MHC Class II RT1Bu/L antibody | OX-3

Mouse anti Rat MHC Class II RT1Bu/L
- Product Type
- Monoclonal Antibody
- Clone
- OX-3
- Isotype
- IgG1
- Specificity
- MHC Class II RT1Bu/L
| Mouse anti Rat MHC Class II RT1Bu/L antibody, clone OX-3 recognizes a polymorphic determinant of the rat RT1B MHC class II antigen, reacting with haplotypes u and l. The literature reports reactivity with Lewis, Wistar and AO strain rats but not BN, DA or PVG/c strains. This antibody is useful for distinguishing RT1B positive cells from different rat strains, e.g. for recognising cells of donor origin in bone marrow reconstituted radiation chimaeras. The major histocompatibility complex (MHC) is a cluster of genes that are important in the immune response to infections. In rats, this complex is referred to as the RT1 region. In mice, this complex is referred to as the H-2 region. Mouse anti Rat MHC Class II RT1Bu/L antibody, clone OX-3 also cross reacts with mouse strains of the H-2 haplotypes b and s. Analysis of recombinant mouse strains has mapped the OX-3 determinant to the H-2I-A region. This product is routinely tested in flow cytometry on Lewis rat splenocytes. |
- Target Species
- Rat
- Species Cross-Reactivity
-
Target Species Cross Reactivity Mouse - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG - liquid
- Preparation
- MCA45GA: Purified IgG prepared by ion exchange chromatography from tissue culture supernatant
- MCA45R: Purified IgG prepared by affinity chromatography on Protein G from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
-
MCA45GA:
0.09% Sodium Azide - MCA45R: 0.09% Sodium Azide (NaN3)
- Carrier Free
- Yes
- Immunogen
- Rat thymocyte membrane glycoproteins.
- Approx. Protein Concentrations
- IgG concentration 1.0 mg/ml
- Fusion Partners
- Spleen cells from immunized BALB/c mice were fused with cells from the NS1 mouse myeloma cell line.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | 1/100 | ||
| Immunohistology - Frozen | |||
| Immunohistology - Paraffin |
- Flow Cytometry
- Use 10ul of the suggested working dilution to label 106 cells in 100ul
- Immunohistology
- This product does not require protein digestion pre-treatment of paraffin embedded sections e.g. trypsin or pronase prior to staining.
This product does not require antigen retrieval using heat treatment methods prior to staining of paraffin sections.
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG1 Negative Control | MCA1209 | F | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG1 Negative Control | ||||||
References for MHC Class II RT1Bu/L antibody
-
McMaster, W.R. & Williams, A.F. (1979) Identification of Ia glycoproteins in rat thymus and purification from rat spleen.
Eur J Immunol. 9 (6): 426-33. -
McMaster, W.R. & Williams, A.F. (1979) Monoclonal antibodies to Ia antigens from rat thymus: cross reactions with mouse and human and use in purification of rat Ia glycoproteins.
Immunol Rev. 47: 117-37. -
Barclay, A.N. & Mayrhofer, G. (1981) Bone marrow origin of Ia-positive cells in the medulla rat thymus.
J Exp Med. 153 (6): 1666-71. -
Zhang, J. et al. (1997) Expression of major histocompatibility complex molecules in rodent retina. Immunohistochemical study.
Invest Ophthalmol Vis Sci. 38 (9): 1848-57. -
Hahm, K.B. et al. (2000) Loss of TGF-beta signaling contributes to autoimmune pancreatitis.
J Clin Invest. 105 (8): 1057-65. -
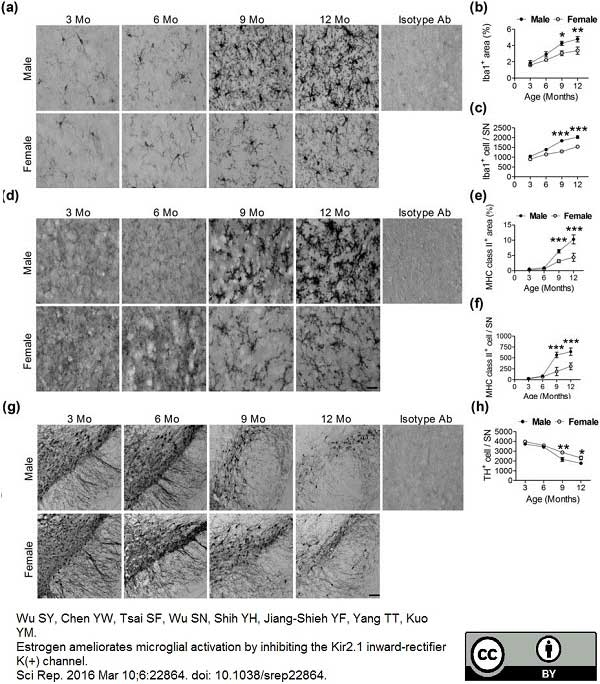
Wu, S.Y. et al. (2016) Estrogen ameliorates microglial activation by inhibiting the Kir2.1 inward-rectifier K(+) channel.
