AMH antibody | 5/6
Mouse anti Human AMH
- Product Type
- Monoclonal Antibody
- Clone
- 5/6
- Isotype
- IgG1
- Specificity
- AMH
Filter by Application:
P WB Reset| Mouse anti Human AMH, clone 5/6 recognizes human anti-mullerian hormone (AMH), originally classified as a foetal testicular hormone that inhibits Mullerian duct development. AMH is expressed post-natally by immature Sertoli cells, and to a lesser degree by granulosa cells. AMH plays a role in testicular differentiation and in the regulation of ovarian follicle growth. AMH is a member of the TGF beta superfamily. It is secreted as a homodimeric ~140 kDa disulphide linked precursor that is cleaved to release the mature ~30 kDa homodimer. |
- Target Species
- Human
- Species Cross-Reactivity
-
Target Species Cross Reactivity Mouse Sheep Squirrel monkey Baboon Rat - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Concentrated tissue culture supernatant - liquid
- Preparation
- Concentrated Tissue Culture Supernatant containing 0.1M Tris/HCl pH7.4 and 5-10% foetal calf serum.
- Preservative Stabilisers
- 0.1% sodium azide (NaN3)
- Immunogen
- Synthetic peptide derived from human AMH (VPTAYAGKLLISLSEERISAHHVPNMVATEC)
- Fusion Partners
- Spleen cells from immunised T/O outbred mice were fused with cells of the SP2/0 myeloma cell line.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Immunohistology - Paraffin 1 | 1/20 | 1/40 | |
| Western Blotting |
- 1This product requires antigen retrieval using heat treatment prior to staining of paraffin sections.Sodium citrate buffer pH 6.0 is recommended for this purpose.
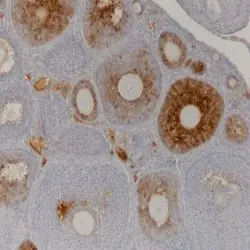
- Histology Positive Control Tissue
- human ovary
References for AMH antibody
-
Van Saen, D. et al. (2010) Meiotic activity in orthotopic xenografts derived from human postpubertal testicular tissue.
Hum Reprod. 26: 282-93. -
Gruijters, M.J et al. (2003) Anti-Müllerian hormone and its role in ovarian function.
Mol Cell Endocrinol. 211 (1-2): 85-90. -
Weenen, C. et al. (2004) Anti-Mullerian hormone expression pattern in the human ovary: potential implications for initial and cyclic follicle recruitment.
Mol Hum Reprod10: 77-83. -
Papanastasopoulos, P. et al. (2009) A case of complete androgen insensitivity syndrome presenting with incarcerated inguinal hernia: an immunohistochemical study.
Fertil Steril. 92: 1169.e11-4. -
Campbell, B.K. (2009) The endocrine and local control of ovarian follicle development in the ewe
Anim. Reprod. 6:159-71 -
Walker, M.L. et al. (2009) Ovarian aging in squirrel monkeys (Saimiri sciureus).
Reproduction. 138: 793-9. -
Sobinoff, A.P. et al. (2011) Understanding the villain: DMBA induced pre-antral ovotoxicity involves selective follicular destruction and primordial follicle activation through PI3K/Akt and mTOR signalling.
Toxicol Sci. 123: 563-75. -
Van Saen, D. et al. (2011) Can pubertal boys with Klinefelter syndrome benefit from spermatogonial stem cell banking?
Hum Reprod. 27: 323-30.
View The Latest Product References
-
David, A. et al. (2012) Effect of cryopreservation and transplantation on the expression of kit ligand and anti-Müllerian hormone in human ovarian tissue
Hum Reprod. 27: 1088-95. -
Kevenaar, M.E. et al. (2006) Serum anti-mullerian hormone levels reflect the size of the primordial follicle pool in mice.
Endocrinology. 147: 3228-34. -
Campbell, B.K. et al. (2012) The role of anti-Müllerian hormone (AMH) during follicle development in a monovulatory species (sheep).
Endocrinology. 153: 4533-43. -
Amorim, C.A. et al. (2013) Successful vitrification and autografting of baboon (Papio anubis) ovarian tissue.
Hum Reprod. 28: 2146-56. -
Parlakgumus, H.A. et al. (2015) GNRH agonists and antagonists in rescue for cyclophosphamide-induced ovarian damage: friend or foe?
Arch Gynecol Obstet. 291 (6): 1403-10. -
Themmen, A.P.N. et al. (2016) The use of anti-Müllerian hormone as diagnostic for gonadectomy status in dogs.
Theriogenology. 86 (6): 1467-74. -
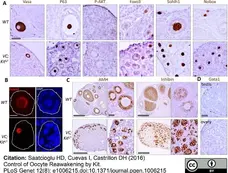
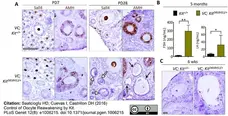
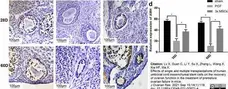
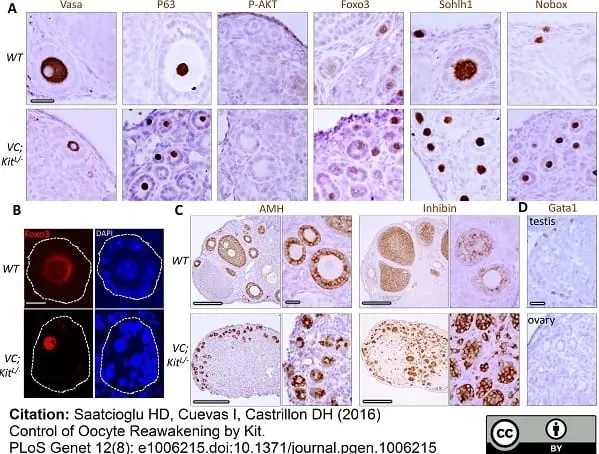
Saatcioglu, H.D. et al. (2016) Control of Oocyte Reawakening by Kit.
