MHC Class I RT1A antibody | OX-18





Mouse anti Rat MHC Class I RT1A
- Product Type
- Monoclonal Antibody
- Clone
- OX-18
- Isotype
- IgG1
- Specificity
- MHC Class I RT1A
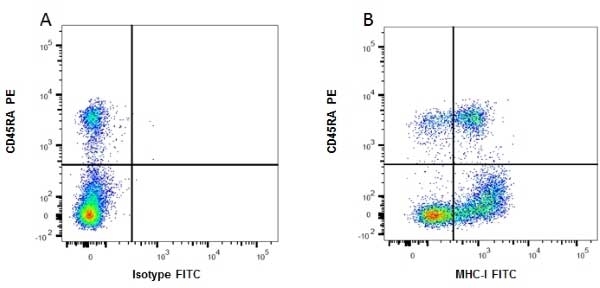
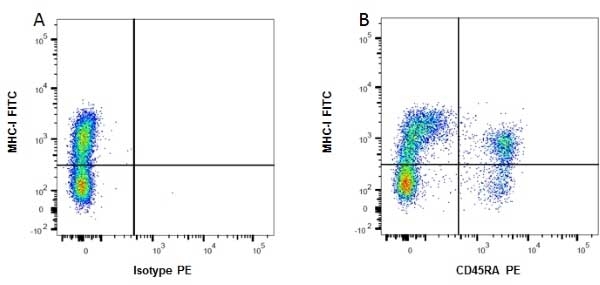
| Mouse anti Rat MHC Class I RT1A antibody, clone OX-18 recognizes a monomorphic determinant of rat MHC Class I (RT1A), expressed by all rat strains. However, quantitative measurements suggest that not all of the class I molecules are recognized. Mouse anti Rat MHC Class I RT1A antibody, clone OX-18 has been used in immunoaffinity purification of rat MHC class I molecules (Fukumoto et al. 1982). Mouse anti Rat MHC Class I RT1A antibody, clone OX-18 is routinely tested in flow cytometry on rat splenocytes. |
- Target Species
- Rat
- Product Form
- Purified IgG - liquid
- Preparation
- MCA51G: Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- MCA51GA, MCA51R: Purified IgG prepared by affinity chromatography on Protein G from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
0.09% Sodium Azide - Carrier Free
- Yes
- Immunogen
- Rat spleen cell glycoproteins
- Approx. Protein Concentrations
- IgG concentration 1.0 mg/ml
- Fusion Partners
- Spleen cells from immunised BALB/c mice were fused with cells of the mouse P3X63Ag8.653 myeloma cell line.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| ELISA | |||
| Flow Cytometry | 1/50 | 1/100 | |
| Immunohistology - Frozen 1 | |||
| Immunohistology - Paraffin | |||
| Immunoprecipitation |
- 1The epitope recognised by this antibody is reported to be sensitive to formaldehyde fixation and tissue processing. Bio-Rad recommends the use of acetone fixation for frozen sections.
- Flow Cytometry
- Use 10ul of the suggested working dilution to label 106 cells in 100ul.
- Immunohistology
- Acetone fixation recommended - the antigen is sensitive to fixation with paraformaldehyde.
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG1 Negative Control | MCA1209 | F | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG1 Negative Control | ||||||
References for MHC Class I RT1A antibody
-
Fukumoto, T. et al. (1982) Mouse monoclonal antibodies against rat major histocompatibility antigens. Two Ia antigens and expression of Ia and class I antigens in rat thymus.
Eur J Immunol. 12 (3): 237-43. -
Bukovský, A. et al. (1984) Association of some cell surface antigens of lymphoid cells and cell surface differentiation antigens with early rat pregnancy.
Immunology. 52: 631-40 -
Osawa, H. et al. (1985) Inhibition of IL 2-dependent proliferation of rat T lymphoblasts by the monoclonal antibody ART62 which reacts with MHC class 1 antigens.
J Immunol. 134 (6): 3901-6. -
Chacon, M.A. & Boulanger, L.M. (2013) MHC class I protein is expressed by neurons and neural progenitors in mid-gestation mouse brain.
Mol Cell Neurosci. 52: 117-27. -
Fujikawa, L.S. et al. (1989) Class II antigens on retinal vascular endothelium, pericytes, macrophages, and lymphocytes of the rat.
