CD90 antibody | F15-42-1










Mouse anti Human CD90
- Product Type
- Monoclonal Antibody
- Clone
- F15-42-1
- Isotype
- IgG1
- Specificity
- CD90
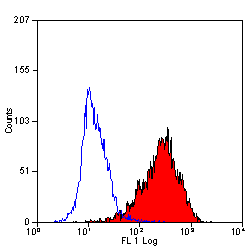
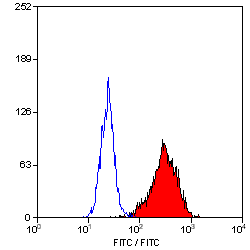
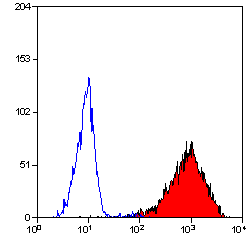
| Mouse anti Human CD90 antibody, clone F15-42-1 recognizes the human CD90 cell surface antigen, a ~25 kDa glycoprotein homologous to rat Thy1. The antigen is expressed by a subset of CD34+ve cells in the bone marrow and by prothymocytes within the thymus. CD90 is also expressed extensively within the brain. Mouse anti Human CD90 antibody, clone F15-42-1 is routinely tested in flow cytometry on the MOLT4 cell line. |
- Target Species
- Human
- Species Cross-Reactivity
-
Target Species Cross Reactivity Cynomolgus monkey - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG - liquid
- Preparation
- MCA90T: Purified IgG prepared by ion exchange chromatography from tissue culture supernatant
- MCA90: Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
0.09% Sodium Azide - Carrier Free
- Yes
- Immunogen
- Purified human brain Thy-1.
- Approx. Protein Concentrations
- IgG concentration 1.0 mg/ml
- Fusion Partners
- Spleen cells from immunized BALB/c mice were fused with cells of the mouse NS-1 myeloma cell line.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | 1/10 | 1/50 | |
| Immunofluorescence | |||
| Immunohistology - Frozen 1 | |||
| Immunohistology - Paraffin | |||
| Immunoprecipitation |
- 1The epitope recognised by this antibody is reported to be sensitive to formaldehyde fixation and tissue processing. Bio-Rad recommends the use of acetone fixation for frozen sections.
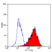
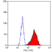
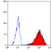
- Flow Cytometry
- Use 10ul of the suggested working dilution to label 106 cells in 100ul.
- Histology Positive Control Tissue
- Human Brain, thymus
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG1 Negative Control | MCA928 | F | 100 Tests |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG1 Negative Control | ||||||
Source Reference
-
McKenzie, J.L. & Fabre, J.W. (1981) Human thy-1: unusual localization and possible functional significance in lymphoid tissues.
J Immunol. 126 (3): 843-50.
References for CD90 antibody
-
Daar, A.S. & Fabre, J.W. (1981) Demonstration with monoclonal antibodies of an unusual mononuclear cell infiltrate and loss of normal epithelial membrane antigens in human breast carcinomas.
Lancet. 2 (8244): 434-8. -
Fiegel, H.C. et al. (2004) Stem-like cells in human hepatoblastoma.
J Histochem Cytochem. 52 (11): 1495-501. -
Hagood, J.S. et al. (2005) Loss of fibroblast Thy-1 expression correlates with lung fibrogenesis.
Am J Pathol. 167 (2): 365-79. -
Tome, M. et al. (2007) Calponin is expressed by subpopulations of connective tissue cells but not olfactory ensheathing cells in the neonatal olfactory mucosa.
BMC Neurosci. 8: 74. -
Diaz-Romero, J. et al. (2008) Immunophenotypic changes of human articular chondrocytes during monolayer culture reflect bona fide dedifferentiation rather than amplification of progenitor cells.
J Cell Physiol. 214: 75-83. -
Pessina, A. et al. (2010) CD45+/CD133+ positive cells expanded from umbilical cord blood expressing PDX-1 and markers of pluripotency.
Cell Biol Int. 34: 783-90. -
Manochantr, S. et al. (2010) Isolation, characterization and neural differentiation potential of amnion derived mesenchymal stem cells.
