Fibroblasts/Epithelial Cells antibody | D7-FIB

Mouse anti Human Fibroblasts/Epithelial Cells
- Product Type
- Monoclonal Antibody
- Clone
- D7-FIB
- Isotype
- IgG2a
- Specificity
- Fibroblasts/Epithelial Cells
| Mouse anti Human fibroblasts/epithelial cells antibody, clone D7-FIB recognizes a ~112kDa molecule expressed on the cell surface of human fibroblasts. The antigen is not expressed by peripheral blood cells, and is found at low levels on a minority of melanoma cell lines. The epitope recognized by clone D7-FIB is sensitive to SDS, but resistant to trypsin, tunicamycin and collagenase. Mouse anti Human Fibroblasts/Epithelial Cells antibody, clone D7-FIB also binds to epithelium, myoepthelium, smooth muscle and some leucocytes. Mouse anti Human Fibroblasts/Epithelial Cells antibody, clone D7-FIB is useful as a cell membrane marker to characterize chondrocyte differentiation giving a positive reaction with dedifferentiated human chondrocytes, and negative with differentiated chondrocytes (van Osch et al. 2001). Mouse anti Human Fibroblasts/Epithelial Cells antibody, clone D7-FIB is routinely tested in flow cytometry on the KG1 cell line. |
- Target Species
- Human
- Species Cross-Reactivity
-
Target Species Cross Reactivity Rat Mouse - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG - liquid
- Preparation
- MCA1399G: Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- MCA1399GT: Purified IgG prepared by affinity chromatography on Protein G from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% sodium azide (NaN3)
- Carrier Free
- Yes
- Immunogen
- Human foreskin fibroblasts.
- Approx. Protein Concentrations
- IgG concentration 1.0 mg/ml
- Fusion Partners
- Spleen cells from immunised BALB/c mice were fused with cells of the mouse SP2 myeloma cell line.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
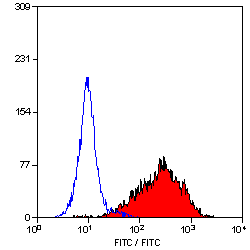
| Flow Cytometry | 1/50 | 1/200 | |
| Immunohistology - Frozen 1 | 1/100 | ||
| Immunohistology - Paraffin | |||
| Immunoprecipitation |
- 1The epitope recognised by this antibody is reported to be sensitive to formaldehyde fixation and tissue processing. Bio-Rad recommends the use of acetone fixation for frozen sections.
- Flow Cytometry
- Use 10μl of the suggested working dilution to label 106 cells in 100μl
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG2a Negative Control | MCA929 | F | 100 Tests |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG2a Negative Control | ||||||
References for Fibroblasts/Epithelial Cells antibody
-
Fearns C & Dowdle EB (1992) The desmoplastic response: induction of collagen synthesis by melanoma cells in vitro.
Int J Cancer. 50 (4): 621-7. -
Jayne, D.G. et al. (1999) A three-dimensional in-vitro model for the study of peritoneal tumour metastasis.
Clin Exp Metastasis. 17 (6): 515-23. -
Kelynack, K.J. et al. (2000) Human renal fibroblast contraction of collagen I lattices is an integrin-mediated process.
Nephrol Dial Transplant. 15 (11): 1766-72. -
Todisco, E. et al. (2002) CD40 ligand-stimulated B cell precursor leukemic cells elicit interferon-gamma production by autologous bone marrow T cells in childhood acute lymphoblastic leukemia.
Leukemia. 16 (10): 2046-54. -
Jones. E.A. et al. (2002) Isolation and characterization of bone marrow multipotential mesenchymal progenitor cells.
Arthritis. Rheum. 46: 3349-60. -
Telfer, J.F. and Brock, J.H. (2002) Expression of ferritin, transferrin receptor, and non-specific resistance associated macrophage proteins 1 and 2 (Nramp1 and Nramp2) in the human rheumatoid synovium.
Ann Rheum Dis. 61: 741-4. -
Petrow, P.K. et al. (2002) Characterisation of the cell type-specificity of collagenase 3 mRNA expression in comparison with membrane type 1 matrix metalloproteinase and gelatinase A in the synovial membrane in rheumatoid arthritis.
Ann Rheum Dis. 61: 391-7. -
Pap, T. et al. (2003) Osteoclast-independent bone resorption by fibroblast-like cells
Arthritis Res Ther. 5: R163-73.
View The Latest Product References
-
Miranda-Carús, M.E. et al. (2004) Rheumatoid arthritis synovial fluid fibroblasts express TRAIL-R2 (DR5) that is functionally active.