Invest Ophthalmol Vis Sci. 30 (1): 66-73. -
Zhai, Y. and Knechtle, S. et al. (1998) Two distinct forms of soluble MHC class I molecules synthesized by different mechanisms in normal rat cells in vitro
Human Immunol. 59: 404-14 -
Baca Jones, C.C. et al. (2009) Rat cytomegalovirus infection depletes MHC II in bone marrow derived dendritic cells.
Virology. 388: 78-90. -
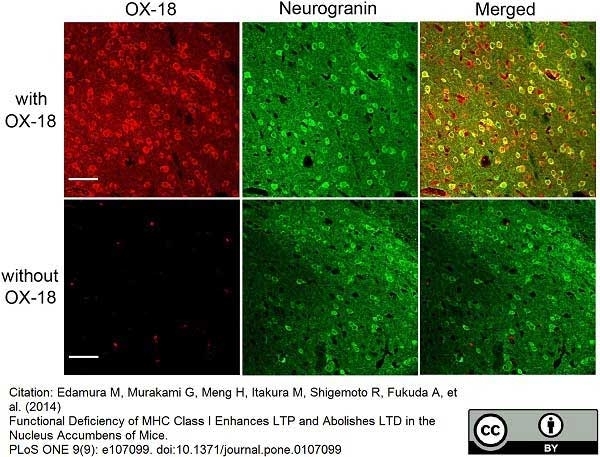
Edamura, M. et al. (2014) Functional deficiency of MHC class I enhances LTP and abolishes LTD in the nucleus accumbens of mice.
PLoS One. 9 (9): e107099.
View The Latest Product References
-
Cunningham, T.L. et al. (2014) Correlations between blood-brain barrier disruption and neuroinflammation in an experimental model of penetrating ballistic-like brain injury.
J Neurotrauma. 31 (5): 505-14. -
Skwirba, M. et al. (2014) Expression of nestin after renal transplantation in the rat.
APMIS. 122 (10): 1020-31. -
Schu, S. et al. (2012) Immunogenicity of allogeneic mesenchymal stem cells.
J Cell Mol Med. 16 (9): 2094-103. -
Dixon-Salazar, T.J. et al. (2014) MHC class I limits hippocampal synapse density by inhibiting neuronal insulin receptor signaling.
J Neurosci. 34 (35): 11844-56. -
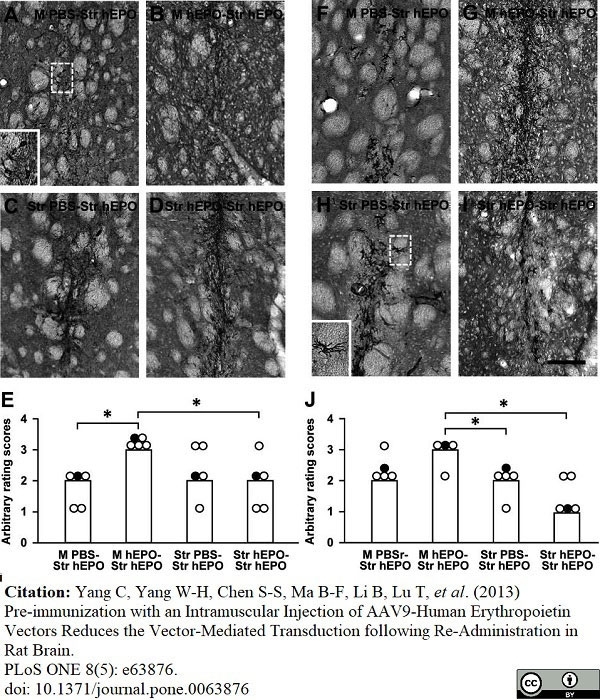
Yang, C. et al. (2013) Pre-immunization with an intramuscular injection of AAV9-human erythropoietin vectors reduces the vector-mediated transduction following re-administration in rat brain.
PLoS One. 8 (5): e63876. -
Elmer BM et al. (2013) MHCI requires MEF2 transcription factors to negatively regulate synapse density during development and in disease.
J Neurosci. 33 (34): 13791-804. -
Picarda E et al. (2014) MHC-derived allopeptide activates TCR-biased CD8+ Tregs and suppresses organ rejection.
J Clin Invest. 124 (6): 2497-512. -
Ma, R. et al. (2013) Structural integrity, ECM components and immunogenicity of decellularized laryngeal scaffold with preserved cartilage.
