CD11R1 antibody | MIL4






Mouse anti Pig CD11R1
- Product Type
- Monoclonal Antibody
- Clone
- MIL4
- Isotype
- IgG1
- Specificity
- CD11R1
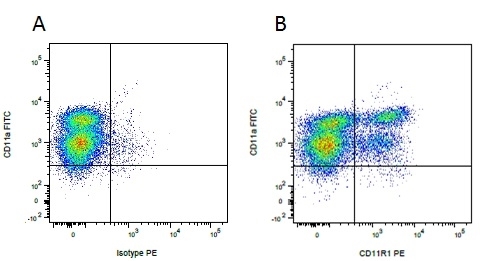
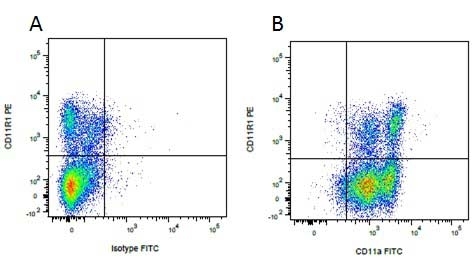
| Mouse anti Pig CD11R1, clone MIL4 recognizes the porcine cell surface antigen classified as CD11R1 at the Third International Workshop on Swine Leukocyte Differentiation Antigens (Haverson et al. 2001). Mouse anti Pig CD11R1, clone MIL4 stains porcine eosinophils, a subset of neutrophils and NK cells, it does not stain monocytes or macrophages (Haverson et al. 1994). Mouse anti Pig CD11R1, clone MIL4 immunoprecipitates a band corresponding to integrin β2 (CD18) of ~95kDa, in common with all other anti CD11 antibodies tested at the workshop and also a band of ~165 kDa corresponding to CD11R1, in a manner identical to the cross reactive anti human CD11b clone, TMG6-5 from peripheral blood mononuclear cell lysates suggesting that porcine CD11R1 is analogous to human CD11b (Dominguez et al. 2001) Mouse anti pig CD11R1, clone MIL4 is cross reactive with the guinea pig and is useful for the identification of a population of guinea pig natural killer cells, Kurloff cells (Takizawa et al. 2004) (Eremin et al. 1980). |
- Target Species
- Pig
- Species Cross-Reactivity
-
Target Species Cross Reactivity Human Guinea Pig - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% sodium azide (NaN3)
- Carrier Free
- Yes
- Immunogen
- Porcine Lamina Propria Leucocytes.
- Approx. Protein Concentrations
- IgG concentration 1 mg/ml
- Fusion Partners
- Spleen cells from immunized BALB/c mice were fused with cells of the P3 - X63 - Ag.653 myeloma cell line.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | 1/25 | 1/200 | |
| Immunohistology - Frozen | |||
| Immunoprecipitation |
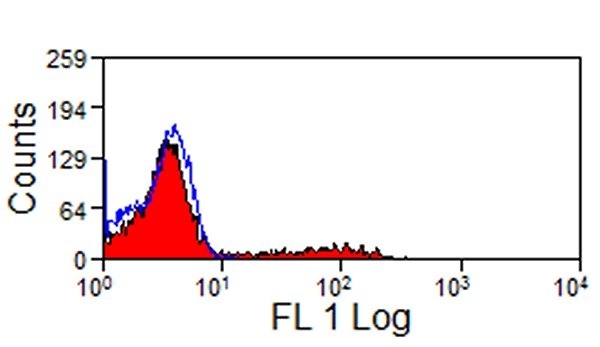
- Flow Cytometry
- Use 10μl of the suggested working dilution to label 1x106 cells in 100μl
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG1 Negative Control | MCA928 | F | 100 Tests |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG1 Negative Control | ||||||
References for CD11R1 antibody
-
Haverson, K. et al. (1994) Characterization of monoclonal antibodies specific for monocytes, macrophages and granulocytes from porcine peripheral blood and mucosal tissues.
J Immunol Methods. 170 (2): 233-45. -
Domínguez, J. et al. (2001) Workshop studies on monoclonal antibodies in the myeloid panel with CD11 specificity.
Vet Immunol Immunopathol. 80 (1-2): 111-9. -
Ordway, D.et al. (2007) The cellular immune response to Mycobacterium tuberculosis infection in the guinea pig.
J Immunol. 179: 2532-41. -
Inman, C.F. et al. (2010) Dendritic cells interact with CD4 T cells in intestinal mucosa.
J Leukoc Biol. 88: 571-8. -
Cheng, Q. et al. (2010) Administered CpG oligodeoxynucleotide induces mRNA expression of CXC and CC chemokines at the intestinal mucosa and PBMCs in piglets.
Int Immunopharmacol. 10: 611-8. -
Shang, S. et al. (2011) Activities of TMC207, rifampin, and pyrazinamide against Mycobacterium tuberculosis infection in guinea pigs.
