CD49f antibody | 450-30A
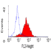
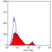
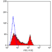
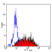

Mouse anti Human CD49f
- Product Type
- Monoclonal Antibody
- Clone
- 450-30A
- Isotype
- IgG1
- Specificity
- CD49f
| Mouse anti Human CD49f antibody, clone 450-30A recognizes the human VLA-6 cell surface antigen, also known as the alpha 6 integrin and as CD49f. CD49f is expressed by platelets, weakly by monocytes and by a subset of lymphocytes. CD49f is also widely expressed on epithelial tissues. |
- Target Species
- Human
- Product Form
- Purified IgG - liquid
- Preparation
- MCA1457, MCA1457T: Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- MCA1457GA: Purified IgG prepared by affinity chromatography on Protein G from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% sodium azide (NaN3)
- Carrier Free
- Yes
- Immunogen
- alpha 6 beta 4 integrin purified from A431 cells.
- Approx. Protein Concentrations
- IgG concentration 1.0 mg/ml
- Fusion Partners
- Spleen cells from immunized BALB/c mice were fused with cells of the mouse SP2/0 myeloma cell line.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
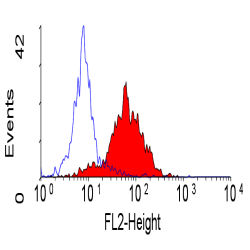
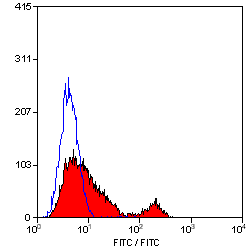
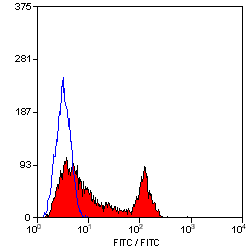
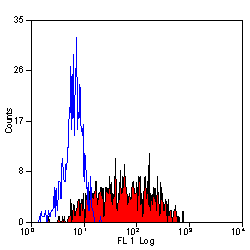
| Flow Cytometry | 1/10 | 1/50 | |
| Immunohistology - Frozen 1 | 1/50 | 1/100 | |
| Immunohistology - Paraffin | |||
| Western Blotting |
- 1The epitope recognised by this antibody is reported to be sensitive to formaldehyde fixation and tissue processing. Bio-Rad recommends the use of acetone fixation for frozen sections.
- Flow Cytometry
- Use 10μl of the suggested working dilution to label 106 cells in 100μl
- Histology Positive Control Tissue
- Human tonsil
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG1 Negative Control | MCA928 | F | 100 Tests |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG1 Negative Control | ||||||
References for CD49f antibody
-
Kennel, S.J. et al. (1990) Second generation monoclonal antibodies to the human integrin alpha 6 beta 4.
Hybridoma. 9 (3): 243-55. -
Cavers, M. et al. (2002) Differential expression of beta1 and beta2 integrins and L-selectin on CD4+ and CD8+ T lymphocytes in human blood: comparative analysis between isolated cells, whole blood samples and cryopreserved preparations.
Clin Exp Immunol. 127: 60-5. -
Liu, L. et al. (2003) Priming of eosinophil migration across lung epithelial cell monolayers and upregulation of CD11b/CD18 are elicited by extracellular Ca2+.
Am J Respir Cell Mol Biol. 28: 713-21. -
Maurice, S. et al. (2007) Isolation of progenitor cells from cord blood using adhesion matrices.
Cytotechnology. 54: 121-33. -
Kaczmarek, M. et al. (2011) Evaluation of the phenotype pattern of macrophages isolated from malignant and non-malignant pleural effusions.
Tumour Biol. 32: 1123-32. -
López, J. et al. (2012) Cancer-initiating cells derived from established cervical cell lines exhibit stem-cell markers and increased radioresistance.
BMC Cancer. 12: 48. -
Keller, P.J. et al. (2012) Defining the cellular precursors to human breast cancer.
Proc Natl Acad Sci U S A. 109: 2772-7. -
Aldridge, V. et al. (2012) Human mesenchymal stem cells are recruited to injured liver in a β1-integrin and CD44 dependent manner.
Hepatology. 56 (3): 1063-73.
View The Latest Product References
-
Steinberg, F. et al. (2012) SNX17 protects integrins from degradation by sorting between lysosomal and recycling pathways.
J Cell Biol. 197 (2): 219-30. -
Goyer, B. et al. (2018) Extracellular Matrix and Integrin Expression Profiles in Fuchs Endothelial Corneal Dystrophy Cells and Tissue Model.
Tissue Eng Part A. 24 (7-8): 607-15. -
Amirian, M. et al. (2022) VASA protein and gene expression analysis of human non-obstructive azoospermia and normal by immunohistochemistry, immunocytochemistry, and bioinformatics analysis.
Sci Rep. 12 (1): 17259. -
Hashemi Karoii, D. et al. (2023) Altered G-Protein Transduction Protein Gene Expression in the Testis of Infertile Patients with Nonobstructive Azoospermia.
DNA Cell Biol. 42 (10): 617-37.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Human ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up