J Med Assoc Thai. 93 Suppl 7: S183-91. -
Karlsen, T.A. et al. (2010) Human primary articular chondrocytes, chondroblasts-like cells, and dedifferentiated chondrocytes: differences in gene, microRNA, and protein expression and phenotype.
Tissue Eng Part C Methods. 17: 219-27.
View The Latest Product References
-
Hauser, P.V. et al. (2010) Stem cells derived from human amniotic fluid contribute to acute kidney injury recovery.
Am J Pathol. 177: 2011-21. -
Yin, S. et al. (2010) Chondrogenic transdifferentiation of human dermal fibroblasts stimulated with cartilage-derived morphogenetic protein 1.
Tissue Eng Part A. 16: 1633-43. -
Gieseke, F. et al. (2010) Human multipotent mesenchymal stromal cells use galectin-1 to inhibit immune effector cells.
Blood. 116: 3770-9. -
Holzwarth, C. et al. (2010) Low physiologic oxygen tensions reduce proliferation and differentiation of human multipotent mesenchymal stromal cells.
BMC Cell Biol. 11:11 -
Meng, J. et al (2011) Contribution of human muscle-derived cells to skeletal muscle regeneration in dystrophic host mice.
PLoS One. 6: e17454. -
Cizeau, J. et al. (2011) Fusogenics: a recombinant immunotoxin-based screening platform to select internalizing tumor-specific antibody fragments.
J Biomol Screen. 16: 90-100. -
Cox, G. et al. (2011) The use of the reamer-irrigator-aspirator to harvest mesenchymal stem cells.
J Bone Joint Surg Br. 93: 517-24. -
Shafaei, H. et al. (2011) Effects of human placental serum on proliferation and morphology of human adipose tissue-derived stem cells.
Bone Marrow Transplant. 46: 1464-71. -
Paul, G. et al. (2012) The adult human brain harbors multipotent perivascular mesenchymal stem cells.
PLoS One. 7: e35577. -
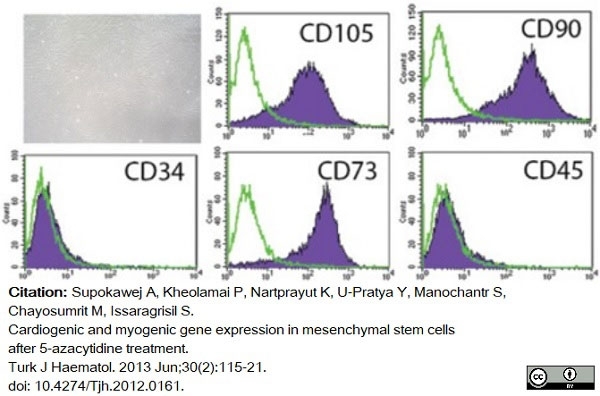
Supokawej, A. et al. (2013) Cardiogenic and myogenic gene expression in mesenchymal stem cells after 5-azacytidine treatment.
Turk J Haematol. 30 (2): 115-21. -
Escobar, C.H. & Chaparro, O. (2016) Xeno-Free Extraction, Culture, and Cryopreservation of Human Adipose-Derived Mesenchymal Stem Cells.
Stem Cells Transl Med. 5 (3): 358-65. -
Shinoda, K. et al. (2016) Thy1+IL-7+ lymphatic endothelial cells in iBALT provide a survival niche for memory T-helper cells in allergic airway inflammation.
Proc Natl Acad Sci U S A. 113 (20): E2842-51. -
Kamprom, W. et al. (2016) Endothelial Progenitor Cell Migration-Enhancing Factors in the Secretome of Placental-Derived Mesenchymal Stem Cells.
Stem Cells Int. 2016: 2514326. -
Vaquero, J. et al. (2016) An approach to personalized cell therapy in chronic complete paraplegia: The Puerta de Hierro phase I/II clinical trial.
Cytotherapy. 18 (8): 1025-36. -
Zhang, X. et al. (2017) Regeneration of hyaline-like cartilage in situ with SOX9 stimulation of bone marrow-derived mesenchymal stem cells.
