SLA Class II DQ antibody | K274.3G8
Mouse anti Pig SLA Class II DQ
- Product Type
- Monoclonal Antibody
- Clone
- K274.3G8
- Isotype
- IgG1
- Specificity
- SLA Class II DQ
Filter by Application:
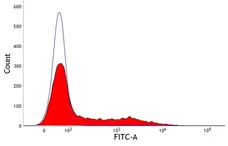
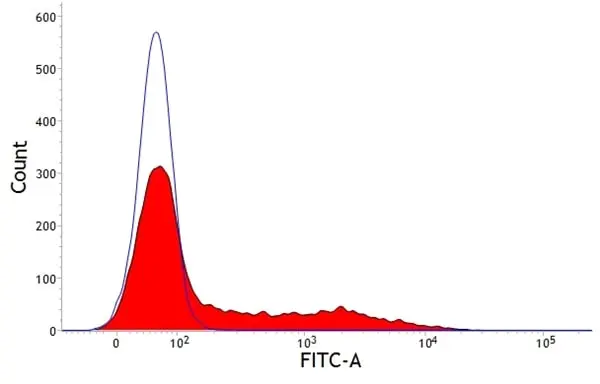
F Reset| Mouse anti Pig SLA Class II DQ antibody, clone K274.3G8 recognizes SLA DQ molecules which are expressed on all B cells, antigen presenting cells and on certain subsets of resting and activated T cells. The major histocompatibility complex (MHC) is a cluster of genes that are important in the immune response to infections. In pigs, this is referred to as the swine leukocyte antigen (SLA) region. There are 3 major MHC class II proteins encoded by the SLA which are SLA DP, SLA DQ and SLA DR. |
- Target Species
- Pig
- Species Cross-Reactivity
-
Target Species Cross Reactivity Bovine - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% sodium azide (NaN3)
- Carrier Free
- Yes
- Immunogen
- Porcine peripheral blood lymphocytes
- Approx. Protein Concentrations
- IgG concentration 1.0 mg/ml
- Fusion Partners
- Spleen cells from immunized mice were fused with cells of the P3-X63-Ag.653 myeloma cell line
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | 1/10 | 1/100 | |
| Immunohistology - Frozen | |||
| Immunohistology - Paraffin |
- Flow Cytometry
- Use 10μl of the suggested working dilution to label 1x106 cells in 100μl
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG1 Negative Control | MCA928 | F | 100 Tests |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG1 Negative Control | ||||||
References for SLA Class II DQ antibody
-
Brodersen, R. et al. (1998) Analysis of the immunological cross reactivities of 213 well characterized monoclonal antibodies with specificities against various leucocyte surface antigens of human and 11 animal species.
Vet Immunol Immunopathol. 64: 1-13. -
Paillot, R. et al. (2001) Functional and phenotypic characterization of distinct porcine dendritic cells derived from peripheral blood monocytes.
Immunology 102: 396-404. -
Makala, L.H. et al. (2001) Ontogeny of pig discrete Peyer's patches: expression of surface antigens.
J Vet Med Sci. 63 (6): 625-36. -
Faure, J.P. et al. (2002) Polyethylene glycol reduces early and long-term cold ischemia-reperfusion and renal medulla injury.
J Pharmacol Exp Ther. 302: 861-70. -
Hauet, T. et al. (2002) Polyethylene glycol reduces the inflammatory injury due to cold ischemia/reperfusion in autotransplanted pig kidneys.
Kidney Int. 62: 654-67. -
Yang, P. et al. (2002) Immune cells in the porcine retina: distribution, characterization and morphological features.
Invest Ophthalmol Vis Sci. 43: 1488-92. -
Sarradell, J. et al. (2003) A morphologic and immunohistochemical study of the bronchus-associated lymphoid tissue of pigs naturally infected with Mycoplasma hyopneumoniae.
Vet Pathol. 40: 395-404. -
Edamura, K. et al. (2005) Effect of long-term culture on the expression of antigens and adhesion molecule in single porcine pancreatic endocrine cells.
Xenotransplantation. 12 (4): 327-32.
View The Latest Product References
-
Maasilta, P.K. et al. (2005) Immune cells in a heterotopic lamb-to-pig bronchial xenograft model.
Transpl Int. 18: 1100-8. -
Jayle, C. et al. (2007) Comparison of protective effects of trimetazidine against experimental warm ischemia of different durations: early and long-term effects in a pig kidney model.
Am J Physiol Renal Physiol. 292: F1082-93. -
Park, J.Y. et al. (2008) Characterization of interaction between porcine reproductive and respiratory syndrome virus and porcine dendritic cells.
J Microbiol Biotechnol. 18: 1709-16. -
Facci, M.R. et al. (2010) A comparison between isolated blood dendritic cells and monocyte-derived dendritic cells in pigs.
Immunology. 129 (3): 396-405. -
Inman, C.F. et al. (2010) Dendritic cells interact with CD4 T cells in intestinal mucosa.
J Leukoc Biol. 88: 571-8. -
Weesendorp E et al. (2013) Phenotypic modulation and cytokine profiles of antigen presenting cells by European subtype 1 and 3 porcine reproductive and respiratory syndrome virus strains in vitro and in vivo.
Vet Microbiol. 167 (3-4): 638-50. -
Debeer, S. et al. (2013) Comparative histology and immunohistochemistry of porcine versus human skin.
Eur J Dermatol. 23 (4): 456-66. -
LeLuduec, J.B. et al. (2016) Intradermal vaccination with un-adjuvanted sub-unit vaccines triggers skin innate immunity and confers protective respiratory immunity in domestic swine.
Vaccine. 34 (7): 914-22. -
Vreman, S. et al. (2018) Neonatal porcine blood derived dendritic cell subsets show activation after TLR2 or TLR9 stimulation.
Dev Comp Immunol. 84: 361-70. -
Loss, H. et al. (2018) Effects of a pathogenic ETEC strain and a probiotic Enterococcus faecium strain on the inflammasome response in porcine dendritic cells.
Vet Immunol Immunopathol. 203: 78-87. -
Ladowski, J.M. et al. (2018) Swine Leukocyte Antigen Class II Is a Xenoantigen.
Transplantation. 102 (2): 249-54.
Further Reading
-
Piriou-Guzylack, L. (2008) Membrane markers of the immune cells in swine: an update.
Vet Res. 39: 54.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Pig ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up