PLoS Genet. 12 (8): e1006215. -
Calvopina, J.H. et al. (2015) The Aorta-Gonad-Mesonephros Organ Culture Recapitulates 5hmC Reorganization and Replication-Dependent and Independent Loss of DNA Methylation in the Germline.
Stem Cells Dev. 24 (13): 1536-45. -
Camlin, N.J. et al. (2016) Maternal Smoke Exposure Impairs the Long-Term Fertility of Female Offspring in a Murine Model.
Biol Reprod. 94 (2): 39. -
Ohta, K. et al. (2012) Male differentiation of germ cells induced by embryonic age-specific Sertoli cells in mice.
Biol Reprod. 86 (4): 112. -
Bazzano, M.V. et al. (2015) Obesity induced by cafeteria diet disrupts fertility in the rat by affecting multiple ovarian targets.
Reprod Biomed Online. 31 (5): 655-67. -
Díaz, P.U. et al. (2018) Altered Expression of Anti-Müllerian Hormone during the Early Stage of Bovine Persistent Ovarian Follicles.
J Comp Pathol. 158: 22-31. -
Cacciottola, L. et al. (2020) Long-Term Advantages of Ovarian Reserve Maintenance and Follicle Development Using Adipose Tissue-Derived Stem Cells in Ovarian Tissue Transplantation.
J Clin Med. 9 (9)Sep 15 [Epub ahead of print]. -
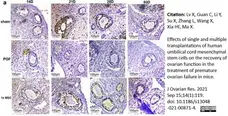
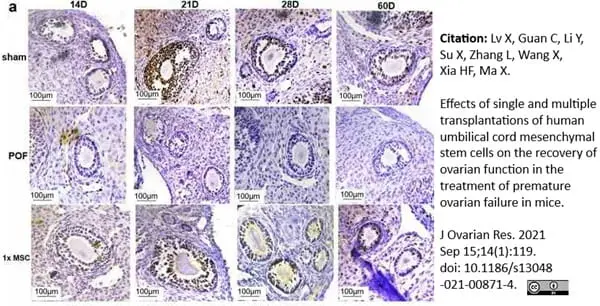
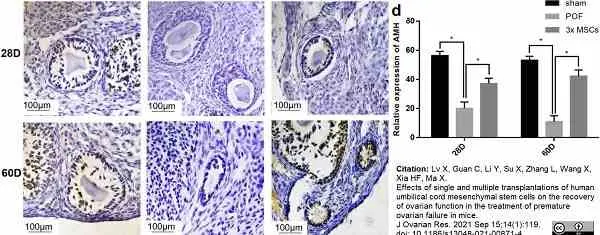
Lv, X. et al. (2021) Effects of single and multiple transplantations of human umbilical cord mesenchymal stem cells on the recovery of ovarian function in the treatment of premature ovarian failure in mice.
J Ovarian Res. 14 (1): 119. -
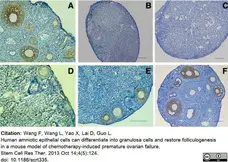
Wang, F. et al. (2013) Human amniotic epithelial cells can differentiate into granulosa cells and restore folliculogenesis in a mouse model of chemotherapy-induced premature ovarian failure.
Stem Cell Res Ther. 4 (5): 124. -
de Michele, F. et al. (2018) In vitro. formation of the blood-testis barrier during long-term organotypic culture of human prepubertal tissue: comparison with a large cohort of pre/peripubertal boys.
Mol Hum Reprod. 24 (5): 271-82. -
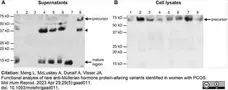
Meng, L. et al. (2023) Functional analysis of rare anti-Müllerian hormone protein-altering variants identified in women with PCOS.
Mol Hum Reprod. 29 (5): gaad011. -
Amorim, C.A. et al. (2019) Long-term follow-up of vitrified and autografted baboon (Papio anubis) ovarian tissue.
Hum Reprod. 34 (2): 323-334. -
Van Saen, D. et al. (2020) Characterization of the stem cell niche components within the seminiferous tubules in testicular biopsies of Klinefelter patients.
Fertil Steril. 113 (6): 1183-95.e3. -
Sarrible, G.B. et al. (2025) Effects of coenzyme q10 supplementation on metabolic and reproductive outcomes in obese rats.
J Ovarian Res. 18 (1): 22. -
Alvaro Mercadal, B. et al. (2015) AMH mutations with reduced in vitro bioactivity are related to premature ovarian insufficiency.
Hum Reprod. 30 (5): 1196-202. -
Parlakgumus, H.A. et al. (2014) Atorvastatin for ovarian torsion: effects on follicle counts, AMH, and VEGF expression.
Eur J Obstet Gynecol Reprod Biol. 175: 186-90. -
Hervet, C. et al. (2025) Differential impact of porcine reproductive and respiratory virus and swine Influenza A virus infections on respiratory Lymph Nodes B cells and macrophages.
Mol Immunol. 188: 98-110.
- Synonyms
- anti Mullerian Hormone
- RRID
- AB_2226471
- UniProt
- P03971
- Entrez Gene
- AMH
- GO Terms
- GO:0001880 Mullerian duct regression
- GO:0005179 hormone activity
- GO:0005615 extracellular space
- GO:0007267 cell-cell signaling
- GO:0007506 gonadal mesoderm development
- GO:0007530 sex determination
- GO:0008083 growth factor activity
- GO:0030154 cell differentiation
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Human ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up