PLoS One. 12 (6): e0180138. -
Garikipati, V. N.S. et al. (2018) Isolation and characterization of mesenchymal stem cells from human fetus heart.
PLoS One. 13 (2): e0192244. -
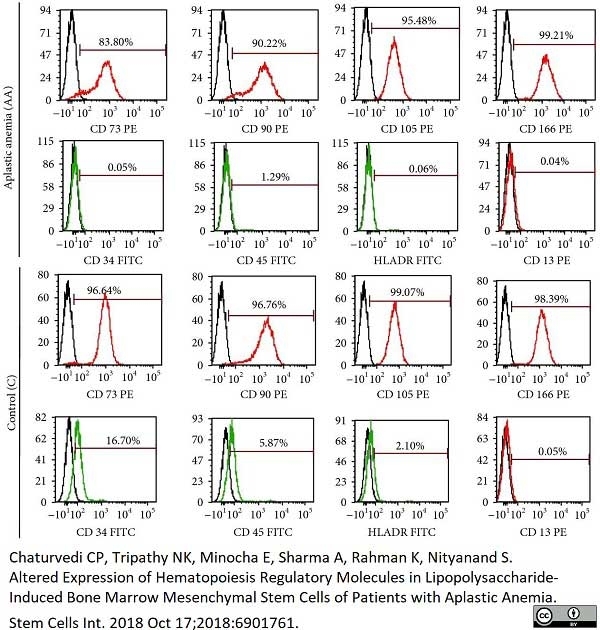
Chaturvedi, C.P. et al. (2018) Altered Expression of Hematopoiesis Regulatory Molecules in Lipopolysaccharide-Induced Bone Marrow Mesenchymal Stem Cells of Patients with Aplastic Anemia.
Stem Cells Int. 2018: 6901761. -
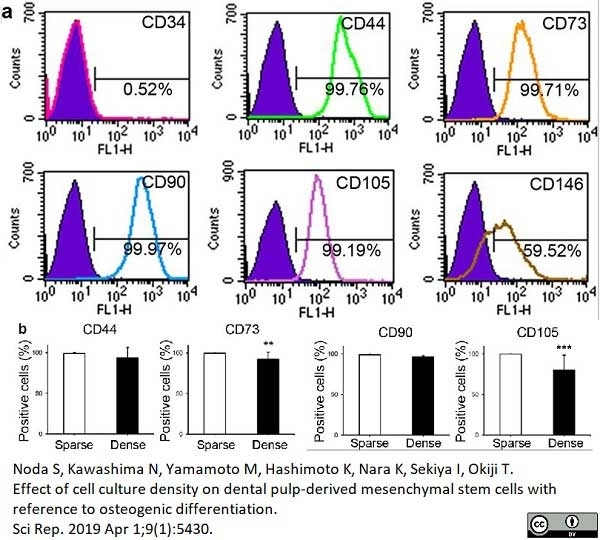
Noda, S. et al. (2019) Effect of cell culture density on dental pulp-derived mesenchymal stem cells with reference to osteogenic differentiation.
Sci Rep. 9 (1): 5430. -
Song, H. et al. (2019) MIF/CD74 axis participates in inflammatory activation of Schwann cells following sciatic nerve injury.
J Mol Histol. 50 (4): 355-67. -
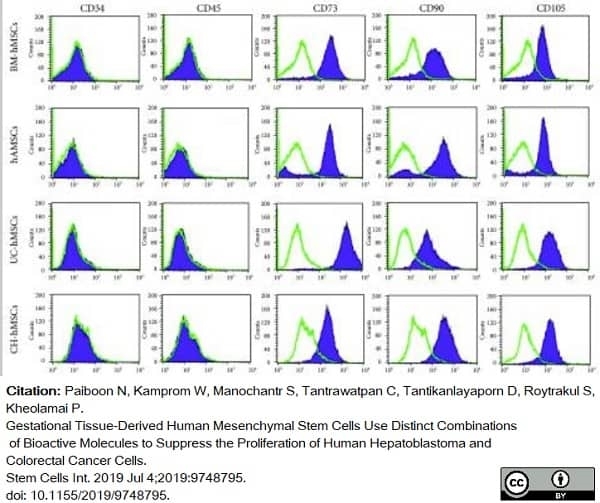
Paiboon, N. et al. (2019) Gestational Tissue-Derived Human Mesenchymal Stem Cells Use Distinct Combinations of Bioactive Molecules to Suppress the Proliferation of Human Hepatoblastoma and Colorectal Cancer Cells.
