CD206 antibody | 15-2
Mouse anti Human CD206
- Product Type
- Monoclonal Antibody
- Clone
- 15-2
- Isotype
- IgG1
- Specificity
- CD206
Filter by Application:
IF Reset| Mouse anti Human CD206 monoclonal antibody, clone 15-2 recognizes human macrophage mannose receptor 1, also known as CD206 or C-type lectin domain family 13 member D-like. CD206 is a ~175 kDa single pass type I transmembrane glycoprotein belonging to the group of pattern recognition receptors (Paveley et al. 2011). CD206 has multiple carbohydrate recognition motifs and acts as a receptor for bacteria, fungi and other pathogens (Ezekowitz et al. 1990). CD206 is predominantly expressed in tissue macrophages and dendritic cells (Engering et al. 1997) and can also be found in a subpopulation of endothelial cells (Pack et al. 2007) and sperm cells (Cardona-Maya et al. 2006). CD206 can also be detected in a soluble form in human plasma and is elevated in patients with acute sepsis (Rødgaard-Hansen et al. 2013). Mouse anti CD206, clone 15-2 has been used extensively to monitor mannose receptor modulation in macrophages treated with a wide range of cytokines and growth factors (Chang et al. 2004) and to indicate CD206 as a marker for alternative activation of macrophages (Joerink et al. 2011). |
- Target Species
- Human
- Product Form
- Purified IgG - liquid
- Preparation
- Antibody purified from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- < 0.1% sodium azide (NaN3)
- Immunogen
- Purified human mannose receptor.
- Approx. Protein Concentrations
- IgG concentration 0.5 mg/ml
- Fusion Partners
- Spleen cells from immunised Balb/c mice where fused with cells of the SP2/0 Ag.14 mouse myeloma cell line.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | |||
| Immunofluorescence | |||
| Immunohistology - Frozen | |||
| Western Blotting 1 |
- 1Clone 15-2 recognises a protein of approximately 175kDa under non-reducing conditions.
- Flow Cytometry
- Use 10μl of the suggested working dilution to label 106 cells in 100μl
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG1 Negative Control | MCA928 | F | 100 Tests |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG1 Negative Control | ||||||
Source Reference
-
Barrett-Bergshoeff, M. et al. (1997) Monoclonal antibodies against the human mannose receptor that inhibit the binding of tissue-type plasminogen activator.
Thromb Haemost. 77: 718-24.
Antibody Characterization Reference
-
Noorman, F. et al. (1997) Monoclonal antibodies against the human mannose receptor as a specific marker in flow cytometry and immunohistochemistry for macrophages.
J Leukoc Biol. 61 (1): 63-72.
References for CD206 antibody
-
Koning, N. et al. (2009) Distribution of the immune inhibitory molecules CD200 and CD200R in the normal central nervous system and multiple sclerosis lesions suggests neuron-glia and glia-glia interactions.
J Neuropathol Exp Neurol. 68: 159-67. -
Emara, M. et al. (2011) Recognition of the major cat allergen Fel d 1 through the cysteine-rich domain of the mannose receptor determines its allergenicity.
J Biol Chem. 286:13033-40. -
Chang, S.K. et al. (2008) B lymphocyte stimulator regulates adaptive immune responses by directly promoting dendritic cell maturation.
J Immunol. 180: 7394-403. -
MacKinnon, A.C. et al. (2008) Regulation of alternative macrophage activation by galectin-3.
J Immunol. 180: 2650-8. -
Lai, W.K. et al. (2006) Expression of DC-SIGN and DC-SIGNR on human sinusoidal endothelium: a role for capturing hepatitis C virus particles.
Am J Pathol. 169: 200-8. -
Ueno, N. et al. (2009) Differences in human macrophage receptor usage, lysosomal fusion kinetics and survival between logarithmic and metacyclic Leishmania infantum chagasi promastigotes.
Cell Microbiol. 11: 1827-41. -
Kato, M. et al. (2000) Expression of multilectin receptors and comparative FITC-dextran uptake by human dendritic cells.
Int Immunol. 12:1511-9. -
Chang, Y.C. et al. (2004) Modulation of macrophage differentiation and activation by decoy receptor 3.
J Leukoc Biol. 75: 486-94.
View The Latest Product References
-
Yamamoto, H. et al. (2011) Sphingosylphosphorylcholine and lysosulfatide have inverse regulatory functions in monocytic cell differentiation into macrophages.
Arch Biochem Biophys. 506: 83-91. -
He, L.Z. et al. (2007) Antigenic targeting of the human mannose receptor induces tumor immunity.
J Immunol. 178: 6259-67. -
Sturge, J. et al. (2007) Mannose receptor regulation of macrophage cell migration.
J Leukoc Biol. 82: 585-93. -
Torrelles, J.B. et al. (2006) Fine discrimination in the recognition of individual species of phosphatidyl-myo-inositol mannosides from Mycobacterium tuberculosis by C-type lectin pattern recognition receptors.
J Immunol. 177 (3): 1805-16. -
Larsson, K. et al. (2015) COX/mPGES-1/PGE2 pathway depicts an inflammatory-dependent high-risk neuroblastoma subset.
Proc Natl Acad Sci U S A. 112 (26): 8070-5. -
Stankevich, K.S. et al. (2015) Surface modification of biomaterials based on high-molecular polylactic acid and their effect on inflammatory reactions of primary human monocyte-derived macrophages: perspective for personalized therapy.
Mater Sci Eng C Mater Biol Appl. 51: 117-26. -
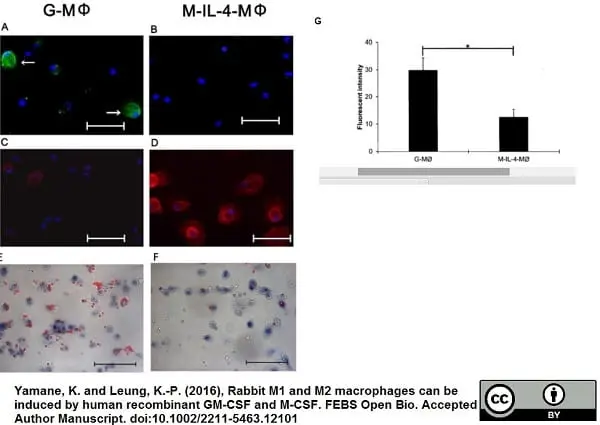
Yamane, K. & Leung, K.P. (2016) Rabbit M1 and M2 macrophages can be induced by human recombinant GM-CSF and M-CSF.
FEBS Open Bio. 6 (9): 945-53. -
Karna, S.L. et al. (2016) RNA-Seq Transcriptomic Responses of Full-Thickness Dermal Excision Wounds to Pseudomonas aeruginosa Acute and Biofilm Infection.
PLoS One. 11 (10): e0165312. -
Özçelik H et al. (2015) Harnessing the multifunctionality in nature: a bioactive agent release system with self-antimicrobial and immunomodulatory properties.
Adv Healthc Mater. 4 (13): 2026-36. -
Argueta-Donohué J et al. (2016) Differential phagocytosis of Leishmania mexicana promastigotes and amastigotes by monocyte-derived dendritic cells.
Microbiol Immunol. 60 (6): 369-81.
- Synonyms
- Mannose Receptor C Type 1
- RRID
- AB_323520
- UniProt
- P22897
- Entrez Gene
- MRC1
- GO Terms
- GO:0004872 receptor activity
- GO:0005887 integral to plasma membrane
- GO:0005537 mannose binding
- GO:0006898 receptor-mediated endocytosis
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Human ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up