Biomaterials. 34 (7): 1790-8. -
Lu, X.C. et al. (2015) Dual Therapeutic Effects of C-10068, a Dextromethorphan Derivative, Against Post-Traumatic Nonconvulsive Seizures and Neuroinflammation in a Rat Model of Penetrating Ballistic-Like Brain Injury.
J Neurotrauma. 32 (20): 1621-32. -
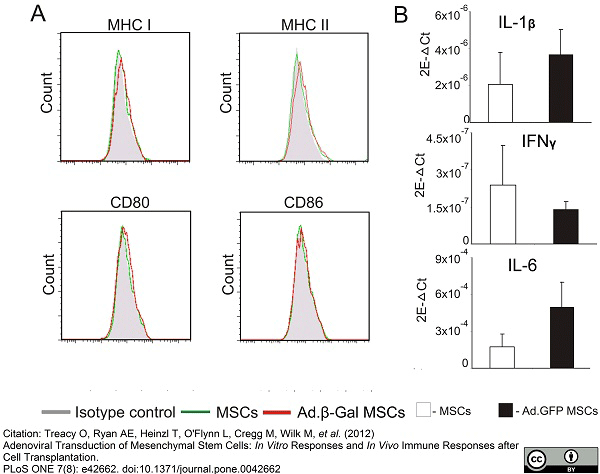
Treacy, O. et al. (2012) Adenoviral transduction of mesenchymal stem cells: in vitro responses and in vivo immune responses after cell transplantation.
PLoS One. 7 (8): e42662. -
Inácio, R.F. et al. (2012) Interferon beta modulates major histocompatibility complex class I (MHC I) and CD3-zeta expression in PC12 cells.
Neurosci Lett. 513 (2): 223-8. -
Yang, Y.M. et al. (2013) Microglial TNF-α-dependent elevation of MHC class I expression on brain endothelium induced by amyloid-beta promotes T cell transendothelial migration.
Neurochem Res. 38 (11): 2295-304. -
Zhang, J.et al. (2017) Changes in Expressions of Major Histocompatibility Complex Class I, Paired-Immunoglobulin-Like Receptor B, and Cluster of Differentiation 3ζ in Motor Cortical Representations of the Brachial Plexus After Avulsion in Rats.
World Neurosurg. 106: 211-8. -
Otto, C. et al. (2012) Immunisation with an allogeneic peptide promotes the induction of antigen-specific MHC II(pos) CD4+ rat T cells demonstrating immunostimulatory properties.
Transpl Immunol. 26 (4): 220-9. -
Coiro, P. et al. (2015) Impaired synaptic development in a maternal immune activation mouse model of neurodevelopmental disorders.
Brain Behav Immun. pii: S0889-1591(15)00417-1. -
Bombeiro, A.L. et al. (2017) Importance of major histocompatibility complex of class I (MHC-I) expression for astroglial reactivity and stability of neural circuits in vitro.
Neurosci Lett. 647: 97-103. -
Iinuma, C. et al. (2015) Establishment of a vascular endothelial cell-reactive type II NKT cell clone from a rat model of autoimmune vasculitis.
Int Immunol. 27 (2): 105-14. -
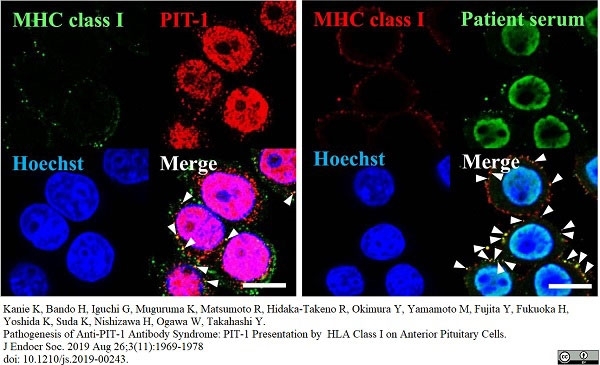
Kanie, K. et al. (2019) Pathogenesis of Anti-PIT-1 Antibody Syndrome: PIT-1 Presentation by HLA Class I on Anterior Pituitary Cells.
J Endocr Soc. 3 (11): 1969-78. -
Li, P. et al. (2020) Neuronal NLRC5 regulates MHC class I expression in Neuro-2a cells and also during hippocampal development.
J Neurochem. 152 (2): 182-94.
- RRID
- AB_322393
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Rat ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up