Stem Cells Int. 2019: 9748795. -
Sanjurjo-Rodriguez, C. et al. (2020) Gene Expression Signatures of Synovial Fluid Multipotent Stromal Cells in Advanced Knee Osteoarthritis and Following Knee Joint Distraction.
Front Bioeng Biotechnol. 8: 579751. -
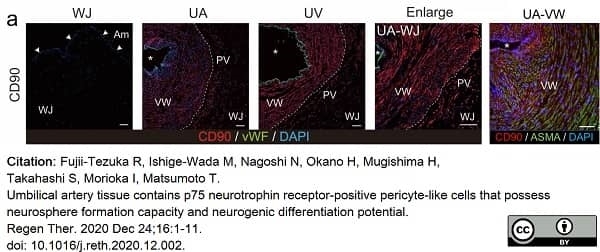
Fujii-Tezuka, R. et al. (2021) Umbilical artery tissue contains p75 neurotrophin receptor-positive pericyte-like cells that possess neurosphere formation capacity and neurogenic differentiation potential.
Regen Ther. 16: 1-11. -
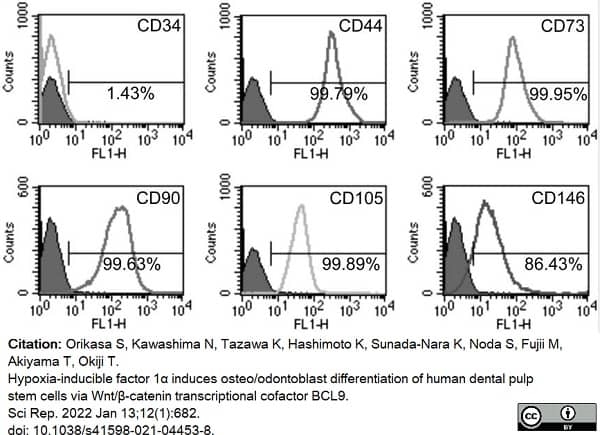
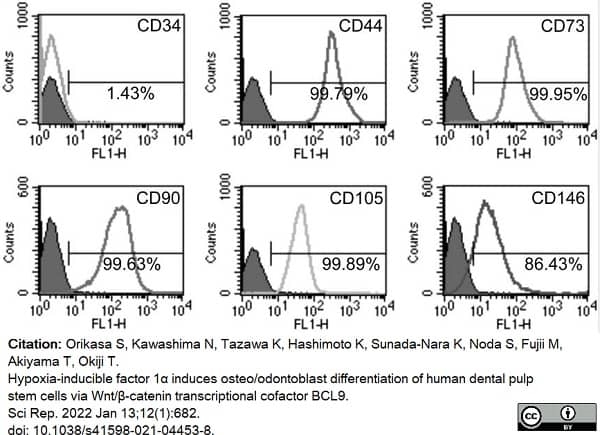
Orikasa, S. et al. (2022) Hypoxia-inducible factor 1α induces osteo/odontoblast differentiation of human dental pulp stem cells via Wnt/β-catenin transcriptional cofactor BCL9.
Sci Rep. 12 (1): 682. -
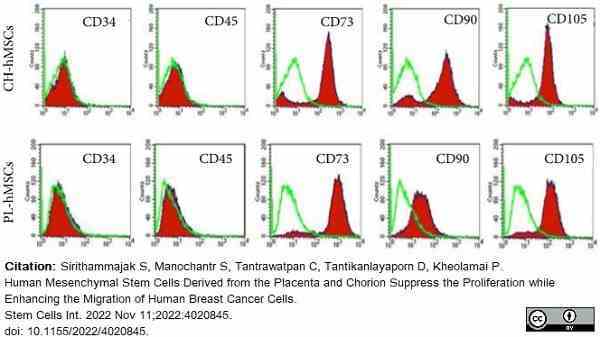
Sirithammajak, S. et al. (2022) Human Mesenchymal Stem Cells Derived from the Placenta and Chorion Suppress the Proliferation while Enhancing the Migration of Human Breast Cancer Cells.