Arthritis Rheum. 50: 2786-93. -
Manoussaka, M.S. et al. (2005) Flow cytometric characterisation of cells of differing densities isolated from human term placentae and enrichment of villous trophoblast cells.
Placenta. 26 (4): 308-18. -
De Bari, C. et al. (2006) Mesenchymal multipotency of adult human periosteal cells demonstrated by single-cell lineage analysis.
Arthritis Rheum. 54 (4): 1209-21. -
Jones, E.A. et al. (2006) Optimization of a flow cytometry-based protocol for detection and phenotypic characterization of multipotent mesenchymal stromal cells from human bone marrow.
Cytometry B Clin Cytom. 70: 391-9. -
English, A. et al. (2007) A comparative assessment of cartilage and joint fat pad as a potential source of cells for autologous therapy development in knee osteoarthritis.
Rheumatology (Oxford). 46: 1676-83. -
Martinez, C. et al. (2007) Human bone marrow mesenchymal stromal cells express the neural ganglioside GD2: a novel surface marker for the identification of MSCs.
Blood. 109 (10): 4245-8. -
Morito, T. et al. (2008) Synovial fluid-derived mesenchymal stem cells increase after intra-articular ligament injury in humans.
Rheumatology (Oxford). 47 (8): 1137-43. -
Nimura, A. et al. (2008) Increased proliferation of human synovial mesenchymal stem cells with autologous human serum: comparisons with bone marrow mesenchymal stem cells and with fetal bovine serum.
Arthritis Rheum. 58: 501-10. -
Scutt, N. et al. (2008) Tissue specific characteristics of cells isolated from human and rat tendons and ligaments.
J Orthop Surg Res. 3: 32. -
De Bari, C. et al. (2008) A biomarker-based mathematical model to predict bone-forming potency of human synovial and periosteal mesenchymal stem cells.
Arthritis Rheum. 58 (1): 240-50. -
Kanayama, M. et al. (2009) Alpha9 integrin and its ligands constitute critical joint microenvironments for development of autoimmune arthritis.
J Immunol. 182: 8015-25. -
Pountos, I. et al. (2011) NSAIDS inhibit in vitro MSC chondrogenesis but not osteogenesis: implications for mechanism of bone formation inhibition in man.
J Cell Mol Med. 15: 525-34. -
Sekiya, I. et al. (2012) Human mesenchymal stem cells in synovial fluid increase in the knee with degenerated cartilage and osteoarthritis.
J Orthop Res. 30: 943-9. -
Behl, B. et al. (2013) Biological effects of cobalt-chromium nanoparticles and ions on dural fibroblasts and dural epithelial cells.
Biomaterials. 34 (14): 3547-58. -
Asano, T. et al. (2014) α9β1 integrin acts as a critical intrinsic regulator of human rheumatoid arthritis.
Rheumatology (Oxford). 53 (3): 415-24. -
Papageorgiou, I. et al. (2014) Interaction of micron and nano-sized particles with cells of the dura mater.
J Biomed Mater Res B Appl Biomater. 102 (7): 1496-505. -
Iyyanki, T. et al. (2015) Harvesting technique affects adipose-derived stem cell yield.
Aesthet Surg J. 35 (4): 467-76. -
Schminke, B. et al. (2015) The pathology of bone tissue during peri-implantitis.
J Dent Res. 94 (2): 354-61. -
Grognuz, A. et al. (2016) Human Fetal Progenitor Tenocytes for Regenerative Medicine.
Cell Transplant. 25 (3): 463-79. -
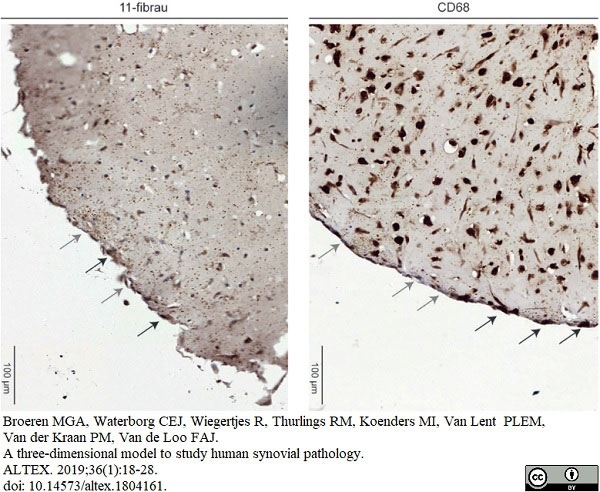
Broeren, M.G.A. (2019) A three-dimensional model to study human synovial pathology
ALTEX. 36 (1): 18-28.
- RRID
- AB_2180567
MCA1399G
MCA1399GT
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Human ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up