Antimicrob Agents Chemother. 55 (1): 124-31. -
Rank, R.G. et al. (2012) Effect of Inflammatory Response on In Vivo Competition between Two Chlamydial Variants in the Guinea Pig Model of Inclusion Conjunctivitis.
Infect Immun. 80: 612-9. -
Suda, Y. et al. (2014) Immunobiotic Lactobacillus jensenii as immune-health promoting factor to improve growth performance and productivity in post-weaning pigs.
BMC Immunol. 15: 24.
View The Latest Product References
-
Shegarfi, H. et al. (2015) Regulation of CCN1 (Cyr61) in a porcine model of intestinal ischemia/reperfusion.
Innate Immun. 21 (5): 453-62. -
Yeruva, L. et al. (2015) Chlamydial variants differ in ability to ascend the genital tract in the guinea pig model of chlamydial genital infection.
Infect Immun. 83 (8): 3176-83. -
Auray, G. et al. (2016) Characterization and Transcriptomic Analysis of Porcine Blood Conventional and Plasmacytoid Dendritic Cells Reveals Striking Species-Specific Differences.
J Immunol. 197 (12): 4791-806. -
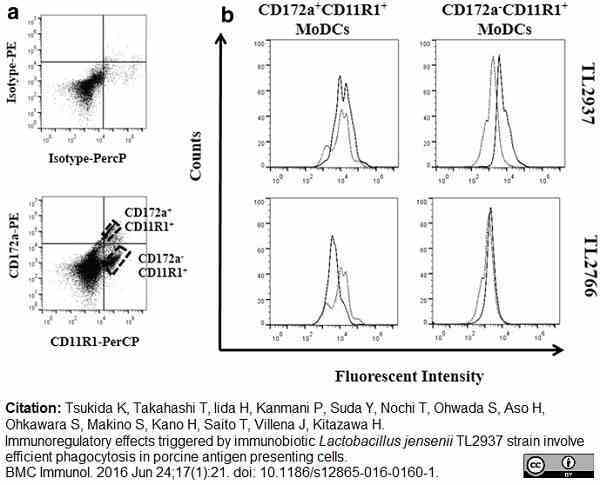
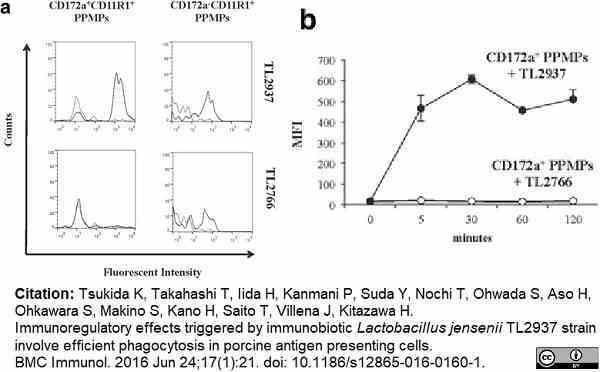
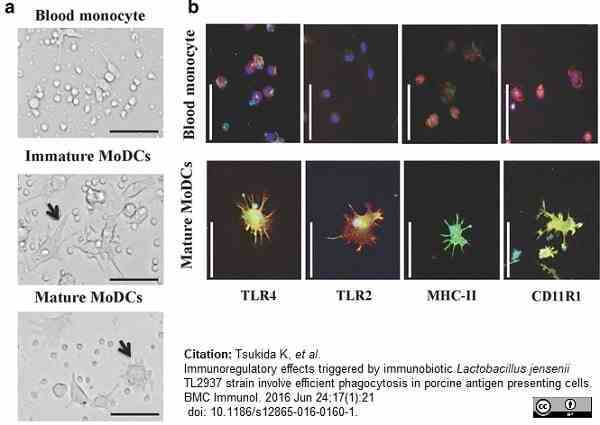
Tsukida, K. et al. (2016) Immunoregulatory effects triggered by immunobiotic Lactobacillus jensenii TL2937 strain involve efficient phagocytosis in porcine antigen presenting cells.
BMC Immunol. 17 (1): 21. -
Sautter, C.A. et al. (2018) Phenotypic and functional modulations of porcine macrophages by interferons and interleukin-4.
Dev Comp Immunol. 84: 181-92. -
Iida, H. et al. (2019) Paraimmunobiotic Bifidobacteria Modulate the Expression Patterns of Peptidoglycan Recognition Proteins in Porcine Intestinal Epitheliocytes and Antigen Presenting Cells.
Cells. 8(8):891. -
Ferret-Bernard, S. et al. (2020) Maternal Supplementation of Food Ingredient (Prebiotic) or Food Contaminant (Mycotoxin) Influences Mucosal Immune System in Piglets.
Nutrients. 12 (7): 2115. -
Álvarez, B. et al. (2023) Porcine Macrophage Markers and Populations: An Update.
Cells. 12 (16): 2103.
Further Reading
-
Piriou-Guzylack, L. (2008) Membrane markers of the immune cells in swine: an update.
Vet Res. 39: 54.
MCA1220GA
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Pig ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up