurofilaments share antigenic determinants.
Nature. 298 (5869): 84-6. -
Weber, K. et al. (1983) Neurofilaments, a subclass of intermediate filaments: structure and expression.
Cold Spring Harb Symp Quant Biol. 48 Pt 2: 717-29. -
Connolly ,A.A. et al. (1987) A comparative study of a silver stain and monoclonal antibody reactions on Alzheimer's neurofibrillary tangles.
J Neurol Neurosurg Psychiatry. 50: 1221-4. -
Doering, L.C. (1991) Transplantation of fetal CNS tissue into the peripheral nervous system: a model to study aberrant changes in the neuronal cytoskeleton.
J Neural Transplant Plast. 2: 193-205. -
McCarthy, P.W. et al. (1992) RT97- and calcitonin gene-related peptide-like immunoreactivity in lumbar intervertebral discs and adjacent tissue from the rat.
J Anat. 180: 15-24. -
Murphy, A. et al. (1993) Neurofilament expression in human T lymphocytes.
Immunology. 79: 167-70. -
Sann, H. et al. (1995) RT97: a marker for capsaicin-insensitive sensory endings in the rat skin.
Cell Tissue Res. 282 (1): 155-61. -
Tonge, D.A. et al. (1996) Expression of a developmentally regulated, phosphorylated isoform of microtubule-associated protein 1B in sprouting and regenerating axons in vitro.
Neuroscience. 73 (2): 541-51.
View The Latest Product References
-
Johnstone, M. et al. (1997) The neurofilament antibody RT97 recognises a developmentally regulated phosphorylation epitope on microtubule-associated protein 1B.
J Anat. 191 ( Pt 2): 229-44. -
Wang, S. et al. (2000) Progressive optic axon dystrophy and vacuslar changes in rd mice.
Invest Ophthalmol Vis Sci. 41: 537-45. -
Yabe, J.T. et al. (2001) Neurofilaments consist of distinct populations that can be distinguished by C-terminal phosphorylation, bundling, and axonal transport rate in growing axonal neurites.
J Neurosci. 21: 2195-205. -
Shin, D.H. et al. (2003) The correspondence between the labeling patterns of antibody RT97, neurofilaments, microtubule associated protein 1B and tau varies with cell types and development stages of chicken retina.
Neurosci Lett. 342: 167-70. -
Kuwamura, M. et al. (2004) Cerebral ganglioneuroblastoma in a golden retriever dog.
Vet Pathol. 41: 282-4. -
Lorber, B. et al. (2004) Stimulated regeneration of the crushed adult rat optic nerve correlates with attenuated expression of the protein tyrosine phosphatases RPTPalpha, STEP, and LAR.
Mol Cell Neurosci. 27: 404-16. -
Reynolds, J. et al. (2005) Age-dependent changes in Fibroblast growth factor 2 (FGF-2) expression in mouse cerebellar neurons.
J Cell Mol Med. 9: 398-406. -
Logan, A. et al. (2006) Neurotrophic factor synergy is required for neuronal survival and disinhibited axon regeneration after CNS injury.
Brain. 129: 490-502. -
Veeranna, et al. (2008) Neurofilament tail phosphorylation: identity of the RT-97 phosphoepitope and regulation in neurons by cross-talk among proline-directed kinases.
J Neurochem. 2008 Oct;107(1): 35-49. -
Vidal-Sanz, M. et al. (2015) Retinal neurodegeneration in experimental glaucoma.
Prog Brain Res. 220: 1-35. -
Rovere, G. et al. (2015) Comparison of Retinal Nerve Fiber Layer Thinning and Retinal Ganglion Cell Loss After Optic Nerve Transection in Adult Albino Rats.
Invest Ophthalmol Vis Sci. 56 (8): 4487-98. -
Wang, J. et al. (2017) MicroRNA regulation in an animal model of acute ocular hypertension.
Acta Ophthalmol. 95 (1): e10-e21. -
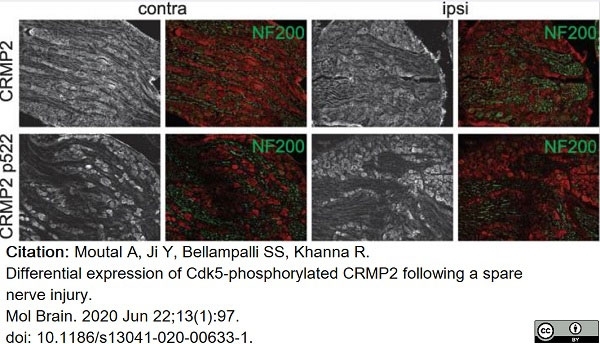
Moutal, A. et al. (2020) Differential expression of Cdk5-phosphorylated CRMP2 following a spared nerve injury.
Mol Brain. 13 (1): 97.
- Synonyms
- Neurofilament Heavy Polypeptide
- RRID
- AB_1102789
- UniProt
- P16884
- Entrez Gene
- Nefh
MCA1321GA
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Rat ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up