Stem Cells Int. 2022: 4020845. -
Arenal, Á. et al. (2022) Effects of Cardiac Stem Cell on Postinfarction Arrhythmogenic Substrate.
Int J Mol Sci. 23 (24): 16211. -
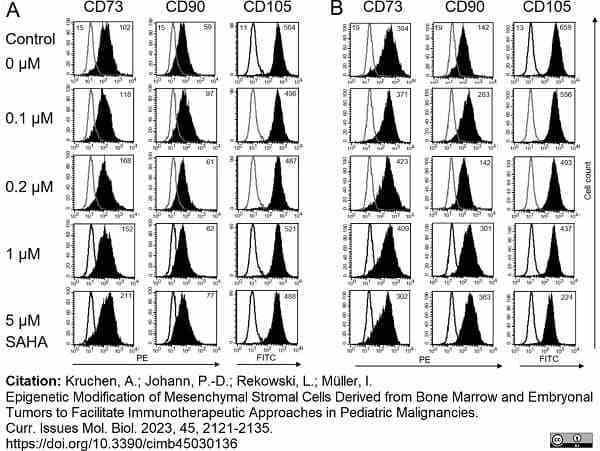
Kruchen, A. et al. (2023) Epigenetic Modification of Mesenchymal Stromal Cells Derived from Bone Marrow and Embryonal Tumors to Facilitate Immunotherapeutic Approaches in Pediatric Malignancies.
Curr Issues Mol Biol. 45 (3): 2121-35. -
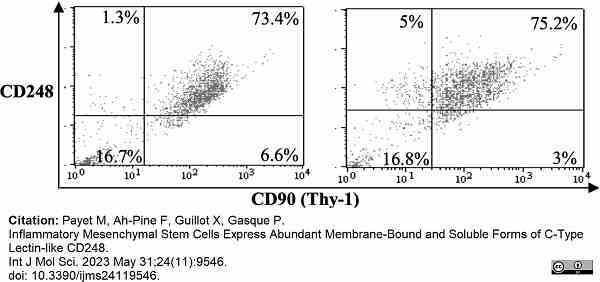
Payet, M. et al. (2023) Inflammatory Mesenchymal Stem Cells Express Abundant Membrane-Bound and Soluble Forms of C-Type Lectin-like CD248.
Int J Mol Sci. 24 (11): 9546. -
Tiraihi, T. et al. (2023) A Sequential Culturing System for Generating Epithelial-Like Stem Cells from Human Mesenchymal Stem Cells Derived from Adipose Tissue
Cell Tissue Biol. 17 (6): 639-52. -
Tripathy, N.K. et al. (2018) Cardiomyogenic Heterogeneity of Clonal Subpopulations of Human Bone Marrow Mesenchymal Stem Cells.
J Stem Cells Regen Med. 14 (1): 27-33.
- Synonyms
- THY1
- RRID
- AB_871984
- UniProt
- P04216
- Entrez Gene
- THY1
- GO Terms
- GO:0001525 angiogenesis
- GO:0007010 cytoskeleton organization
- GO:0005783 endoplasmic reticulum
- GO:0005100 Rho GTPase activator activity
- GO:0005887 integral to plasma membrane
- GO:0005178 integrin binding
- GO:0006469 negative regulation of protein kinase activity
- GO:0016337 cell-cell adhesion
- GO:0030336 negative regulation of cell migration
- View More GO Terms
- GO:0030426 growth cone
- GO:0034235 GPI anchor binding
- GO:0043547 positive regulation of GTPase activity
- GO:0045121 membrane raft
- GO:0046549 retinal cone cell development
- GO:0048041 focal adhesion assembly
- GO:0050771 negative regulation of axonogenesis
- GO:0050852 T cell receptor signaling pathway
- GO:0050860 negative regulation of T cell receptor signaling pathway
- GO:0050870 positive regulation of T cell activation
- GO:0051281 positive regulation of release of sequestered calcium ion into cytosol
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Human ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up