Sci Rep. 6: 22864. -
Fisher, R.A. et al. (1996) Induction of long-term graft tolerance and donor/recipient chimerism.
J Surg Res. 60 (1): 181-5. -
Keller, R. et al. (1988) Modulation of major histocompatibility complex (MHC) expression by interferons and microbial agents. Independent regulation of MHC class II expression and induction of tumoricidal activity in bone marrow-derived mononuclear phagocytes.
Scand J Immunol. 28 (1): 113-21.
View The Latest Product References
-
Streit, W.J. et al. (1989) Peripheral nerve lesion produces increased levels of major histocompatibility complex antigens in the central nervous system.
J Neuroimmunol. 21 (2-3): 117-23. -
Roggin, K.K. et al. (2001) Macrophage phenotype during cholestatic injury and repair: the persistent inflammatory response.
J Pediatr Surg. 36 (1): 220-8. -
Reutzel-Selke A et al. (2003) Short-term immunosuppressive treatment of the donor ameliorates consequences of ischemia/ reperfusion injury and long-term graft function in renal allografts from older donors.
Transplantation. 75 (11): 1786-92. -
Heidenhain, C. et al. (2003) The impact of immune-activating processes following transplantation on chronic allograft nephropathy.
Kidney Int. 64 (3): 1125-33. -
Hahm, K.B. et al. (2001) Loss of transforming growth factor beta signalling in the intestine contributes to tissue injury in inflammatory bowel disease.
Gut. 49 (2): 190-8. -
Pascher A et al. (2006) Rat cytomegalovirus infection interferes with anti-CD4 mAb-(RIB 5/2) mediated tolerance and induces chronic allograft damage.
Am J Transplant. 6 (9): 2035-45. -
Hartmann CB et al. (2005) Immunotoxicity of gallium arsenide on antigen presentation: comparative study of intratracheal and intraperitoneal exposure routes.
J Immunotoxicol. 2 (1): 1-9. -
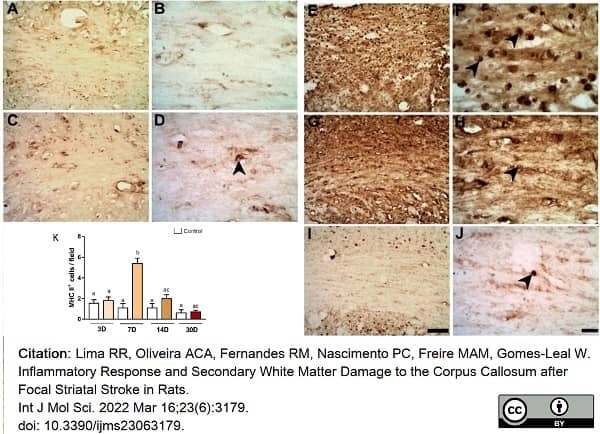
Lima, R.R. et al. (2022) Inflammatory Response and Secondary White Matter Damage to the Corpus Callosum after Focal Striatal Stroke in Rats.
Int J Mol Sci. 23 (6)Mar 16 [Epub ahead of print]. -
Duhalde Vega, M et al. (2022) PD-1/PD-L1 blockade abrogates a dysfunctional innate-adaptive immune axis in critical β-coronavirus disease.
Sci Adv. 8 (38): eabn6545.
Further Reading
-
Barclay, A.N. (1981) The localization of populations of lymphocytes defined by monoclonal antibodies in rat lymphoid tissues.
Immunology. 42 (4): 593-600.
- RRID
- AB_322406
MCA45GA
MCA45R
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Rat ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up